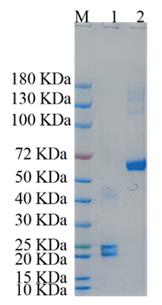
Full Length Protein
🧪 RAP1A-2584H
Source: E.coli
Species: Human
Tag: His
Conjugation:
Protein Length: 1-184 aa

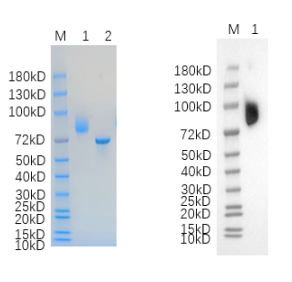
🧪 EGF-04H
Source: E.coli
Species: Human
Tag:
Conjugation:
Protein Length:
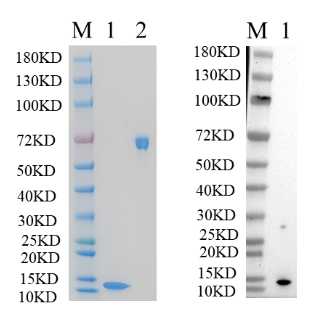
🧪 S100A4-409H
Source: E.coli
Species: Human
Tag: His
Conjugation:
Protein Length: 1-101 aa
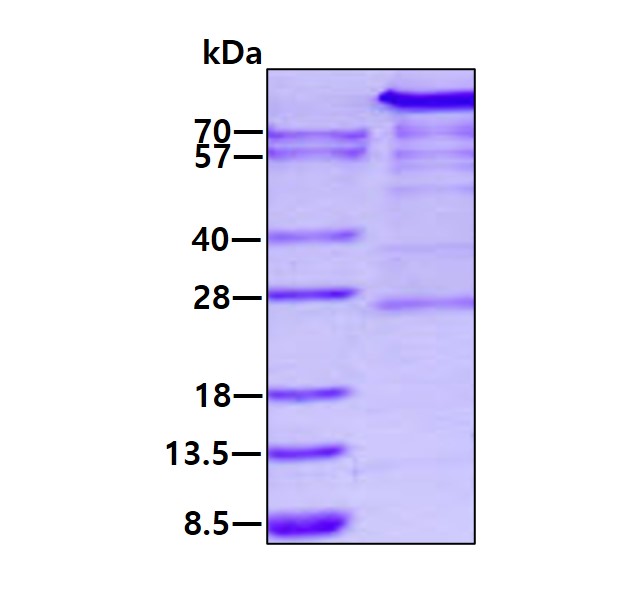
 Full L.
Full L.
🧪 HK2-41H
Source: E.coli
Species: Human
Tag: His
Conjugation:
Protein Length: 1-917aa
🧪 Capn2-2632R
Source: E.coli
Species: Rat
Tag: His
Conjugation:
Protein Length: 1-700aa
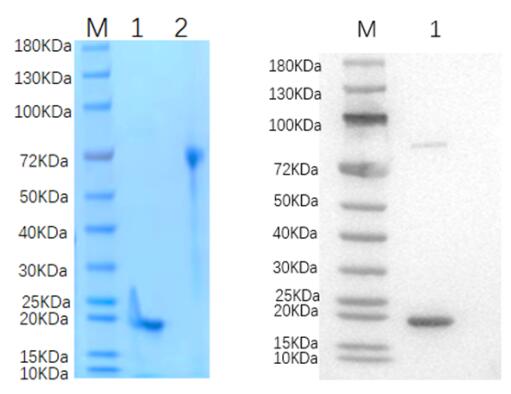
🧪 GAD1-1180H
Source: Insect Cells
Species: Human
Tag: His
Conjugation:
Protein Length: 1-594 aa

