Cell & Tissue Lysates

🧪 ESD-6541HCL
Source: HEK293
Species: Human
Tag: Non
Conjugation:
Protein Length:

🧪 ATF1-8632HCL
Source: HEK293
Species: Human
Tag: Non
Conjugation:
Protein Length:
🧪 IL21R-001CCL
Source: Human Cells
Species: Cynomolgus
Tag: Non
Conjugation:
Protein Length:

🧪 IL12A-001CCL
Source: Human Cells
Species: Cynomolgus
Tag: Non
Conjugation:
Protein Length:

🧪 COLEC10-001CCL
Source: Human Cells
Species: Cynomolgus
Tag: Non
Conjugation:
Protein Length:

SubCategories
Background
Overview
Cell and tissue lysates are the types of samples commonly used in biological research. They are mixtures obtained by physically or chemically destroying cell membranes and intracellular structures and releasing cell contents. The pyrolysis methods can be divided into chemical pyrolysis, enzyme pyrolysis and physical pyrolysis. Chemical cracking and enzyme cracking are milder methods and rarely cause DNA breakage, while physical cracking, although high yield, is a more violent reaction and may cause DNA breakage. When choosing a cleavage method, it is necessary to consider the stability and activity of the target molecule, as well as the possible effects on other components within the cell during the cleavage process. For example, chemical cracking may result in the introduction of certain chemicals, while physical cracking may result in DNA breakage caused by shear forces.
It should be noted that in order to prevent protein degradation and nucleic acid degradation during cleavage, it is usually necessary to add protease inhibitors and nuclease inhibitors. The cleavage process should be carried out at low temperatures to reduce enzyme activity and protein degradation. Follow-up analysis should be performed immediately after cracking, or stored properly to avoid sample degradation.
Compositions of the lysates
The composition of the lysate depends on the lysate method and the starting material, but usually contains the following components:
Protein: soluble protein within the cell; Membrane binding proteins may require additional washing steps to separate; Specific proteins in organelles.
Nucleic acid: DNA, which may need to be removed by DNase processing; RNA, too, may need to be removed or separated by RNA enzyme processing.
Organelle fragments: fragments of organelles such as mitochondria, endoplasmic reticulum, and Golgi apparatus; These fragments may contain specific enzymes and proteins that are useful for studying cellular metabolism and signaling pathways.
Metabolites: including small metabolites (such as amino acids, sugars, lipids) and large metabolites (such as nucleotides, coenzymes); Analysis of metabolites can reveal changes in metabolic state and pathways of cells.
Ions and other small molecules: ions inside and outside the cell (e.g., Na+, K+, Ca2+, Mg2+); Low molecular weight compounds such as antioxidants, signaling molecules, etc.
Extracellular material: If extracellular fluid is included in the lysis process, then the lysate may also contain extracellular matrix components, serum proteins, etc.
Comparison of lysis techniques
Mechanical lysis:
The mechanical lysis subjects a cell to a physical force, such as compression within a confined region, where the mechanical stress results in membrane rupture, or to sharp, physical structures which inhibit a cell’s path and punctures the membrane in a uncontrolled manner. The compression method, while capable of producing fast (~ms), complete lysis, results in a nonuniform diffusion of cellular content, and the possibility of cellular debris adhering to parts of the compression region. Similarly, mechanical lysis against physical structures typically results in cellular debris sticking to the physical structures and poor diffusion due to incomplete membrane rupture. Additionally, neither of these mechanisms lends itself to selective lysis.
