Native Proteins
🧪 CDA002
Source: Human Ascites Fluid
Species: Human
Tag: Non
Conjugation:
Protein Length:
🧪 CST3-4309H
Source: Human
Species: Human
Tag: Non
Conjugation:
Protein Length:

🧪 THBS1-31514TH
Source:
Species: Human
Tag: Non
Conjugation:
Protein Length:

🧪 KNG1-1844H
Source: Human Plasma
Species: Human
Tag: Non
Conjugation:
Protein Length:
🧪 ANXA11-6905H
Source: E.coli
Species: Human
Tag: Avi
Conjugation:
Protein Length: 1-505 aa

🧪 CED026
Source: Kidney
Species: Bovine
Tag:
Conjugation:
Protein Length:

🧪 CGA-8163H
Source: Human Pregnancy Urine
Species: Human
Tag: Non
Conjugation:
Protein Length:

🧪 LTF-8196H
Source: Human Breast Milk
Species: Human
Tag: Non
Conjugation:
Protein Length:

🧪 SHBG-8259H
Source: Human Serum
Species: Human
Tag: Non
Conjugation:
Protein Length:
🧪 HRP-8336h
Source: horseradish
Species: Horseradish
Tag: Non
Conjugation:
Protein Length:
🧪 ACTB-325H
Source: Human Platelets
Species: Human
Tag: Non
Conjugation:
Protein Length:
🧪 CKMB-165H
Source: Human Cardiac
Species: Human
Tag: Non
Conjugation:
Protein Length:

🧪 gGT-184B
Source: Bovine Kidney
Species: Bovine
Tag: Non
Conjugation:
Protein Length:
🧪 PRL-111S
Source: Sheep Pituitary
Species: Sheep
Tag: Non
Conjugation:
Protein Length:
🧪 GPOSP-40
Source:
Species:
Tag:
Conjugation:
Protein Length:

SubCategories
- ADV Protein
- Aerococcuss viridans Protein
- Agaricus bisporus Protein
- Aleuria aurantia Protein
- Arthrobacter globiformis Protein
- Arthrobacter sp. Protein
- Artocarpus integrifolia Protein
- Aspergillus Niger Protein
- Atlantic salmon Protein
- Bacillus Protein
- Bovine Protein
- Canavalia ensiformis Protein
- Candida Protein
- Canine Protein
- Chicken Protein
- Clostridium Protein
- Cynomolgus Monkey Protein
- Datura stramonium Protein
- Dinophyceae sp. Protein
- Dolichos Biflorus Protein
- Donkey Protein
- Erythrina cristagalli Protein
- Escherichia coli Protein
- Feline Protein
- FSME Protein
- Galanthus nivalis Protein
- Goat Protein
- Griffonia Simplicifolia Protein
- Guinea Pig Protein
- H3N2 Protein
- Hamster Protein
- HBV Protein
- Hippeastrum hybrid Protein
- Horse Protein
- horseradish Protein
- HSV Protein
- Human Protein
- IAV Protein
- Influenza B virus Protein
- Jack Bean Protein
- Lotus Tetragonolobus Protein
- Maackia Amurensis Protein
- Maclura Pomifera Protein
- Megathura crenulata Protein
- Monkey Protein
- Mouse Protein
- MV Protein
- Narcissus Pseudonarcissus Protein
- Other Protein
- Ovine Protein
- Parainfluenza Virus Protein
- Peanut Protein
- Phaseolus vulgaris Protein
- Pigeon Protein
- Pisum Sativum Protein
- Porcine Protein
- Potato Protein
- Pseudomonas sp. Protein
- Rabbit Protein
- Rat Protein
- Red algae Protein
- Rhesus Monkey Protein
- Ricinus Communis Protein
- RV Protein
- S. aureus Protein
- Sambucus nigra Protein
- Sheep Protein
- Snake Protein
- Solanum Tuberosum Protein
- Soybean Protein
- Streptococcus Protein
- Swine Protein
- T.gondii Protein
- Tomato Protein
- Turkey Protein
- Ulex europaeus Protein
- Vibrio Protein
- Vicia Villosa Protein
- VZV Protein
- Wheat Germ Protein
- Wisteria floribunda Protein
- Y. enterocolitica Protein
- Yeast Protein
Background
Overview
Native proteins, also known as natural proteins, are proteins that are purified from their natural sources. These proteins can come from whole blood, serum, and plasma of humans, animals, or reptiles. In addition, microbial and non-microbial (e.g., egg whites, animal skin) are also good sources of native protein. Native proteins are characterized by the fact that they undergo a series of natural in vivo modification processes such as precursor protein cleavage, disulfide bond formation, glycosylation, etc.

