Neurotrophin Family
Available Resources for the Study of Neurotrophin Family
At Creative BioMart, our focus is on driving advancements in neurotrophin family research. We are dedicated to providing researchers with the latest tools and knowledge on neurotrophin receptors and ligands.
- Our product offerings are diverse and essential for gaining a deep understanding of the functions and mechanisms of the neurotrophin family. From recombinant proteins to pre-coupled magnetic beads, cell and tissue lysates, and more, we offer a comprehensive range of products to support research efforts.
- Our team of experts brings a wealth of experience in neurotrophin family research and is committed to developing customized solutions to meet the specific needs of researchers. Additionally, we provide a wealth of resources on involved pathways, protein function, interacting proteins, and other valuable information to support research endeavors and maximize their impact.
Our Featured Products
About Neurotrophin Family
The neurotrophin family consists of a group of proteins called neurotrophins and their corresponding receptors. The neurotrophin family consists of a group of proteins that play critical roles in the development, survival, and function of neurons. This family includes neurotrophin ligands such as nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), neurotrophin-3 (NT-3), and neurotrophin-4 (NT-4). These neurotrophins interact with specific receptors known as neurotrophin receptors, which include Trk receptors (TrkA, TrkB, and TrkC) and the p75 neurotrophin receptor (p75NTR).
Neurotrophin ligands bind to their respective receptors, initiating downstream molecular signaling pathways that regulate neuronal survival, differentiation, and synaptic plasticity. The Trk receptors are receptor tyrosine kinases (RTKs) that activate intracellular signaling cascades upon ligand binding. The p75NTR receptor, on the other hand, can act as a co-receptor and modulate signaling strength and specificity.
These interactions between neurotrophin ligands and their receptors are involved in various biological processes, including neuronal development, axonal growth, synaptic plasticity, and maintenance of neuronal survival and function. Dysregulation of the neurotrophin signaling system has been associated with several neurological disorders, making the study of the neurotrophin family and its receptors of great importance in neuroscience research.
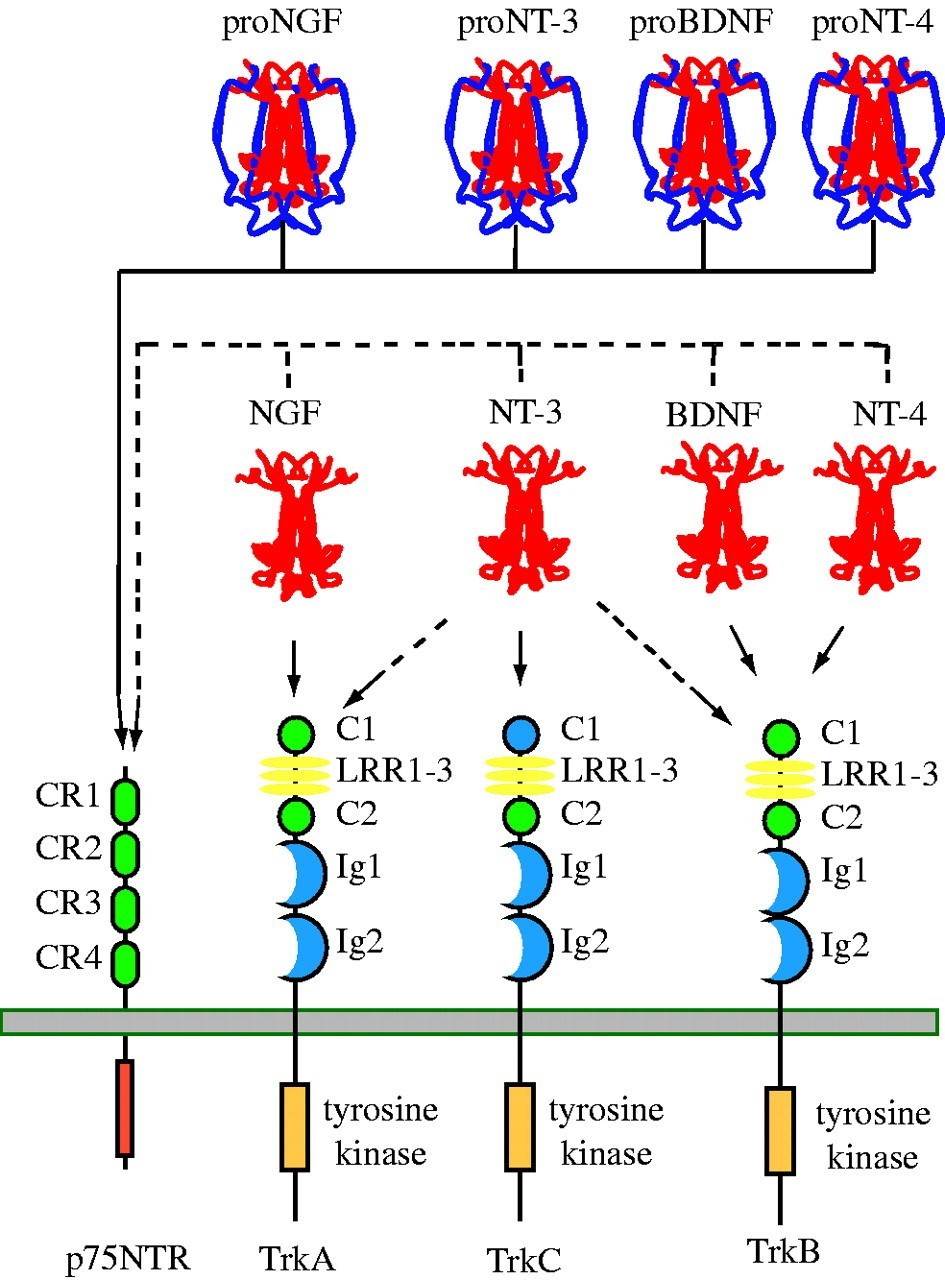 Fig.1 Neurotrophin–receptor interactions. (Reichardt L F, 2006)