Chemical lysis:
The most widely used method for bulk assays which translates well to single cells, is the introduction of a chemical detergent which solubilizes lipids and proteins in the plasma membrane, creating pores which leads to complete lysis. Two common detergents, Triton X-100 and SDS, each exhibit different lysis capabilities, with the former typically inducing lysis in ~30s while preserving enzyme activity; whereas, the latter usually results in faster lysing (< 2s) but denatures membrane and cellular proteins. Denatured proteins are typically unfavorable as they quickly aggregate, forming an insoluble, randomly organized structure. While chemical lysis does not require specialized equipment apart from a mixing method, and various detergents exist to enable selective lysis, it is evident that the detergents can significantly impact the outcome of the experiment due to long times to lyse, leading to excessive diffusion of cellular content. Detergents also tend to denature and break up protein complexes, while also adding an additional reagent that may eventually need to be removed from the cell lysates prior to a specific assay.
Enzymatic lysis:
Enzymatic lysis is a gentle cleavage method that uses the biocatalytic action of specific enzymes to break down the cell wall or membrane, which releases the contents of the cell by selectively destroying the external structure of the cell. This method is particularly suitable for microorganisms with strong cell walls such as bacteria and fungi, as well as plant cells, because it is able to efficiently release intracellular proteins, nucleic acids, and other biomolecules without destroying sensitive biomolecules within the cell. In the process of enzymatic cleavage, it is first necessary to select suitable enzymes, such as lysozyme for the cleavage of bacterial cell walls, cellulase and pectinase for the cleavage of plant cell walls. The cells are then suspended in a lysis buffer containing these enzymes and incubated at the right temperature and pH to promote the breakdown of the cell wall. The efficiency of cleavage can be assessed by microscopic observation or biochemical analysis, and can be optimized by adjusting enzyme concentration, incubation time, and temperature. Finally, the unlysed cells and cell fragments are removed by physical methods such as centrifugation to obtain lysates that can be used for subsequent biological research. The advantage of enzymatic cracking is that it is highly selective and controllable, but it may take longer and cost more.
What We Offer
Creative BioMart now offers the collection of lysates of different types which include Over-expression lysates, Tissue lysates, Cell line lysates, Stem cell lysates and Membrane lysates. We offer tissue and cell lysates which can be used to evaluate antibody quality and serve as an excellent positive control for electrophoresis, western blotting, immunoprecipitation, enzymatic activity analysis, protein-protein interaction analysis and tissue specific expression identification, and over-expression lysates to provide a cost-effective option as assay standards in Western blot. Our lysates are validated and are available in a variety of normal, tumor and disease characterized tissues.
Over-expression lysates: Over-expression lysates are derived from cells or organisms that have been genetically engineered to produce a specific protein in greater quantities than they would under normal conditions. This is typically achieved by using recombinant DNA technology, where the gene encoding the protein of interest is inserted into a plasmid and transformed into a host cell, such as bacteria (E. coli), yeast, or mammalian cells.
Tissue lysates: Tissue lysates are prepared by homogenizing and solubilizing tissues from animals or plants to release cellular contents. This process breaks down the cell membranes and organelles, resulting in a mixture of proteins, nucleic acids, and other biomolecules.
Cell line lysates: Cell line lysates are obtained by breaking open a population of cultured cells, such as those derived from cancer (cancer cell lines) or immortalized normal cells. The cells are typically lysed using detergents, enzymes, or mechanical disruption. Cell line lysates represent the proteome and transcriptome of the specific cell line and can be used to study the effects of genetic alterations or environmental factors on cellular processes.
Stem cell lysates: Stem cell lysates are prepared from stem cells, which are undifferentiated cells with the potential to develop into many different cell types. These cells can be derived from various sources, such as embryonic stem cells or adult stem cells. The composition of stem cell lysates is characterized by a diverse range of proteins involved in self-renewal, differentiation, and other stem cell-specific processes.
Membrane lysates: Membrane lysates are specialized preparations that focus on the components of cellular membranes, including the plasma membrane and membranes of organelles such as mitochondria and the endoplasmic reticulum. These lysates are typically prepared using methods that selectively disrupt the membrane structures while preserving membrane-associated proteins.