In biochemistry, the native state of a protein is its properly folded and assembled form with operative structure and function. In biophysics, the native state of a protein is defined by a low amount of entropy with a relatively low intramolecular enthalpy. Compared to recombinant proteins generated using protein engineering techniques, native proteins are obtained from proper organisms and are unaltered by outer factors such as heat, chemicals and enzyme reaction, and retain their naturally folded state.
Functions
There are many different types of native proteins, and every native protein has a specific function. This is crucial for living organisms.
Building cells and tissues: Proteins are the main components of cells and tissues, and they play a key role in the structural maintenance, growth and repair of cells.
Catalyzing biological reactions: Certain proteins, such as enzymes, are able to accelerate chemical reactions and act as biocatalysts that are essential for metabolic processes.
Regulation of metabolic processes: Proteins are involved in regulating metabolic activities in the body and maintaining physiological balance.
Provide immune protection: Proteins such as antibodies play a role in the immune system to protect the body from pathogens.
Carry out material transport and storage: for example, hemoglobin is responsible for the transport of oxygen, while other proteins are involved in the storage of nutrients.
Support movement and structure: Muscle proteins such as actin and myosin are involved in muscle contraction and support the movement of the body.
Involved in information transfer: Certain proteins such as hormones and receptors are involved in signaling between cells, regulating multiple functions of the organism.

Applications
Scientific Research: Native protein-DNA couplings are used in immunoPCR. Native proteins can be a control for immunobioassays such as ELISA and other antibody assays. Also they can used for the study of protein-protein interactions or cell-based functional assays.
Medical Research: Native proteins are used in research to study their structure, function, and interactions with other molecules. This knowledge can lead to the development of new drugs, materials, and technologies. In addition, by detecting certain protein markers, it is possible to diagnose disease, monitor disease progression, and evaluate the effectiveness of treatment.
Drug Development: By studying and understanding the structure and function of proteins in their natural state, Native proteins can be used for the screening and confirmation of drug targets. In addition, natural polymers, especially protein-based biomaterials, can better solve the problems of synthetic materials, so they have a wide range of applications in drug delivery.
Food Industry: Native proteins are an important part of a healthy diet, as they provide the body with the building blocks it needs to function properly. They can be found in a variety of foods, including meat, fish, eggs, dairy products, and plant-based sources such as beans and nuts.
Biotechnology: Native proteins are used in the production of biofuels, enzymes, and other industrial chemicals. They can also be used in genetic engineering to create new strains of plants and animals with desirable traits.
Environmental Science: Native proteins are used in environmental monitoring and remediation efforts, such as detecting pollutants in water and soil or removing heavy metals from contaminated sites.

