Assay kits
SubCategories
Background
Overview
A test kit is a medical device used for qualitative or quantitative detection of biological samples such as human body fluids and blood to reflect the health status of an individual. These kits usually contain all the chemicals, materials, and instructions needed to perform a specific test. Their main advantages are simple operation, short detection time, suitable for clinical medical institutions, centers for disease control and prevention, inspection and testing institutions and other units.
When using the test kit, it is also necessary to pay attention to some matters, such as wearing protective equipment such as masks and gloves during operation to avoid cross infection; Ensure that the operating surface is clean and avoid testing in an unclean environment; Do not expose the kit to high temperature, humidity, direct sunlight and other conditions, so as not to affect the accuracy of the test results.

Procedure of Kits
The method of using a test kit usually includes several basic steps. The details are as follows:
Preparation: Wash your hands or use a quick hand sanitizer before using the test kit to prevent contamination of the sample or reagent. Read the instructions to understand how different brands or types of kits might operate. Check whether the kit and contents are within the shelf life, whether there is any missing or damaged, to ensure the effectiveness and integrity of the kit. Ensure that the temperature and humidity of the test environment meet the requirements to avoid impact on the test results.
Experimental procedure: Unpack the sample extraction solution and pour all of it into the sample extraction tube. Follow instructions and collect samples. The collected sample is placed into a sample extraction tube containing the extraction solution and mixed thoroughly to release the indicator. The liquid in the sample extraction tube is characterized. Judge the results according to the instructions.
Result interpretation: The test result may be a data range or a color mark. The test result can be obtained according to the corresponding data and results given in the instruction manual; If none is found, the test is invalid and needs to be retested.
In summary, it is important to follow the instructions when using the test kit, because different brands of kits may have different operating processes and results interpretation. At the same time, ensure that the operation is carried out in a suitable environment to ensure the accuracy of the test results.
Advantages of Kits
The detection kit has the following advantages over other detection methods:
Easy to operate: Kits are often designed to be user-friendly and can be performed by non-specialists under simple instructions, which greatly reduces the reliance on professional technicians. And the result judgment is objective, reducing the possibility of human error.
Rapid detection: The kits are designed to make the testing process much faster, and some kits are able to provide results within minutes, which is very useful in situations where rapid diagnosis is required.
High sensitivity: With the progress of science and technology, the detection sensitivity of the kit continues to improve, and it can detect lower concentrations of pathogens or other substances under test.
High selectivity: Modern kits are able to specifically identify target substances and accurately detect specific ingredients even in complex mixtures.
Equipment portability: The supporting testing equipment is becoming more and more intelligent and miniaturized, easy to carry and field use, especially suitable for field or resource limited environment for testing.
Comprehensive detection: Some kits can detect multiple components at the same time, and this integrated technology greatly shortens the detection cycle and improves efficiency.
Research Progress
The evolution of test kits can be traced to the evolution of the In Vitro Diagnostics (IVD) industry. The in vitro diagnostics industry has undergone four major technological revolutions, each of which has led to significant advances and innovations in the development of test kits.
Biochemical diagnosis: This is the earliest in vitro diagnostic technology, mainly relying on chemical reactions to detect various biochemical indicators in the blood, such as blood sugar, cholesterol and so on. These tests usually require specialized laboratory equipment and technicians.
Enzymology diagnosis: With the development of enzymology, people began to use specific enzymes as biocatalysts to carry out more accurate biochemical reactions, thus improving the sensitivity and specificity of detection.
Immunodiagnostics: The advent of immunodiagnostic technology marks an important stage in the development of test kits. By using specific responses of antibodies and antigens, trace amounts of pathogens or biomarkers can be detected. The application of this technology is very wide, including infectious diseases, autoimmune diseases and many other fields.
Molecular diagnosis: The development of molecular diagnostic technology, especially the invention of polymerase chain reaction (PCR) technology, makes the detection kit can detect a very small number of nucleic acid sequences, which greatly promotes the diagnostic ability in the field of genetic diseases, infectious diseases and so on.
With the advancement of technology, the application of detection kits has also continued to expand, from the initial biochemical detection to the current ability to detect a variety of pathogens, tumor markers, genetic variants, and so on. Especially during the new coronavirus epidemic, the development and application of novel coronavirus antigen test kits have demonstrated the important role of test kits in responding to public health emergencies.