Applications
Cell and tissue lysates have a wide range of applications in biomedical research, providing the basis for a variety of experimental techniques by releasing proteins, nucleic acids, metabolites and other biomolecules within cells. The following are some of the main applications of cell and tissue lysates:
Immune response detection: Cell and tissue lysates contain a large number of endogenous proteins and antigens that can be used to detect immune system responses. By evaluating antibodies against specific lysate antigens, it can help diagnose autoimmune diseases such as rheumatoid arthritis and multiple sclerosis.
Drug screening: In the field of drug development, cell-lysate antigens are used to screen and identify drug candidates that may be effective for specific diseases. Many drugs' mechanisms of action involve interactions with proteins or other molecules inside cells, and cell lysates provide a platform that allows researchers to quickly assess the effects of drugs.
Proteomic studies: Protein expression analysis: Quantitative and qualitative analysis of proteins in lysates to study the expression level of specific proteins in cells or tissues; Protein interaction: Immunocoprecipitation (Co-IP) or yeast two-hybridization experiments are conducted using proteins in lysates to explore the interaction between proteins; Protein function study: The enzyme activity of the enzyme protein in the lysate was determined to study its biological function.
Cell therapy and regenerative medicine: Cell culture: The use of specific cell lysates as a complement to cell culture to support cell growth and differentiation; Tissue engineering: Bioactive molecules from lysates are used in tissue engineering to promote tissue regeneration and repair.
Forensics and biosafety: DNA fingerprinting: genetic analysis of DNA extracted from lysates for forensic identification and paternity testing; Pathogen detection: detection of pathogen components in lysates for the diagnosis and epidemiological study of infectious diseases.

Case Study
Case Study 1: Recombinant Human ESD 293 Cell Lysate (ESD-6541HCL)
High levels of cholesterol in the blood can lead to non-alcoholic fatty liver disease (NAFLD) and hardening of the arteries. Although there are more and more publications and patent applications for lowering blood cholesterol with small chemical molecules, clinically effective drugs are limited and there is a need to discover new targets and drugs to relieve high cholesterol. Esterase D (ESD) is abundant in the liver, and its role in cholesterol metabolism is unclear. In this paper, a new ESD activator, small chemical molecule fluorescent pyrazoline derivative 5 (FPD5), can effectively reverse the high blood cholesterol level and prevent fatty liver and arteriosclerosis in high-fat apoE-/- diet mice. FPD5 can reduce the formation of foam cells induced by oxidized low density lipoprotein (oxLDL). Further exploring the mechanism of FPD5 on blood cholesterol regulation, it was found that FPD5-triggered ESD aggregated in lysosomes. It also interacts with Jun activation domain binding protein 1 (JAB1). ESD acts as a deacetylase to remove Thr89 acetylation of JAB1 and improve its activity. Thus, the ATP-binding box transporter A1 (ABCA1) accelerates cholesterol effluence.
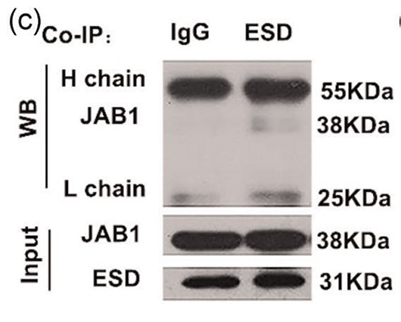
(Xinpeng Chen, 2021)
Fig1. Co‐immunoprecipitation (co‐IP) of ESD with the JAB1 protein from HEK293T cells.
Case Study 2: Recombinant Human ATF1 293 Cell Lysate (ATF1-8632HCL)
Multiple genome-wide studies have identified associations between the outcome of human immunodeficiency virus (HIV) infection and polymorphisms in and around the gene encoding the HIV coreceptor CCR5, but the functional basis of the strongest of these associations, rs1015164A/G, has been unclear. We report that rs1015164 marks a mutation in the activation transcription factor 1 binding site, which controls the expression of antisense long non-coding RNA (lncRNA) CCR5AS. Knockdown or enhancement of CCR5AS expression leads to corresponding changes in CCR5 expression on CD4+ T cells. CCR5AS interferes with the interaction between the RNA-binding protein Raly and the 3' untranslated region of CCR5, protecting CCR5 messenger RNA from RALy-mediated degradation. Reducing CCR5 expression by inhibiting CCR5AS in vitro can reduce CD4+ T cell infection with CCR5-CCR5-addicted HIV. These data represent a rare determination of the functional importance of genome-wide disease associations in which lncRNA expression influences HIV infection and disease progression.
