-
- ExosomesVirus-like Nanoparticles (VNPs)
- LiposomesHeparosanCyclodextrins
- Polymeric NanocarrierPolymer-Drug ConjugatesHydrogelResponsive Polymer
- Inquiry


- Home
- Biopolymer Drug Carriers Design
- Cyclodextrins
Cyclodextrins
Cyclodextrins (CDs) are a family of cyclic oligosaccharides with hydrophilic outer surface and lipophilic cavity. They possess a unique ability to act as molecular containers by entrapping drugs in their internal cavity. No covalent bonds are formed or broken during drug CD complex formation, therefore serving as a convenient route to increase aqueous solubility of poorly soluble drugs, and also to increase their bioavailability and stability. In general, the ability of natural cyclodextrins and their hydrophilic derivatives to entrap guest molecules can be modulated by their cavity size and substituent group on the sugar ring. With our strong expertise in cyclodextrin-based nanotechnology, Creative BioMart continues contributing to the development of novel drug delivery systems. Currently there are more than 50 different pharmaceutical products worldwide containing drug/cyclodextrin complexes on the market. Below table lists some of the cyclodextrin-based carriers that have been used for anticancer drugs.
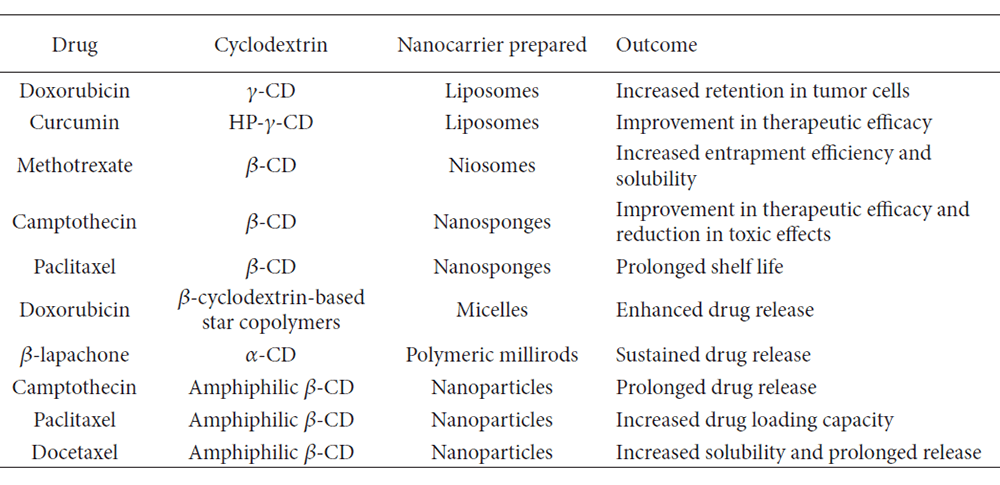
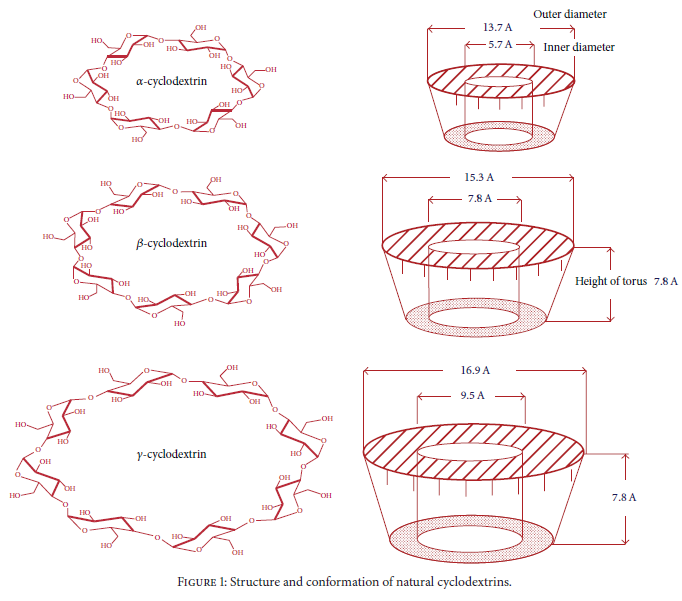 Structure and conformation of natural cyclodextrins. Reprinted from Gidwani, Bina, et al. BioMed research international 2015.
Structure and conformation of natural cyclodextrins. Reprinted from Gidwani, Bina, et al. BioMed research international 2015.
-