Case Study
Case Study 1: Active Native Bovine TSH Protein (TSH-1315B)
Thyroid hormones (THs) are crucial regulators of human metabolism and early development. During the safety assessment of plant protection products, the human relevance of chemically induced TH perturbations observed in test animals remains uncertain. European regulatory authorities request follow-up in vitro studies to elucidate human-relevant interferences on thyroid gland function or TH catabolism through hepatic enzyme induction. However, human in vitro assays based on single molecular initiating events poorly reflect the complex TH biology and related liver-thyroid axis. To address this complexity, the researchers present human three-dimensional thyroid and liver organoids with key functions of TH metabolism. The thyroid model resembles in vivo-like follicular architecture and a TSH-dependent triiodothyronine synthesis over 21 days, which is inhibited by methimazole. Activation of the nuclear receptors PXR and AHR was demonstrated via the induction of specific CYP isoenzymes by rifampicin, pregnenolone-16α-carbonitrile, and β-naphthoflavone. However, this nuclear receptor activation, assumed to regulate UDP-glucuronosyltransferases and sulfotransferases, appeared to have no effect on gT4 and sT4 formation in this human-derived hepatic cell line model.
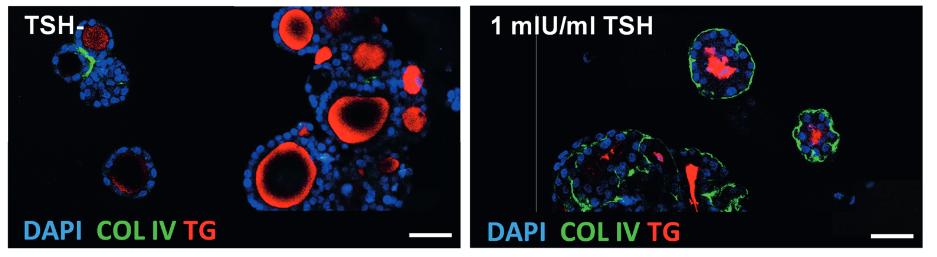
(Julia Kühnlenz, 2023)
Fig1. Representative immunofluorescent staining of the unstimulated (TSH-) and TSH-stimulated (1 mIU/mL TSH) 3D thyroid model after being statically cultured for 21 days. Protein expression of thyroglobulin (TG, red), collagen IV (COL IV, green), and nuclei (DAPI, blue) is shown. Scale 30 μm.
Case Study 2: Native Human Breast Milk Lactoferrin APO (LTF-8196H)
The global spread of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and the associated disease COVID-19, requires therapeutic interventions that can be rapidly identified and translated to clinical care. Traditional drug discovery methods have a >90% failure rate and can take 10 to 15 y from target identification to clinical use. In contrast, drug repurposing can significantly accelerate translation.
From a library of 1,425 US Food and Drug Administration (FDA)-approved compounds and clinical candidates, they identified 17 hits that inhibited SARS-CoV-2 infection and analyzed their antiviral activity across multiple cell lines. Additionally, they found that inhibitors of the Ras/Raf/MEK/ERK signaling pathway exacerbate SARS-CoV-2 infection in vitro.