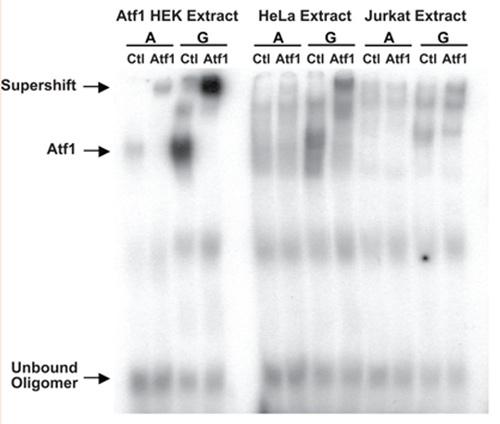
(Smita Kulkarni, 2019)
Fig2. An electrophoretic mobility shift assay (EMSA) was performed with 32P-labelled oligonucleotides containing the core binding sequence for ATF1 containing the A or G variant of rs2027820.
Case Study 3: Recombinant Human SMN1 293 Cell Lysate (SMN1-1660HCL)
In recent years, due to the high conductivity, mechanical strength and large surface area of carbon nanomaterials, the integration of carbon nanomaterials into electrochemical sensors has aroused great interest. However, no comparative study on aptamer sensing applications of different carbon nanomaterials has been reported. This study reports a comparative study of the properties of six carbon electrode materials (carbon, graphene (G), graphene oxide (GO), multi-walled carbon nanotubes (MWCNT), single-walled carbon nanotubes (SWCNT), and carbon nanofibers (CNF)) on physical adsorption-prepared hemoglobin A1C (HbA1c) apperomer sensors. The aptamer is non-covalently fixed to six nanoelectrodes through the π-π stacking interaction between the DNA nucleobase and the surface of the carbon material, thus forming a barrier for electron transfer. However, once the target protein binds to the aptamer, the aptamer dissociates from the surface, thereby enhancing electron transfer, which is the basis of detection. Comparison of aptamer adsorption, sensor response and selectivity of different nanomaterials showed that swcnts sensors had better performance. The detection limits of total hemoglobin (tHb) and glycosylated hemoglobin (HbA1c) by Voan swcnts aptamer sensors were 0.13 pg/mL and 0.03 pg/mL, respectively, showing high sensitivity and selectivity. It is selective for blood proteins such as cystic fibrosis transmembrane conduction regulator (CFTR), survival motor neuron (SMN), cytoplasmic division dedication factor 8 (DOCK8), signal transducer and transcriptional activator 3 (STAT3).
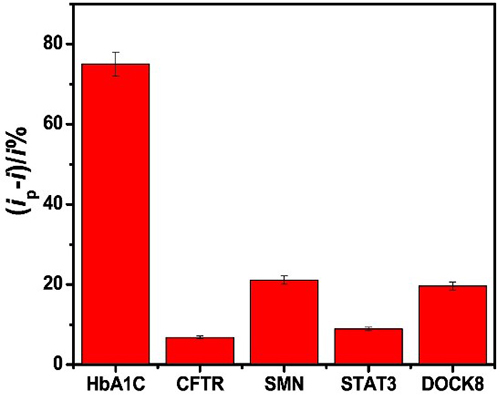
(Shimaa Eissa, 2019)
Fig3. The HbA1c aptamer assay selectivity against 1 ng/mL of HbA1c, CFTR, SMN, STAT3 and DOCK8.
