AMOT
-
Official Full Name
angiomotin
-
Overview
This gene belongs to the motin family of angiostatin binding proteins characterized by conserved coiled-coil domains and C-terminal PDZ binding motifs. The encoded protein is expressed predominantly in endothelial cells of capillaries as well as larger vessels of the placenta where it may mediate the inhibitory effect of angiostatin on tube formation and the migration of endothelial cells toward growth factors during the formation of new blood vessels. Alternative splicing results in multiple transcript variants encoding different isoforms. [provided by RefSeq, Jul 2008] -
Synonyms
AMOT; angiomotin; angiomotin p80 isoform; angiomotin p130 isoform;
- Recombinant Proteins
- Cell & Tissue Lysates
- Protein Pre-coupled Magnetic Beads
- Human
- Mouse
- E.coli
- HEK293
- HEK293T
- In Vitro Cell Free System
- Mammalian Cell
- Wheat Germ
- GST
- His
- His (Fc)
- Avi
- Myc
- DDK
- N/A
- N
- Involved Pathway
- Protein Function
- Interacting Protein
- AMOT Related Articles
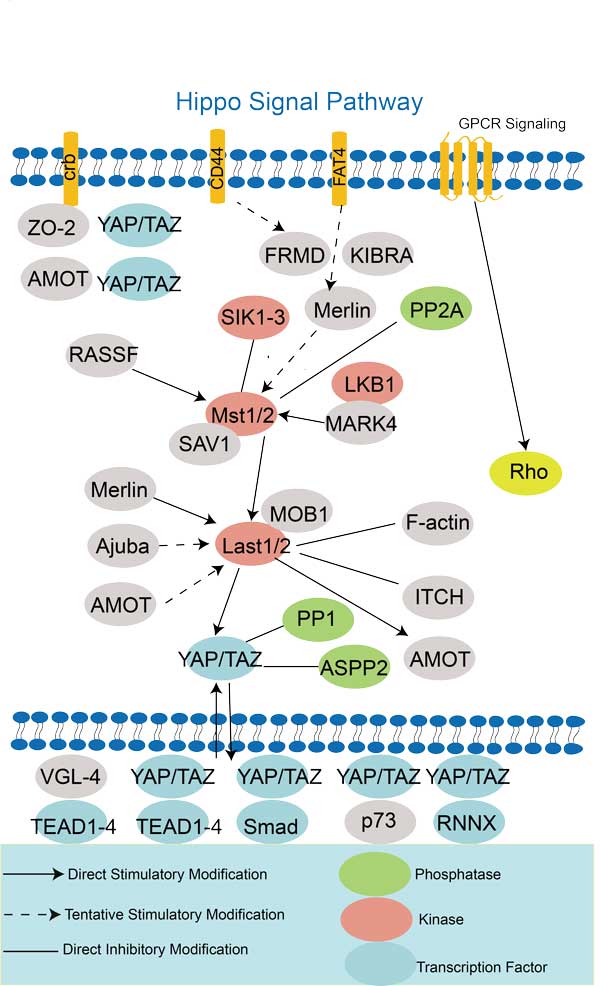
- AMOT Related Signal Pathway
AMOT involved in several pathways and played different roles in them. We selected most pathways AMOT participated on our site, such as , which may be useful for your reference. Also, other proteins which involved in the same pathway with AMOT were listed below. Creative BioMart supplied nearly all the proteins listed, you can search them on our site.
| Pathway Name | Pathway Related Protein |
|---|
AMOT has several biochemical functions, for example, . Some of the functions are cooperated with other proteins, some of the functions could acted by AMOT itself. We selected most functions AMOT had, and list some proteins which have the same functions with AMOT. You can find most of the proteins on our site.
| Function | Related Protein |
|---|
AMOT has direct interactions with proteins and molecules. Those interactions were detected by several methods such as yeast two hybrid, co-IP, pull-down and so on. We selected proteins and molecules interacted with AMOT here. Most of them are supplied by our site. Hope this information will be useful for your research of AMOT.
NF2; MPP5; YAP1; AMOTL1
- Q&As
- Reviews
Q&As (28)
Ask a questionYes, AMOT has been associated with various human diseases and disorders. In addition to cancer, it has been implicated in cardiovascular diseases, including heart development and vascular angiogenesis. Moreover, AMOT is involved in neuronal development and neurodevelopmental disorders such as autism spectrum disorder.
Targeting AMOTL1 or its associated pathways holds potential therapeutic implications. Due to its involvement in cancer progression, inhibiting AMOTL1 activity or disrupting its interactions with other proteins could be explored as a strategy to interfere with tumor growth and metastasis.