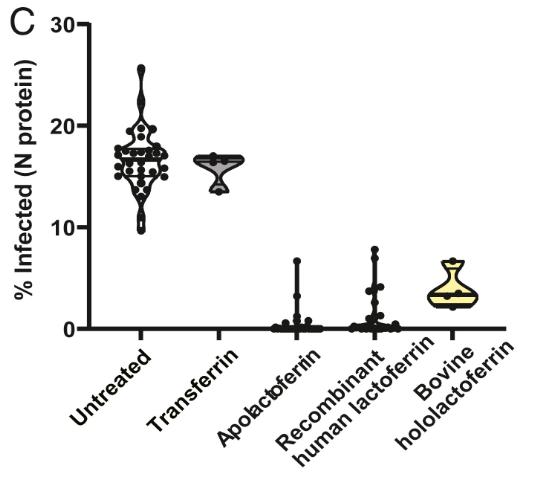
(Carmen Mirabelli, 2021)
Fig2. Percentage of SARS-CoV-2–infected Huh7 cells upon treatment with bovine apolactoferrin and hololactoferrin, native human lactoferrin, and transferrin at a concentration of 6,250 nM.
Case Study 3: Native Human Chorionic Gonadotropin (CGA-8163H)
Both stray and free-roaming owned dogs contribute to the serious global dog overpopulation problem. Many dog owners are unwilling to have their pet castrated for various reasons, including a reluctance to have their dog's behavior changed. A non-surgical method of sterilizing both stray and owned dogs would help to prevent unwanted litters. The aim of this study was to compare the use of a solution of 20% CaCl2 in 95% ethanol injected into the testicles or into the head of the epididymis. The animals were examined at 0, 3, 6, and 9 months for sperm quality, concentration of testosterone in serum, and side effects; at 0 and 5 months with contrast-enhanced ultrasound (CEUS) to enhance the morphological aspects/alteration of the testicular parenchyma or epididymis; and at 9 months when all were castrated for histological examination. All dogs treated with CaCl2 became sterile with azoospermia achieved over the 9-month study. The concentration of testosterone in serum significantly decreased following intratesticular treatment with CaCl2. No adverse effects were noted.
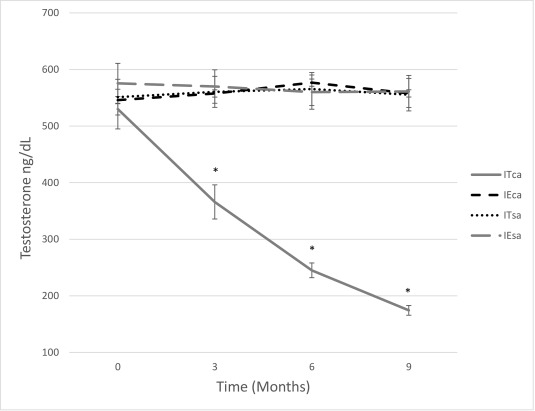
(Raffaella Leoci, 2019)
Fig3. Effects of injection on serum testosterone levels in each group over time. No significant differences in testosterone levels were found over time in the other three groups: intraepididymal injection CaCl2 (IEca), intraepididymal injection saline (IEsa), and intratesticular injection saline (ITsa) (each n = 37).
Case Study 4: Native Human Serum Sex Hormone Binding Globulin (SHBG-8259H)
Vitamin D deficiency is associated with an increased risk of prostate cancer mortality and is hypothesized to contribute to prostate cancer aggressiveness and disparities in African American populations. The prostate epithelium was recently shown to express megalin, an endocytic receptor that internalizes circulating globulin-bound hormones, which suggests regulation of intracellular prostate hormone levels. This contrasts with passive diffusion of hormones that is posited by the free hormone hypothesis. Here, the researchers demonstrate that megalin imports testosterone bound to sex hormone-binding globulin into prostate cells.
