Immunoglobulins superfamily
- Related links
- Recombinant Proteins
- Native Proteins
- GMP Proteins
- Fluorescent Dyes
- Immunoglobulins information
- Immunoglobulin Superfamily Members
- Immunoglobulins
- Immune System
- Human Leukocyte Antigen (HLA)
IgSF CAMs
Co-stimulatory/inhibitory molecules
Growth Factor Receptors
Receptor tyrosine kinases
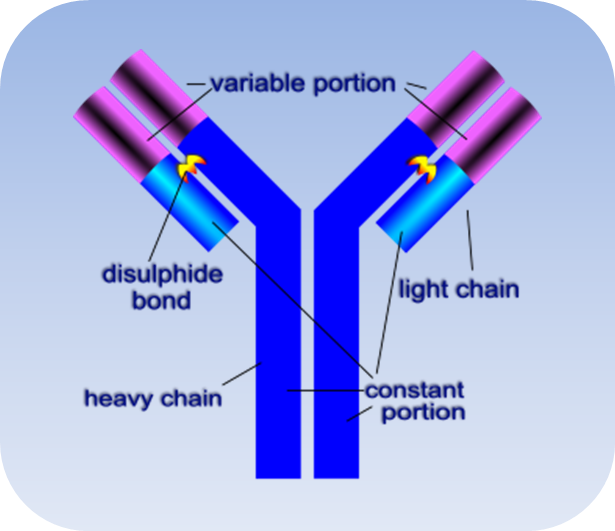
What is Immunoglobulins superfamily (IgSF)?
The immunoglobulin superfamily (IgSF) is a class of proteins that are associated with the adhesion, binding and recognition processes of cells. The term "immunoglobulin superfamily" (IgSF) initially referred to Igs and other proteins involved in the immune response and sharing the same 3D topology. The subsequent discovery of the Ig fold in proteins not functionally related to Igs led to the definition of new functional families, structurally similar to the Igs, such as that of the cytokine receptors or of the bacterial proteins containing the fibronectin type III module.
Proteins which have similar structure to immunoglobulins are classified into IgSF. In recent years, increasing numbers of novel members of the IgSF have been identified. Membership of the IgSF of a newly identified protein is usually based on the conservation in folding and in sequence of specific features found within the Ig molecules. These criteria include domain size (-100 amino acids), the number of strands, and the general topology of the Ig domain or Ig fold. All IgSFs possess at least one immunoglobulin domain or fold.
The IgSF superfamily is composed of surface antigen receptors, co-receptors of the immune system, cell adhesion molecules, some cytokine receptors and molecules involved in antigen presentation to lymphocytes. Most of the IgSF members play roles in the immune system. But the sperm-specific protein Izumo is essential for sperm-egg fusion.
The major functional characteristics of IgSF members have been classed in groups as follows:
Molecular transport: Antibiotic proteins actively transport chromophores through different cellular compartments. In vertebrates, hemocyanins transport oxygen molecules. The Ig receptor (Poly IgR) transports Igs through the epithelial wall.
Morphoregulation: The proteins of the extracellular matrix are involved in the architectural organization and elasticity of a large number of tissues. Fibronectin acts as an "adhesive" that conserves tissue integrity. Cell adhesion molecules exposed on the embryonic cell surfaces (such as cadherin molecules) favor the organization of these cells into differentiated tissues.
Cell phenotype markers: e.g., tumor-cell markers and surface molecules of hematopoietic cells. The latter allow us to evaluate the cell type (B or T cells of the immune system) or the state of cell differentiation (B or pre-B cells, activated T cells, transformed cells, etc.).
Cell adhesion molecules: The cellular immune response requires many cell adhesion molecules of the IgSF, such as CD2, CD4, CD8, and MHC (major histocompatibility complex molecules). Some cell phenotype markers are also involved in cell adhesion, e.g., B7, B29, CD19, CD3, CD7, etc.
Virus receptors: e.g., PVR, CD4, ICAM-1, and Bgp molecules, which are the respective receptors of poliovirus, HIV, rhinovirus, and MHV virus, in addition to their constitutive functions.
Shape recognition and toxin neutralization: The property of sequence polymorphism and structural variability of the immunoglobulin molecules allows the immune system to adapt for different antigens.
Viral and bacterial molecules: In bacteria, IgSF members are domains within enzymes. They also can be involved in pili assembly and/or synthesis. In viruses, they act as receptors of cellular mediators (interferons, interleukins, etc.), or, as surface proteins, they may enhance virus virulence and dissemination.
Others: Some other original functions have been observed in the IgSF, such as the regulation of gene transcription (NF-kB), cell migration (VCAM, PECAM-1, etc.), or cell death marking (PD-1). Finally, a number of Ig-like domains do not possess any as-yet-identified biological function.
Immunoglobulin (Ig) domain is the common feature of immunoglobulin superfamily members. This domain is named after immunoglobulin proteins with about 70-110 amino acids. Immunoglobulin domains contain a characteristic structure called Ig-fold with two sheets of antiparallel beta strands forming a sandwich-like structure by cysteine residues in the B and F strands. Immunoglobulin domains also have a complementarity determining region which decides the specificity of antibodies binding their ligands.
There are more than 750 members have been identified in immunoglobulin superfamily which is the most populous family in human genome. Most of these protein members can be found in our products index. Creative-Biomart supplies abundant products related to immunoglobulin superfamily for scientific experiments.
Immunoglobulins superfamily related literatures
1. Inoue N, Ikawa M, Isotani A, et al. The immunoglobulin superfamily protein Izumo is required for sperm to fuse with eggs[J]. Nature, 2005, 434(7030): 234-238.
2. Harpaz Y, Chothia C. Many of the immunoglobulin superfamily domains in cell adhesion molecules and surface receptors belong to a new structural set which is close to that containing variable domains[J]. Journal of molecular biology, 1994, 238(4): 528-539.
3. Maness P F, Schachner M. Neural recognition molecules of the immunoglobulin superfamily: signaling transducers of axon guidance and neuronal migration[J]. Nature neuroscience, 2007, 10(1): 19-26.
4. Juliano R L. Signal transduction by cell adhesion receptors and the cytoskeleton: functions of integrins, cadherins, selectins, and immunoglobulin-superfamily members[J]. Annual review of pharmacology and toxicology, 2002, 42(1): 283-323.
5. Halaby D M, Mornon J P E. The immunoglobulin superfamily: an insight on its tissular, species, and functional diversity[J]. Journal of molecular evolution, 1998, 46(4): 389-400.