🧪 SOD3-1038H
Source: E.coli
Species: Human
Tag: Non
Conjugation:
Protein Length: 1-240 aa
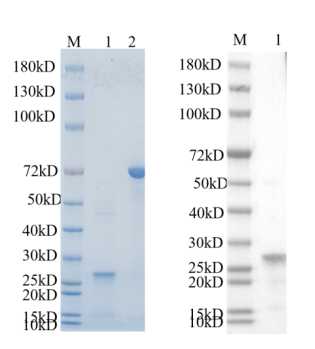
 Full L.
Full L.
🧪 KRT19-7239H
Source: E.coli
Species: Human
Tag: His
Conjugation:
Protein Length: 1-400 aa
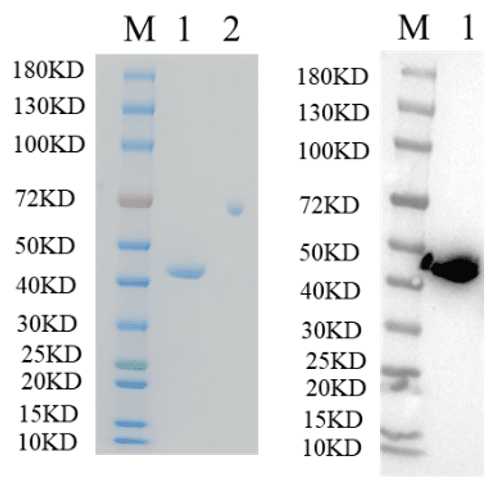
🧪 FGF4-520H
Source: E.coli
Species: Human
Tag:
Conjugation:
Protein Length:
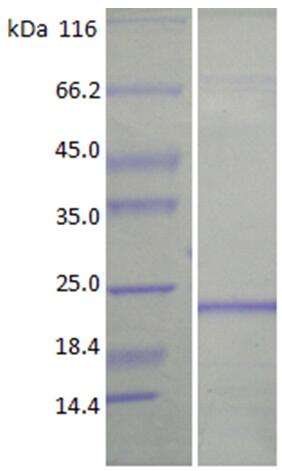
🧪 Bcl2-3582M
Source: E.coli
Species: Mouse
Tag: His
Conjugation:
Protein Length: 1-236 aa
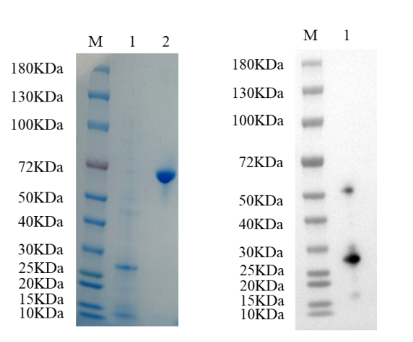
🧪 DDX3Y-3751H
Source: E.coli
Species: Human
Tag: His
Conjugation:
Protein Length: 1-660 aa

🧪 SLC30A8-03H
Source: Sf9 Cells
Species: Human
Tag: His
Conjugation:
Protein Length:
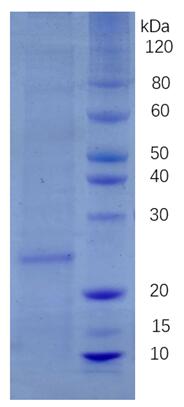
🧪 GUCY2C-7391M
Source: Human Cells
Species: Mouse
Tag: His
Conjugation:
Protein Length:
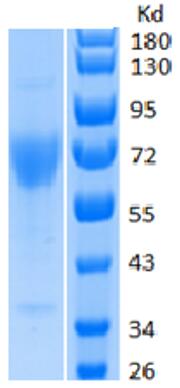
🧪 LIPE-9128M
Source: HEK293
Species: Mouse
Tag: His
Conjugation:
Protein Length: 1-759 aa
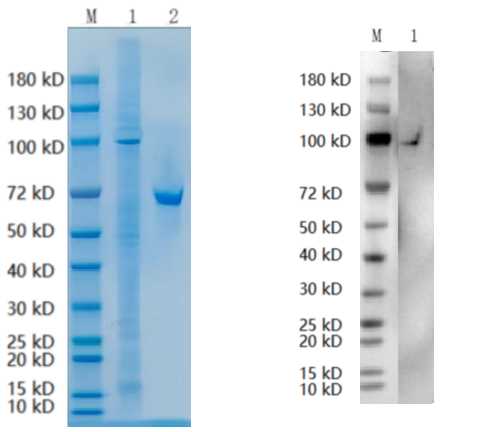
 Full L.
Full L.
🧪 SIAE-15119M
Source: HEK293
Species: Mouse
Tag: His
Conjugation:
Protein Length: 22-541 aa
Background
Overview
A full-length protein is a complete protein sequence from the N terminal to the C terminal. The complete amino acid sequence of the protein can be obtained by full length protein sequencing, which is essential for understanding the biological function of the protein. It helps scientists fully understand the function of proteins, including how they interact with other molecules, as well as their localization and mechanism of action within cells. The study of full length proteins can reveal the role of specific proteins in the development of diseases, which is of great significance for the discovery of new therapeutic targets and methods. In addition, structural analysis of full length proteins can provide detailed information about the three-dimensional structure of proteins, which is of guiding significance for understanding their functions and designing experiments.