Megalin expression was regulated and suppressed by 25-hydroxyvitamin D (25D) in cell lines, patient-derived prostate epithelial cells, and prostate tissue explants. In patients, the relationships between hormones support this regulatory mechanism, as prostatic DHT levels are higher in African American men and are inversely correlated with serum 25D status. Megalin levels are reduced in localized prostate cancer by Gleason grade.
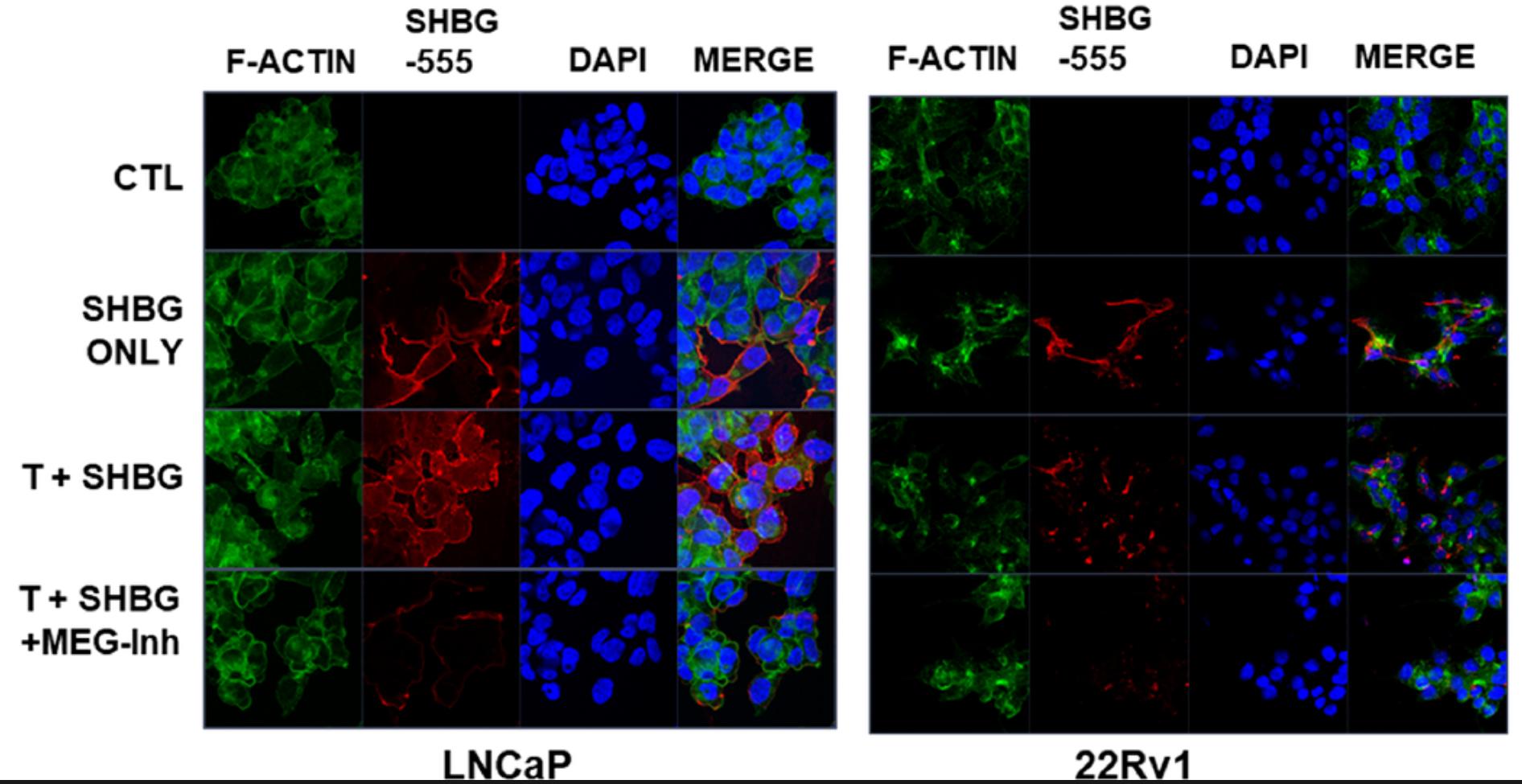
(Jason Garcia, 2023)
Fig4. Prostate cells express megalin and import SHBG-bound T. Visualization (× 63) of SHBG-555 (red) import into cells with DAPI (blue) nuclear and F-actin (green) cytoskeletal counterstains. LNCAP and 22Rv1 cells at 80% confluency were incubated with 25 nmol/L T ± 125 nmol/L human SHBG.
Case Study 5: Native Human MUC16 protein (MUC16-1H)
To investigate whether pre-season self-reported mental toughness and self-regulation strategies predicts post-season stress, mental health, group cohesion, and satisfaction in varsity athletes. Sixty-seven varsity athletes from a U-Sport Canadian university. Athletes completed pre- and post-season measures of mental toughness, emotion regulation, self-control, group cohesion, and satisfaction. Multiple mediation models were employed during analyses. Pre-season emotion regulation was a significant mediator for the relationships between mental toughness and post-season ratings of perceived stress, perceived mental health, and group cohesion. Self-control was not a significant mediator. In conclusion, emotion regulation plays a significant role in an athlete's experience of perceived stress, mental health issues, and their ratings of group cohesiveness. Research on student-athlete health may benefit from examining emotion regulation training techniques.
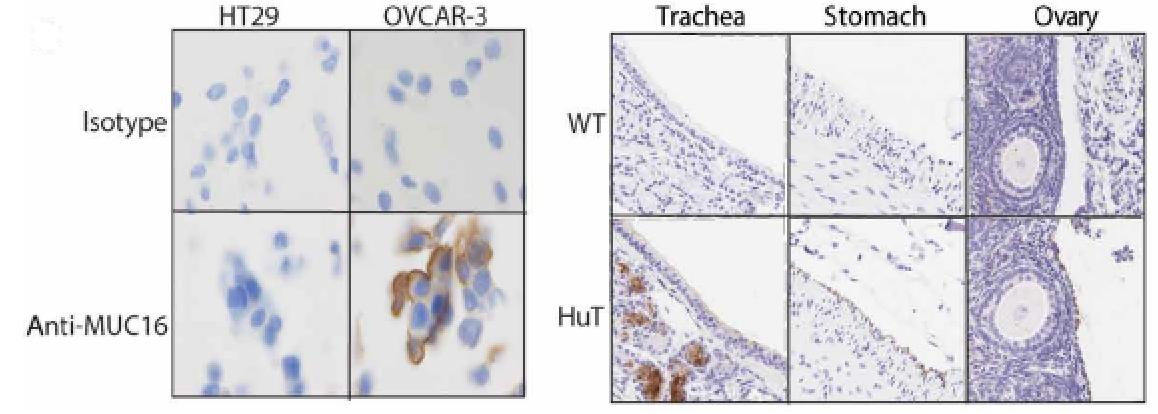
(Alison Crawford, 2023)
Fig5. Left: IHC analysis of MUC16 expression on OVCAR-3 and HT29 cells. Right: IHC analysis
of MUC16 expression in the trachea, stomach, and ovary from wild-type (WT) or HuT mice.
Advantages
- Wide Coverage: More than 1700 native proteins from over 20 different species and nearly 100 different sources.