Yes, AMOTL1 can interact with several other proteins and genes. One of its well-known interacting partners is the transcriptional co-activators YAP and TAZ. AMOTL1 interacts with YAP and TAZ to regulate cell proliferation, migration, and organ size through the Hippo signaling pathway.
Currently, there are no known genetic disorders specifically associated with mutations in the AMOTL1 gene. However, variations in the AMOTL1 gene have been linked to an increased risk of developing myopia. Myopia is a common eye disorder characterized by nearsightedness, and genetic factors, including AMOTL1 variations, have been found to contribute to its development. Further research is needed to uncover any potential genetic disorders associated with AMOTL1 mutations and to better understand the impact of these variations on human health.
Genetic alterations or mutations in AMOTL1 have been identified in some human diseases. For example, AMOTL1 gene fusions have been found in certain types of cancers, including gliomas and gastrointestinal stromal tumors (GISTs). These alterations may lead to dysregulated AMOTL1 function and contribute to tumorigenesis. Additionally, variations in the AMOTL1 gene have been associated with increased risk of developing eye conditions such as myopia.
AMOTL1 has multiple functions in cellular processes. It can regulate cell migration by modulating actin cytoskeleton dynamics and interacting with other proteins involved in cell motility. It also participates in the regulation of cell junction integrity by associating with tight junction components and controlling their localization. Additionally, it plays a role in angiogenesis, the formation of new blood vessels, by regulating endothelial cell behavior and morphogenesis.
Currently, there are no drugs or compounds specifically targeting the AMOT protein approved for clinical use. However, research efforts are aimed at identifying small molecules or therapeutic strategies that could modulate AMOT function and potentially be used in targeted therapies in the future.
Yes, AMOTL1 has been shown to interact with and regulate signaling pathways involved in cell growth and survival. It can associate with the Hippo pathway, which controls organ size and tumorigenesis, and modulate the activity of YAP and TAZ, the main effectors of this pathway. AMOTL1 can also interact with the Wnt/β-catenin pathway and influence its signaling output. These interactions contribute to the regulation of cellular functions mediated by AMOTL1.
Yes, AMOTL1 is involved in various developmental processes. It is essential for angiogenesis during embryonic development, contributing to the formation of blood vessels in tissues and organs. AMOTL1 also plays a role in organ development, particularly in heart development. It regulates cardiac morphogenesis and chamber formation through its interactions with YAP and TAZ during embryonic and fetal stages.
Yes, AMOTL1 can interact with various proteins and molecules, enabling its involvement in different cellular processes. It can directly interact with components of the tight junction complex, such as ZO proteins, and regulate cell junction integrity. AMOTL1 can also interact with cytoskeletal proteins, including actin and myosin, to influence cell shape and motility. Furthermore, it can interact with proteins involved in signaling pathways, such as angiomotin, YAP, and β-catenin, to modulate their activity and downstream effects.
Yes, AMOT has been shown to interact with and regulate multiple signaling pathways. One example is the Hippo pathway, where AMOT can associate with the core components of the pathway, such as YAP and TAZ, and modulate their activity and localization. AMOT can also interact with and regulate the activity of the PI3K-Akt pathway, MAPK/Erk pathway, and the Wnt/β-catenin pathway. These interactions and regulations contribute to AMOT-mediated cellular functions, including cell proliferation, migration, and epithelial-mesenchymal transition (EMT).
The AMOT protein localizes to different cellular compartments, including the cytoplasm, nucleus, and cell membrane. Its localization can be regulated and varies depending on cellular conditions and signaling pathways.
The AMOT protein is particularly associated with the regulation of cell polarity, cell adhesion, and the Hippo signaling pathway. Dysregulation of these processes can contribute to various diseases, including cancer and cardiovascular disorders.
Targeting the AMOT protein for therapeutic interventions is an area of ongoing research. Modulating AMOT's interactions, stability, or expression could potentially impact diseases where AMOT dysregulation is implicated, such as cancer or cardiovascular diseases. However, further studies are needed to understand the potential therapeutic implications and safety of targeting AMOT in specific diseases.
Yes, animal models, particularly mice, have been utilized to study AMOTL1 function and its role in diseases. Genetically modified mouse models with altered AMOTL1 expression or specific mutations have provided insights into the protein's involvement in angiogenesis, tissue development, and cancer progression. These animal models allow for in-depth investigations of AMOTL1 biology in a physiological context and aid in understanding its functions and potential therapeutic implications.
