Cellular Senescence and Pathways in Aging
Creative BioMart Cellular Senescence and Pathways in Aging Product List
Immunology Background
About Cellular Senescence and Pathways in Aging
Cellular senescence and pathways in aging are a group of molecules that control and regulate physiological rhythms in living organisms. Physiological Cellular senescence is a state of irreversible growth arrest characterized by the inability of cells to divide and proliferate. It is a complex biological process that plays a significant role in aging and age-related diseases. Senescent cells accumulate in tissues over time, contributing to tissue dysfunction and the development of various age-related pathologies. Understanding the mechanisms and pathways involved in cellular senescence is crucial for unraveling the molecular basis of aging and identifying potential therapeutic strategies for age-related disorders.
Cellular senescence is closely associated with the aging process. As cells age, they undergo various changes that lead to the loss of their proliferative capacity and functional decline. Senescent cells display distinct morphological features, such as enlarged and flattened morphology, as well as increased senescence-associated beta-galactosidase (SA-β-gal) activity. These cells also exhibit alterations in gene expression profiles, including the upregulation of cell cycle inhibitors, such as p16INK4a and p21Cip1/Waf1, and the senescence-associated secretory phenotype (SASP), characterized by the secretion of pro-inflammatory cytokines, growth factors, and matrix metalloproteinases.
The accumulation of senescent cells in tissues contributes to the aging process. Senescent cells exhibit impaired tissue repair and regeneration capabilities, leading to the progressive decline of organ function. Additionally, senescent cells secrete various factors that can have detrimental effects on neighboring cells and tissues, promoting chronic inflammation, tissue fibrosis, and other age-related pathologies.
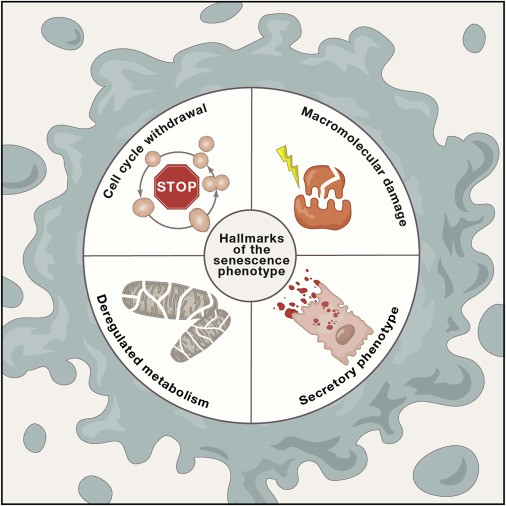 Fig.1 The Hallmarks of the Senescence Phenotype. (Gorgoulis V, et al., 2019)
Fig.1 The Hallmarks of the Senescence Phenotype. (Gorgoulis V, et al., 2019)
Senescent cells exhibit the following four interdependent hallmarks: (1) cell-cycle withdrawal, (2) macromolecular damage, (3) secretory phenotype (SASP), and (4) deregulated metabolism
Pathways Involved in Cellular Senescence and Aging
Several key pathways and mechanisms contribute to the development and maintenance of cellular senescence:
- Telomere Attrition: Telomeres are repetitive DNA sequences that protect the ends of chromosomes. During each cell division, telomeres progressively shorten due to the "end replication problem." Once telomeres reach a critical length, cells undergo replicative senescence, triggering a permanent growth arrest. Telomere attrition is considered a hallmark of cellular aging.
- DNA Damage Response (DDR): Accumulation of DNA damage is another trigger for cellular senescence. DNA damage activates the DDR pathway, leading to the activation of checkpoint kinases, such as ATM (ataxia-telangiectasia mutated) and ATR (ataxia-telangiectasia and Rad3-related), and the induction of cell cycle arrest. Persistent DNA damage, arising from various sources, including oxidative stress and genotoxic insults, can lead to chronic activation of DDR and the establishment of senescence.
- Oncogene-Induced Senescence (OIS): Activation of certain oncogenes, such as Ras and BRAF, can induce cellular senescence as a tumor-suppressive mechanism. Oncogene activation triggers a cascade of signaling events that lead to cell cycle arrest and senescence. OIS is commonly observed in benign tumors and premalignant lesions.
- Senescence-Associated Secretory Phenotype (SASP): Senescent cells secrete a variety of factors collectively known as SASP. These include pro-inflammatory cytokines, chemokines, growth factors, and matrix metalloproteinases. SASP components can promote chronic inflammation, alter tissue microenvironments, and contribute to age-related pathologies.
- Senescence-Associated Chromatin Remodeling: Senescence is accompanied by extensive changes in chromatin structure and gene expression. These changes involve alterations in histone modifications, DNA methylation patterns, and the formation of senescence-associated heterochromatin foci (SAHF). Senescence-associated chromatin remodeling contributes to the stable maintenance of the senescent state.
- Mitochondrial Dysfunction: Mitochondrial dysfunction, characterized by increased reactive oxygen species (ROS) production and impaired mitochondrial function, is associated with cellular senescence. ROS accumulation can induce cellular senescence by causing oxidative damage to macromolecules and activating stress response pathways.
Understanding the intricate signaling networks and pathways involved in cellular senescence is essential for deciphering the molecular mechanisms underlying aging. Targeting these pathways holds promise for developing interventions to alleviate age-related disorders and promote healthy aging.