Fig.1 Neurotrophin–receptor interactions. (Reichardt L F, 2006)
Neurotrophin Family in Disease
- Alzheimer's Disease: Alzheimer's disease is characterized by the accumulation of amyloid-beta plaques and neurofibrillary tangles in the brain. Studies have shown that the expression and signaling of neurotrophin receptors, particularly TrkA and TrkB, are reduced in the brains of individuals with Alzheimer's disease. Additionally, decreased levels of neurotrophin ligands, such as NGF and BDNF, have been observed in the affected brain regions. The impairment of neurotrophin signaling contributes to synaptic dysfunction, neuronal loss, and cognitive decline associated with Alzheimer's disease.
- Parkinson's Disease: In Parkinson's disease, there is degeneration of dopaminergic neurons in the substantia nigra region of the brain. Alterations in neurotrophin signaling have been implicated in the pathogenesis of Parkinson's disease. Reduced levels of neurotrophin ligands, including GDNF (glial cell line-derived neurotrophic factor), have been observed in the brains of Parkinson's patients. The downregulation of neurotrophin receptors, such as TrkB, can lead to impaired neuronal survival and function, contributing to the development and progression of the disease.
- Major Depressive Disorder: Major depressive disorder (MDD) is characterized by persistent feelings of sadness, loss of interest, and impaired functioning. Dysregulation of neurotrophin signaling, particularly BDNF, has been implicated in the pathophysiology of depression. Reduced levels of BDNF and alterations in TrkB receptor expression have been observed in individuals with MDD. The disruption of neurotrophin signaling pathways in key brain regions involved in mood regulation can contribute to the development and persistence of depressive symptoms.
- Peripheral Neuropathies: Peripheral neuropathies, such as diabetic neuropathy and chemotherapy-induced peripheral neuropathy, are characterized by nerve damage and sensory disturbances. Altered expression of neurotrophin receptors and ligands has been implicated in these conditions. Reduced levels of neurotrophin ligands, including NGF and NT-3, and alterations in Trk receptor expression have been observed in the peripheral nerves of individuals with neuropathies. Restoring the balance of neurotrophin signaling may have therapeutic potential in promoting nerve regeneration and alleviating symptoms associated with peripheral neuropathies.