Future Development
With the continuous progress of science and technology, the accuracy, convenience and rapidity of test kits will continue to improve, and more new detection technologies and products may be developed in the future to meet the growing medical and health needs. Future technological innovations in test kits may focus on the following directions:
High-throughput Sequencing technology (NGS): With the development of high-throughput sequencing technology, the application of NGS (Next-Generation Sequencing) in the field of molecular diagnostics is becoming more and more widespread. NGS can detect thousands of genes at the same time, which is suitable for many fields such as tumor concomitant diagnosis, genetic disease screening, infectious disease detection and so on. In the future, NGS technology may further reduce costs, improve accuracy and simplify operational processes, making test kits more efficient and widely available.
Point-of-care testing (POCT): Point-of-care technology allows testing to be done quickly at the sampling site, reducing sample transportation and laboratory processing time. In the future, POCT technology may be further developed to provide more rapid, convenient and accurate test results, especially in the fields of emergency, critical illness, infectious disease prevention and control.
Biosensor technology: Biosensors can detect specific biomolecules with high sensitivity, and more biosensors may be developed in the future to detect various biomarkers, thus promoting the technological innovation of detection kits.
Nanotechnology: The application of nanotechnology in the biomedical field brings new possibilities for detection kits, such as using nanoparticles as signal amplifiers or carriers to improve the sensitivity and selectivity of detection.

What We Offer
Creative BioMart offers thousands of assay kits in a variety of formats, including Activity, Metabolism, Epigenetic, Cellular, Signaling and Detections.
Our assay kits are ready to use and provide all necessary reagents, instructions, plates and analysis protocols to help you quickly obtain accurate and reproducible results. With our assay kits, you will receive highly sensitive, precise, and accurate results across a wide dynamic range with fast, efficient processing.
Activity Kits: They are used to evaluate the physiological state of cells, including cell growth, proliferation, toxicity, etc. These kits are typically based on a cell's metabolic activity, membrane integrity, or specific cell function indicators. Widely used in drug screening, cell biology research, toxicity testing, cell culture conditions optimization and other fields
Metabolism Kits: They are used to measure the metabolic activities of cells or organisms, such as energy metabolism, glucose metabolism, lipid metabolism, etc. These kits can provide detailed information about cellular metabolic pathways. They are widely used in the study of metabolic related diseases, assessment of metabolic disorders, drug metabolism research and nutrition research.
Epigenetic Kits: Focus on epigenetic changes such as DNA methylation, histone modification, etc. These kits can detect mechanisms that regulate the expression of specific genes. They are of great value in the research of cancer, genetic disease, developmental biology and the influence of environmental factors on gene expression.
Cellular Kits: They usually contain a range of reagents and tools for the study of cell culture, cell proliferation, apoptosis, etc. They are widely used in cell biology, molecular biology, drug development and disease model research.
Signaling Kits: They are used to study the activation state of intracellular signal transduction pathway, including kinase and phosphatase activity, expression and regulation of signal molecules. They play an important role in cell signaling research, drug screening and disease mechanism research.
Detections Kits: Including a variety of kits for detecting specific biomarkers, pathogens, genetic variants, and more. These kits are usually highly sensitive and specific. They are very important in clinical diagnosis, disease surveillance, epidemiological research, genetic disease screening and other fields.
Case Study
Case Study 1: Cathepsin D Activity Fluorometric Assay Kit (Kit-0182)
Proteasomes and lysosomes are responsible for homeostasis of proteins, lipids and carbohydrates within cells. Many reports have shown that the proteolytic pathway alters function during neurodegeneration and aging. Lewy body dementia (DLB) is one of the main forms of dementia, and the proteolytic changes of DLB have not been fully studied. This study shows that the components of proteasome and lysosome selectively alter gene expression and enzyme function. Specifically, PSMB8, an inducible proteasome beta subunit, showed elevated mRNA levels and protein levels in the DLB brain compared to age-matched controls. The caspase-like peptidase activity of DLB was significantly decreased by the test kit, and there was no statistical significance in trypsin-like/chemotactic trypsin-like activity. The mRNA levels of cathepsin B and D in the lysosomes of DLB were increased, but only the activity of cathepsin B was increased.
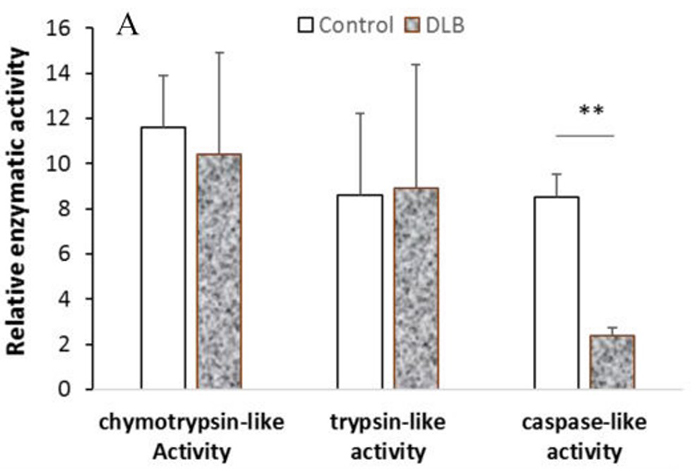
(Qunxing Ding, 2018)
Fig1. Cell lysate was isolated from the SMTG tissue of each subject and the activity of each enzyme was analyzed.