- High Quality: Our native proteins have been tested with different methods to ensure intergrity and high purity.
- Experienced: We have professional research team with experience of many years in the field of molecular and cell biology.
- Customized services: Our consulting services can provide customized solutions to meet customers' specific needs and requirements.
FAQ
-
Q: What is the difference between native protein and synthetic protein?
A: Native proteins are those that occur naturally in living organisms, while synthetic proteins are created in a laboratory through genetic engineering or chemical synthesis. Native proteins have evolved over millions of years to perform specific functions in the body, while synthetic proteins are designed to mimic or enhance these functions. Native proteins are typically more complex and diverse than synthetic proteins, which can be limited in their structure and function. Additionally, native proteins may have unintended side effects or interactions with other molecules in the body, while synthetic proteins can be carefully engineered to minimize these risks.
-
Q: What are the sources of native protein?
A: The sources of native protein are animals and plants. Animals that are commonly consumed for their protein include chicken, beef, pork, fish, and eggs. Plant-based sources of protein include beans, lentils, tofu, nuts, and seeds.
-
Q: What is the extraction and processing process of native proteins?
A: The extraction and processing of native proteins involve several steps: harvesting the raw materials, cleaning and trimming, grinding or chopping, extracting the protein, filtration and separation, washing and desalting, drying.
-
Q: What are the methods and techniques for structural analysis of native proteins?
A: Structural analysis methods are mainly divided into experimental methods and prediction methods. The experimental methods mainly include X-ray crystal diffraction, nuclear magnetic resonance and cryo-electron microscopy.
Resources
References
- Mirabelli C.; et al. Morphological cell profiling of SARS-CoV-2 infection identifies drug repurposing candidates for COVID-19. Proc Natl Acad Sci U S A. 2021;118(36):e2105815118.
- Leoci R.; et al. Effects of intratesticular vs intraepididymal calcium chloride sterilant on testicular morphology and fertility in dogs. Theriogenology. 2019;127:153-160.