Protein expression System
Introduction
Protein expression is a complicated process performing by cells, including how proteins are synthesized, modified and regulated. With the development of genetic engineering, it is easier to control and regulate protein expression and more convenient to get heterologous proteins or recombinant proteins with high purity in large scale. The system in which the protein is expressed plays a key role to get proteins in large scale. So, the protein expression system is required to be easy to culture and maintain, and must grow rapidly and produce large amounts of protein. In addition, many researchers also need proteins or recombinant proteins expressed with proper modification. There are three common used protein expression systems including prokaryotic cell (usual bacteria like E.coli), yeast, insect or mammalian cell expression systems.
Many factors may affect which expression system to choose, including the following:
- How long will spent in expressing the protein
- Easy to handle
- The amount or mass of protein you need
- Post-translational modifications
- Destination of the expressed protein
Brief steps
For recombinant protein expression, the following items are required:
- Preparation of gene that encodes the protein which you are interested
- Generation of cDNA from the respective mRNA
- Selection of suitable expression vector
- Construction the recombinant vector by inserting gene sequence
- Suitable protein expression system to express vector
- Appropriate methods for screening and scaling up best host
The core steps to produce a satisfied recombinant protein are similar in almost all the expression systems as following figure shows.
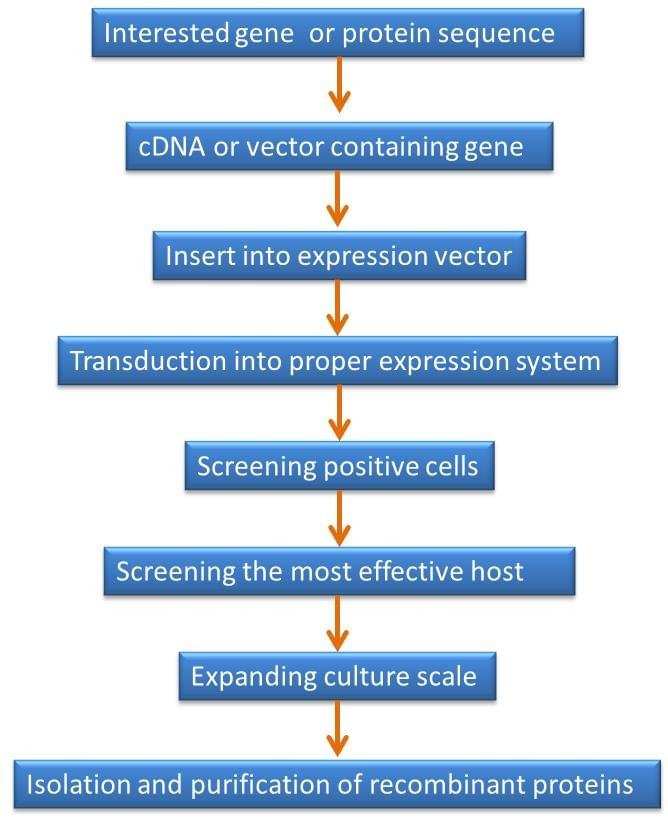
Figure 1. Brief steps for optimizing production of recombinant protein.
Prokaryotic cell expression systems
Bacteria are used to express recombinant proteins for the rapid growth with short doubling time.
It is easy to culture bacteria and the cost is relatively low. E. coli is most common used prokaryotic cell expression systems for its abundant knowledge about its genetics, genome sequence, safety and physiology. However, the E. coli contains toxic pyrogens in its cell membrane and is lack of posttranslational modification. E. coli also cannot release protein production into medium. Thus, production of E. coli system must be tested carefully before application.
Yeast expression systems
Yeasts are able to carry specifically designed plasmids, which makes it easy to construct recombinant vectors. The restriction sites in the plasmid can be used to insert the gene sequence of interest. Yeast also can perform part of posttranslational modifications and can be appropriately scaled up. Saccharomyces cerevisiae and Pichia pastoris are most common used yeast expression systems for their highly developed genetic system, ease of use and lower cost. More important, S. cerevisiae is thought to be safe by FDA.
Mammalian cell expression systems
The mammalian cell expression systems can provide the same modification of proteins in human cells and can secrete protein out of cells. The bioactivities of proteins produced by mammalian cell are almost the same as natural protein. However, the reduced efficiency and levels of the protein expressed in mammalian cell are not satisfied. For commercial application, HEK293 is developed as transient expression system and CHO is developed as stable expression system. CHO cells are commonly used for stable expression. However, the selection and expansion process is relatively complicated and it may take 2-3 months.
The characters of the above three protein expression systems are summarized in Table 1.
| expression system | Protein Yield | Protein Activity | Cell Stability | Posttranslational Modification | Cost |
| E. coli | High | Low | High | Not performed | Low |
| Yeast | High | Middle | High | Part performed | Low |
| Mammalian cell | Low | High | Low | Whole performed | High |

