Protein Design and Engineering
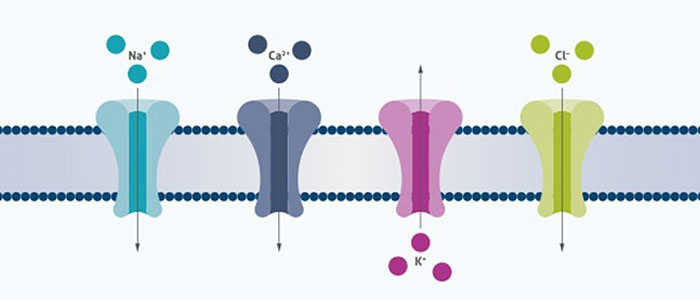
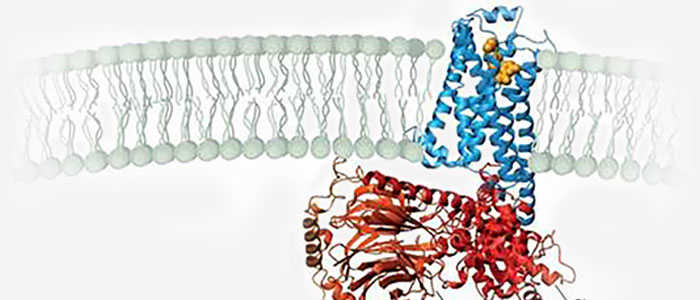
Based upon our protein engineering expertise, we can modulate and optimize challenging protein properties such as activity, affinity, selectivity, stability, resistance to proteolytic degradation, and even improve protein expression and solubility.
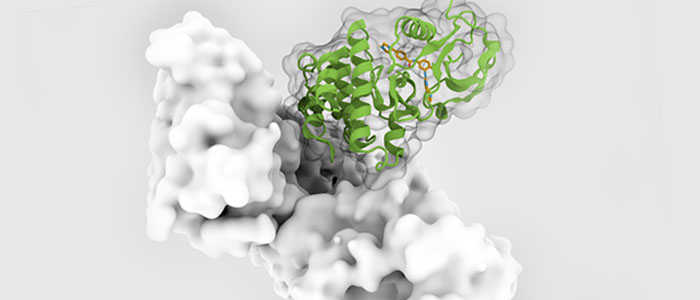
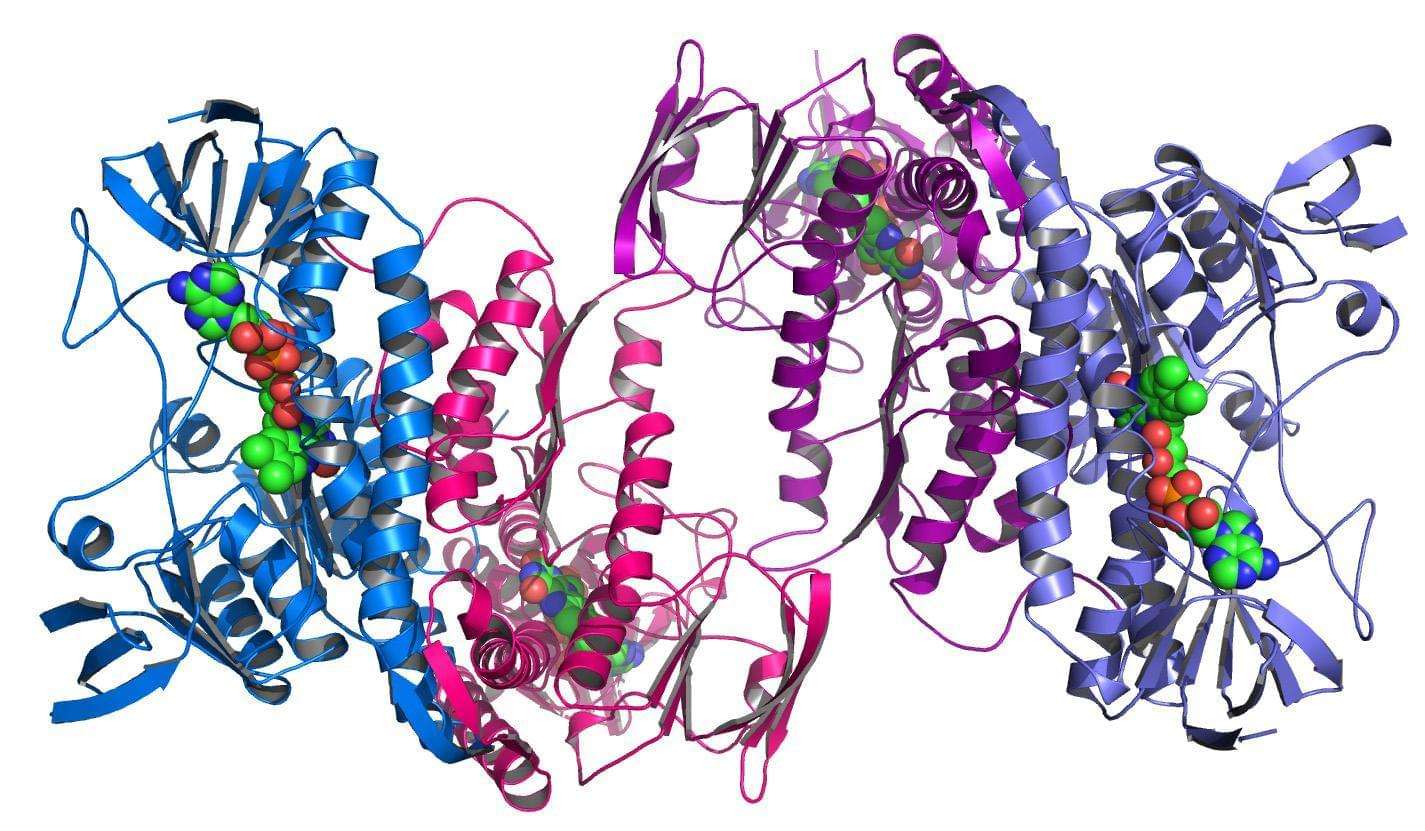
Protein design refers to the ability to alter protein structure to achieve the desired protein function. In order to increase the solubility, stability or ability to crystallize of a given protein, we can use a protein compaction technology based on the deletion of the protein's non-essential floppy loops. Of the two classes of proteins (binding proteins and catalytic proteins), catalytic proteins, or so-called enzymes, are of particular importance to chemical processing because they can be used as chemical catalysts.
Many successful examples using rational design to engineer enzyme stability or activity have been reported. One of them is the above-mentioned redesign of a T4 lysozyme with increased stability by engineering disulfide bonds into the protein. Another notable example is the design of a subtilisin BPN' mutant containing six site-directed mutations, which was 100-fold more stable than the wild-type enzyme in an aqueous environment and 50-fold more stable than the wild-type in anhydrous dimethylformamide.
This approach has been applied to both viral antigens for vaccines development and anti-bacterial targets for drug discovery applications. The resulting functional mini-protein will display a better therapeutic value (therapeutic proteins, antigens) or a superior ability to crystallize (target proteins).
Contact us or send an email at for project quotations and more detailed information.
Quick Links
-

Papers’ PMID to Obtain Coupon
Submit Now -

Refer Friends & New Lab Start-up Promotions