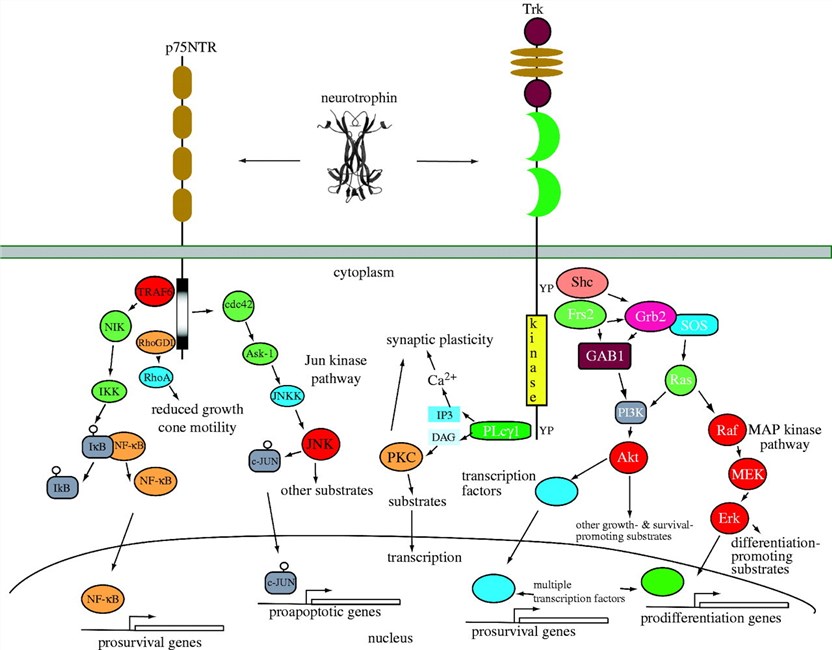 Fig.2 Neurotrophin signalling. (Reichardt L F, 2006)
Fig.2 Neurotrophin signalling. (Reichardt L F, 2006)
Understanding the dysregulation of neurotrophin receptors and ligands in disease provides insights into the underlying mechanisms and potential therapeutic targets. Developing strategies to modulate neurotrophin signaling, either by enhancing ligand availability, promoting receptor expression, or targeting downstream signaling pathways, holds promise for the development of novel treatments for these diseases and disorders.
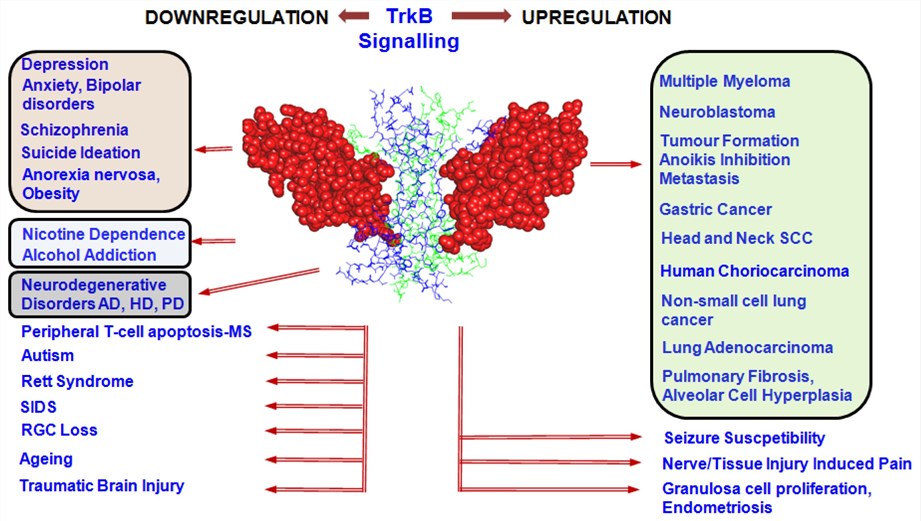 Fig.3 TrkB signalling alterations in various disease conditions. (Gupta VK, et al., 2013)
Fig.3 TrkB signalling alterations in various disease conditions. (Gupta VK, et al., 2013)
An increase (right) or decrease (left) of TrkB expression or its signalling may lead to a range of pathological conditions indicating the significance of the maintenance of a fine equilibrium of the TrkB signalling pathway. TrkB (space-filled)-BDNF (wire-frame) dimeric complex. AD, Alzheimer's Disease; HD, Huntington's Disease; PD, Parkinson's Disease; SIDS, Sudden Infant Death Syndrome; RGC, Retinal Ganglion Cell; MS, Multiple Sclerosis; SCC, Squamous Cell Carcinoma.
We are committed to helping you achieve your scientific goals and make meaningful contributions to research on the roles of the various components of the neurotrophin family and their role in disease. Contact us today to learn more about our products and resources.
References:
- Chao M V. Neurotrophins and their receptors: a convergence point for many signalling pathways[J]. Nature Reviews Neuroscience, 2003, 4(4): 299-309.
- Wong J. Neurotrophin Signaling and Alzheimer's Disease Neurodegeneration− Focus on BDNF/TrkB Signaling[J]. Trends in cell signaling pathways in neuronal fate decision, 2013: 181-194.
- Reichardt L F. Neurotrophin-regulated signalling pathways[J]. Philosophical Transactions of the Royal Society B: Biological Sciences, 2006, 361(1473): 1545-1564.
- Gupta VK, You Y, Gupta VB, Klistorner A, Graham SL. TrkB Receptor Signalling: Implications in Neurodegenerative, Psychiatric and Proliferative Disorders. International Journal of Molecular Sciences. 2013; 14(5):10122-10142.