Challenges
In biological research, the study of full-length proteins is of great significance because their structure often determines their function. However, there are many problems in the process of expression. For example:
Expression challenges: The expression of full-length proteins in prokaryotic cells such as E. coli may be affected by a variety of factors, including protein hydrophilicity, codon rarity, and protein toxicity. For example, proteins that are too hydrophobic are difficult to express, and proteins that contain multiple rare codons linked together may also cause difficulty in expression. In order to solve these problems, researchers need to analyze the protein sequence and secondary structure, and adopt corresponding strategies to optimize the expression conditions.
Translation initiation problems: When expressing full-length proteins, problems with truncated products may be encountered. This may be due to proteolysis or improper initiation of translation. To ensure the acquisition of full-length proteins, expression vectors with fusion labels on both ends can be used to distinguish full-length proteins from truncated proteins by increasing the imidazole concentration at elution.
Transmembrane protein challenges: For specific full-length proteins such as transmembrane proteins, their expression and purification are particularly challenging. To facilitate drug development, the MNP platform extracts high-purity nanoscale cell membrane particles while maintaining the conformation and activity of membrane proteins.

Outlook
The future of full-length protein research is full of promise and challenges, and with the continuous advancement of technology, there are already many biologically important recombinant protein classes on the market today. In the future, full-length proteins are expected to make breakthroughs in many other areas. Including but not limited to:
Improved accuracy of protein structure prediction: With the development of AI-based protein structure prediction technologies such as AlphaFold2, the ability of these technologies to predict the three-dimensional structure of unknown proteins will become even more powerful in the future. This enhancement will not only lead to a better understanding of protein function, but also accelerate the process of drug discovery and biological research.
Research on multi-domain proteins and complexes: At present, there are still challenges in predicting the structure of multi-domain proteins and protein complexes. Future research may lead to breakthroughs in these areas, leading to a more complete understanding of protein complexity and diversity.
Innovative applications of protein design: The application of deep learning techniques has made it possible to design completely new proteins from scratch. In the future, this technology may be further developed to create customized proteins with specific functions, such as enzymes, vaccines, and drug delivery vectors, which will have a profound impact on the pharmaceutical and biotechnology industries.
Improvement of computational tools and databases: With the accumulation of more and more information about protein sequences and their structures, combined with advances in artificial intelligence technology, more efficient computational tools and databases are likely to emerge in the future to support full-length protein research and innovation.

Applications
- Drug development: By preparing recombinant full-length proteins, it is possible to conduct drug-target protein interaction studies, understand the binding mechanism of the drug to the target, and evaluate the activity and specificity of the drug.
- Cell therapy: In the field of cell therapy, recombinant full-length proteins can be used to prepare therapeutic cell products, such as receptor proteins in CAR-T cell therapy for tumor immunotherapy.
- Vaccine development: By expressing recombinant full-length proteins, antigens for use in vaccines can be prepared to induce an immune response to protect humans from pathogens.
Case Study
Case Study 1: Recombinant Human EGF Protein
Re-education of the tumor microenvironment with immune checkpoint inhibitors (ICI) has provided the most significant advancement in cancer management, with impressive efficacy and durable response reported. However, low response rates and a high frequency of immune-related adverse events (irAEs) remain associated with ICI therapies. The latter can be linked to their high affinity and avidity for their target that fosters on-target/off-tumor binding and subsequent breaking of immune self-tolerance in normal tissues. Many multispecific protein formats have been proposed to increase the tumor cell's selectivity of ICI therapies.
In this study, the researchers explored the engineering of a bispecific Nanofitin by the fusion of an anti-epidermal growth factor receptor (EGFR) and anti-programmed cell death ligand 1 (PDL1) Nanofitin modules. While lowering the affinity of the Nanofitin modules for their respective target, the fusion enables the simultaneous engagement of EGFR and PDL1, which translates into a selective binding to tumor cells co-expressing EGFR and PDL1 only.

(Perrine Jacquot, 2023)
Fig1. EGFR phosphorylation level in the presence or the absence of EGF, Cetuximab and B10 Nanofitin studied by Western Blot on A431 cell line.
Case Study 2: Recombinant Human DNM2 Protein
NM23 (NME) is a metastasis suppressor that significantly reduces metastasis without affecting primary tumor size, however, the precise molecular mechanisms are not completely understood. The researchers examined the role of dynamin (DNM2), a GTPase regulating membrane scission of vesicles in endocytosis, in NME1 and NME2 regulation of tumor cell motility and metastasis.
Dynamin inhibitors Iminodyn-22 and Dynole-34-2, or shRNA-mediated downregulation of DNM2, impaired NME's ability to augment endocytosis or suppress tumor cell motility. In a lung metastasis assay, NME1 overexpression failed to significantly suppress metastasis in the DNM2 knockdown MDA-MB-231T cells. Using the EGF-EGFR signaling axis as a model in MDA-MB-231T cells, NME1 decreased pEGFR and pAkt expression in a DNM2-dependent manner, indicating the relevance of this interaction for downstream signaling. NME-DNM2 interaction was confirmed in two-way coimmunoprecipitations.