Case Study 4: Recombinant Human SMN1 293 Cell Lysate (SMN1-1660HCL)
High immunoglobulin E syndrome (HIES) is a rare inherited birth defect, and HIESs is inherited through heterozygous or homozygous mutations in the signaling transducer and transcriptional activator 3 (STAT3) or cell division dedication factor 8 (DOCK8) genes, in autosomal dominant and recessive forms, respectively. Therefore, early detection of human DOCK8 and STAT3 protein levels would aid in the early diagnosis of these diseases and thus in the management of the disease. This study presents the development of immunosensors using electrochemical impedance spectroscopy to detect DOCK8 and STAT3 in a label-free format. The immunosensor was prepared by covalently attaching DOCK8 and STAT3 specific antibodies to gold electrode with cysteamine/phenyl diisothiocyanate linker. This detection is achieved by monitoring the change in charge transfer resistance (Rct) after the iron/ferricyanide REDOX pair binds to the surface of the immunosensor. These biosensors are capable of detecting DOCK8 and STAT3 levels with detection limits of 1.2 and 9.0 pg/ml, respectively. The immunosensor was also applied to the detection of DOCK8 in human serum samples, showing a high recovery rate.
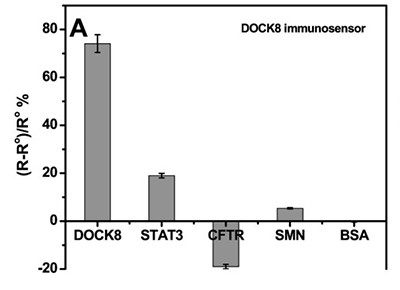
(Shimaa Eissa, 2019)
Fig4. Sensor responses of the DOCK8 immunosensor to 1 ng/mL of BSA, STAT3, CFTR or SMN.
Advantages
- Professional technical support: We have a professional technical team, can provide customers with professional technical support and consulting services. This helps to better understand the properties and applications of the various cracking fluids, so that more reasonable choices can be made.
- Safety and compliance: We comply with relevant biosafety and bioethics regulations to ensure that all cracking services are carried out in a safe environment and in compliance with legal and regulatory requirements.
- Innovative research and development capabilities: We constantly develop new cracking technologies and methods, and develop new products to adapt to changing scientific research needs and market trends.
- Customized services: We can provide customized pyrolysis services according to your specific needs, including the selection of specific pyrolysis conditions, the addition of specific inhibitors to prevent the degradation of certain biomolecules, and the provision of subsequent sample handling and analysis services.
FAQ
-
Q: Does the cracking method used ensure high efficiency and yield?
A: We will choose the pyrolysis method and monitor the pyrolysis process in real time according to the experimental purpose, sample type and other factors. Additional removal of insoluble matter and cell debris increases the yield and purity of the final lysate.
-
Q: Does the lysis process preserve the integrity and activity of all biomolecules within the cell, especially for sensitive proteins and RNA?
A: In practice, in order to protect sensitive proteins and RNA, we will adopt milder means or cleavage methods, cleavage buffers and other means to preserve. It also depends on your specific needs.
References
- Chen X.; et al. A new activator of esterase D decreases blood cholesterol level through ESD/JAB1/ABCA1 pathway. J Cell Physiol. 2021;236(6):4750-4763.
- Kulkarni S.; et al. CCR5AS lncRNA variation differentially regulates CCR5, influencing HIV disease outcome. Nat Immunol. 2019;20(7):824-834.
- Eissa S, Almusharraf AY, Zourob M. A comparison of the performance of voltammetric aptasensors for glycated haemoglobin on different carbon nanomaterials-modified screen printed electrodes. Mater Sci Eng C Mater Biol Appl. 2019;101:423-430.
- Eissa, S.; et al. “Development of Impedimetric Immunosensors for the Diagnosis of DOCK8 and STAT3 Related Hyper‐Immunoglobulin E Syndrome.” Electroanalysis. 2018;30:2021-2027.