Biological Functions or Roles of Cellular Senescence and Pathways in Aging
Cellular senescence and the pathways associated with it play important biological functions and roles in the aging process. Here are some key functions and roles of cellular senescence in aging:
Tumor Suppression: Cellular senescence acts as a tumor-suppressive mechanism by permanently arresting the growth of cells that have accumulated DNA damage or activated oncogenes. Senescence prevents the proliferation of potentially cancerous cells, thereby inhibiting the development and progression of tumors. Senescence induction in response to oncogene activation, known as oncogene-induced senescence (OIS), helps to counteract tumorigenesis.
Tissue Remodeling and Repair: Senescent cells are involved in tissue remodeling and repair processes. During development and wound healing, cellular senescence can promote tissue remodeling by facilitating the clearance of senescent or damaged cells. Senescent cells secrete factors that attract immune cells to clear the senescent cells or promote tissue regeneration and repair.
Aging-Associated Inflammation: Senescent cells secrete a variety of pro-inflammatory cytokines, chemokines, and growth factors as part of the senescence-associated secretory phenotype (SASP). Although SASP serves beneficial roles in tissue repair and immune surveillance, chronic accumulation of senescent cells and persistent SASP can lead to chronic low-grade inflammation, known as inflammation. Inflammation contributes to tissue dysfunction, age-related diseases, and the overall aging process.Stem Cell Exhaustion: Cellular senescence is associated with the decline of tissue regeneration and the depletion of stem cell pools. As stem cells undergo replicative senescence or become senescent due to environmental cues, their regenerative capacity diminishes. This contributes to tissue aging and impaired tissue maintenance and repair.
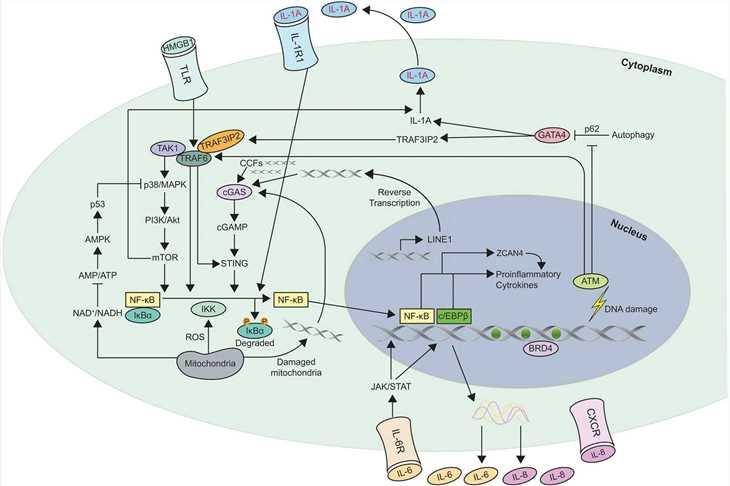 Fig.2 Schematic of the different mechanisms involved in Senescence Associated Secretory Phenotype (SASP) regulation. (Kumari R, et al., 2021)
Fig.2 Schematic of the different mechanisms involved in Senescence Associated Secretory Phenotype (SASP) regulation. (Kumari R, et al., 2021)
Maintenance of Tissue Homeostasis: Cellular senescence helps to maintain tissue homeostasis by eliminating damaged or dysfunctional cells. Senescent cells undergo permanent growth arrest, preventing them from contributing to tissue dysfunction or tumorigenesis. By removing these cells, senescence contributes to the overall health and function of tissues.
Mitigating Genomic Instability: Cellular senescence acts as a safeguard against genomic instability. By halting the proliferation of cells with DNA damage, senescence prevents the transmission of genetic abnormalities to daughter cells. This prevents the accumulation of further mutations and the propagation of damaged genomes.
Epigenetic Regulation: Senescent cells undergo extensive epigenetic changes, including alterations in DNA methylation patterns and histone modifications. These changes contribute to the stable maintenance of the senescent state and help reinforce the growth arrest and senescence-associated gene expression patterns.
Contribution to Aging-Related Diseases: The accumulation of senescent cells in tissues over time is associated with the development of various age-related diseases, including cardiovascular diseases, neurodegenerative disorders, and metabolic disorders. Senescent cells and their secreted factors contribute to tissue dysfunction and the pathogenesis of these diseases.
Understanding the biological functions and roles of cellular senescence and its associated pathways in aging is crucial for developing strategies to mitigate the negative consequences of senescence and promote healthy aging. Targeting senescent cells or modulating senescence-associated pathways holds promise for interventions to delay aging-related diseases and improve overall healthspan in aging individuals.
Research Tools for Cellular Senescence and Pathways in Aging
The cellular senescence mechanism involves a variety of signal controllers and regulatory molecular networks. These controllers include but are not limited to p53/p21, p16/Rb, mTOR, DNA damage, and oxidative reactions. Through these reagents, cells collect internal and external stimuli and trigger related signaling cascades, ultimately leading to the induction of cell cycle molecules and dramatic activity changes. and ischemic or proliferative structures.
To support research in the field of cellular senescence, Creative BioMart provides recombinant proteins and other reagents and tools related to cellular senescence to help researchers conduct research on cellular senescence and support the development of treatments for related diseases. Please click on the relevant aspect molecule/target to view all research reagents. If you have any questions or are interested in these products, please feel free to contact us and our customer service team will be happy to serve you. We look forward to interacting with you!
References:
- Kumari R, Jat P. Mechanisms of Cellular Senescence: Cell Cycle Arrest and Senescence Associated Secretory Phenotype. Front Cell Dev Biol. 2021;9:645593.
- Gorgoulis V, Adams PD, Alimonti A, et al. Cellular Senescence: Defining a Path Forward. Cell. 2019;179(4):813-827.