(Imran Khan, 2019)
Fig2. Two-way co-immunoprecipitation of NME1 and DNM2 was performed on vector and NME1 overexpressing MDA-MB-231T cells using rabbit anti-NME1 and goat anti-DNM2 antibody.
Case Study 3: Active Recombinant Human FGF4 Protein
The present study aimed to explore the functions and molecular mechanisms of miR-511 in breast cancer. A quantitative real-time polymerase chain reaction (qRT-PCR) was used to detect miR-511 levels in breast cancer tissues; a chi-squared test was used to analyze the relationship between miR-511 expression level and pathological parameters of breast cancer patients; TargetScan was used to predict the binding site between the 3'-untranslated region (3'-UTR) of fibroblast growth factor 4 (FGF4) and miR-511; and qRT-PCR, western blot and a luciferase reporter gene assay were conducted to further validate the targeting relationship between miR-511 and FGF4.
The expression level of miR-511 was lower in breast cancer tissues than that in adjacent normal tissues. Low expression of miR-511 was associated with larger tumor size. It was also confirmed that miR-511 directly targeted 3'-UTR of FGF4 and reduced its expression, and FGF4 overexpression reversed the effect of miR-511 on the malignant phenotypes of breast cancer cells.

(Chao Zhang, 2020)
Fig3. Western blot was used to detect the change of FGF4 protein of MDA-MB-231 and MCF-7 cells after up-regulation or down-regulation of miR-511.
Case Study 4: Recombinant Full Length Human CD40 Protein
Fibrocytes appear to participate in inflammation and tissue remodeling in patients with thyroid-associated ophthalmopathy (TAO). These patients have increased frequencies of circulating TSH receptor (TSHR)- and CD40-positive fibrocytes, suggesting TSHR and CD40 may play roles in proinflammatory cytokine production, which ultimately leads to orbital inflammation and tissue remodeling. This study aims to investigate the potential interactions between the TSHR and CD40 signaling pathways and their roles in IL-6 and TNF-α production.
CD40 expression on fibrocytes was assessed using flow cytometry; TSH- and CD40 ligand (CD40L)-stimulated Akt phosphorylation in fibrocytes, by western blot analysis; TSHR-CD40 protein-protein interaction, using co-immunoprecipitation, and CD40-TSHR co-localization, using immunocytochemistry. The results show TSH enhances CD40 expression at a pre-translational level in fibrocytes. Production of IL-6 and TNF-α after costimulation with TSH and CD40L was greater than that after TSH or CD40L stimulation alone. TSH and CD40L costimulation also resulted in greater Akt phosphorylation.

(Tünde Mester, 2016)
Fig4. Co-IP studies using TSHR (left panel) or CD40 antibodies (right panel) show that TSHR antibody pulls down TSHR and CD40, and CD40 antibody also pulls down both proteins, indicating physical contact between the 2 proteins.
Advantages
- Wide Coverage: More than 300 full length proteins from over 20 different species and nearly 10 different sources.
- High Quality: The complete amino acid sequence ensures that the protein function is not distorted. And they have been tested with different methods to ensure intergrity and high purity.
- Quality and industrial testing: We closely monitor the quality and safety of all your products. Our commitment is to fully control manufacturing, from raw materials to finished products, with strict adherence to product specifications, certificates of analysis from independent laboratories, traceability, batch records, and specification sheets.
- Experienced: We have professional research team with experience of many years in the field of molecular and cell biology.
- Fast turnaround: Under the premise of your protein expression and purification, as little as 4-6 weeks.
FAQ
-
Q: When is a recombinant full-length protein needed?
A: By synthesizing full-length proteins, a large number of homogeneous and stable proteins can be obtained for further research and application.
-
Q: How to select a full-length protein expression system?
A: We can provide commonly used expression systems including E. coli, yeast, insect cells, mammalian cells and other expression systems. Specific needs to combine the characteristics of the target protein, folding capacity, activity, time and cost and other factors.
References
- Khan, I.; et al. Metastasis Suppressors NME1 and NME2 Promote Dynamin 2 Oligomerization and Regulate Tumor Cell Endocytosis, Motility, and Metastasis. Cancer Res. 2019;79(18):4689-4702.
- Jacquot, P.; et al. Engineering of a Bispecific Nanofitin with Immune Checkpoint Inhibitory Activity Conditioned by the Cross-Arm Binding to EGFR and PDL1. Biomolecules. 2023;13(4):636.
- Zhang, C.; et al. miR-511 inhibits proliferation and metastasis of breast cancer cells by targeting FGF4. J Gene Med. 2020;22(9):e3168.
- Mester, T.; et al. CD40 Expression in Fibrocytes Is Induced by TSH: Potential Synergistic Immune Activation. PLoS One. 2016;11(9):e0162994.