Yes, the expression of AMOT can be regulated under different physiological or pathological conditions. For example, its expression levels can change during embryonic development, wound healing, and in response to cellular stress or inflammation. Alterations in AMOT expression have also been observed in various diseases, including cancer, cardiovascular disorders, and neurological conditions.
Yes, the AMOT protein undergoes various post-translational modifications, including phosphorylation, ubiquitination, and SUMOylation. These modifications can regulate its localization, interactions, and functions in different cellular processes.
AMOTL1 has been implicated in various diseases and conditions. It has been associated with cancer progression, particularly in gliomas and gastrointestinal stromal tumors (GISTs) where AMOTL1 gene fusions have been identified. In addition, AMOTL1 variations have been correlated with an increased risk of developing myopia, a common eye condition characterized by nearsightedness. Dysregulation of AMOTL1 has also been observed in diseases involving angiogenesis, such as diabetic retinopathy and tumor angiogenesis.
Currently, there are no specific drugs or compounds available for directly modulating AMOTL1 activity. However, targeting signaling pathways associated with AMOTL1, such as the Hippo pathway or Wnt/β-catenin pathway, can indirectly impact AMOTL1 function. Small molecules that selectively inhibit these pathways have been developed and tested in preclinical and clinical studies for various diseases, with potential implications for AMOTL1-related cellular processes. Additionally, exploring protein-protein interactions and post-translational modifications of AMOTL1 may provide avenues for drug discovery in the future.
Yes, animal models, such as mice, have been used to study AMOT function and its involvement in diseases. Genetically modified mice with altered expression or mutations in AMOT genes have provided insights into its roles in development, angiogenesis, cardiac function, and cancer progression. These animal models allow for the investigation of AMOT biology in a complex organism, providing valuable information for understanding its cellular functions and potential therapeutic strategies.
Yes, the AMOT protein can interact with various proteins to regulate its functions and signaling pathways. It interacts with components of tight junctions, cell adhesion molecules, and signaling molecules involved in the Hippo pathway, such as YAP and TAZ.
AMOTL1 is widely expressed throughout different tissues and cell types, although its expression levels can vary depending on the cell type and developmental stage. While it is present in various organs, such as the brain, heart, and kidney, AMOTL1 is particularly enriched in endothelial cells, which line the interior surface of blood vessels. Its high expression in endothelial cells highlights its crucial role in angiogenesis and vascular development.
Yes, genetic variants and mutations in the AMOT gene have been identified in studies and databases. Some of these variants have been associated with various diseases and conditions, including cancer, cardiovascular diseases, and developmental disorders.
AMOTL1 can be regulated at multiple levels within the cell. It is subject to post-translational modifications, including phosphorylation and ubiquitination, which can influence its stability, subcellular localization, and interaction with other proteins. AMOTL1 activity can also be regulated through protein-protein interactions, particularly with components of signaling pathways.
Yes, AMOT can be localized to specific cellular compartments, and its localization can be dynamically regulated. It contains specific protein motifs that determine its cellular localization, such as nuclear localization signals (NLS) and nuclear export signals (NES). AMOT isoforms have been observed in the nucleus, cytoplasm, and at cell-cell junctions. The localization of AMOT can dictate its interactions and functions within specific cellular compartments.
Yes, genetic mutations or alterations in AMOT have been reported in certain human diseases. For example, mutations in AMOT have been identified in individuals with Klippel-Trenaunay syndrome, a rare vascular disorder.
The role of the AMOT protein in cancer is complex and context-dependent. In some cases, it has been described as a tumor suppressor, as it can inhibit cell growth and migration. However, in other circumstances, it can promote tumor growth and invasion, acting as an oncogene. The specific role of AMOT in cancer may vary depending on the cellular context and interacting partners.
There are mouse models and knockout studies targeting the AMOT gene. These studies have provided insights into the function of AMOT and its role in embryonic development, vascular development, and tumor growth.
Customer Reviews (4)
Write a reviewI am extremely satisfied with the high performance and the exceptional support associated with this protein, as it has greatly contributed to the success of my scientific endeavors.
Its purity and consistency have consistently generated trustworthy and reproducible results in my research.
Their expertise and prompt assistance have been instrumental in overcoming challenges and optimizing the use of the AMOT protein in my studies.
the manufacturer's commitment to delivering excellent technical support has proven to be invaluable in troubleshooting any issues that arose during my experiments.
Ask a Question for All AMOT Products
Required fields are marked with *
My Review for All AMOT Products
Required fields are marked with *