Case Study 2: Creatine Assay Kit (Kit-0259)
Antiphospholipid antibodies (apl) cause severe autoimmune diseases characterized by vascular lesions and pregnancy complications. In this study, we found that endosomal lysodiphosphatidic acid (LBPA), presented by CD1-like endothelial protein C receptor (EPCR), is a pathogenic cell surface antigen that is recognized by apl and induces thrombus formation and endosomal inflammatory signaling. apl binds to EPCR-LBPA expressed on innate immune cells to maintain interferon and Toll-like receptor 7-dependent B1a cell expansion and autoantibody production. Albumin in spot-urine samples was detected with mouse albumin ELISA and normalized to creatinine content with the Creatine Assay Kit.
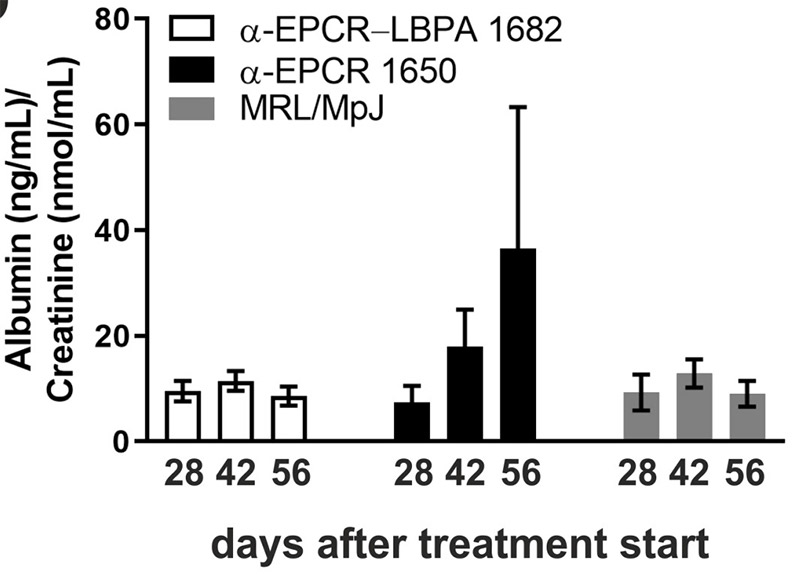
(Nadine Müller-Calleja, 2021)
Fig2. Albuminuria in MRL/MpJ control mice and in MRL-Faslpr mice treated at the age of four weeks for six weeks with the indicated antibodies.
Case Study 3: Thiol Fluorometric Detection Kit (Kit-0816)
In methanogens, one form of gene expression is the expression of copper-regulated alternative forms of methane monooxygenase. A copper-binding compound called methanomycin plays a key role in the absorption of copper by methanoxidation bacteria. Methanobactin is a ribosome synthetic post-translational modified peptide (RiPP) formed by binding the X-Cys dipeptide sequence to copper. The gene encoding methanesin precursor peptides, mbnA, is part of a gene cluster, but the role of other genes in methanesin biosynthesis is unclear. To elucidate the function of these genes, a marker-free deletion of mbnABCMN was constructed in trichosporium Methylosinus OB3b, and then homologous expression of mbnABCM using a wide host range clonal vector was used to determine the function of mbnN, annotated as encoding aminotransferase. The methicotinoid produced by this strain is very different from the wild-type methicotinoid, lacking C-terminal methionine and forming only one of the two oxazolone rings, the results of thiol determination in methanobactin from M. trichosporium OB3b wild-type, ΔmbnAN + pWG101, and ΔmbnAN + pWG102 strains with kits are really different.

(Wenyu Gu, 2016)
Fig3. Quantification of thiol groups in methanobactin from M. trichosporium OB3b wild-type, ΔmbnAN + pWG101, and ΔmbnAN + pWG102 strains as isolated and following reduction by tris(2-carboxyethyl)phosphine under anaerobic conditions.
Case Study 4: Ammonia Colorimetric Assay Kit (Kit-0085)
Hepatic encephalopathy (HE) is a neuropsychiatric complication of cirrhosis associated with poor survival and adverse effects on quality of life (QOL). The drawbacks of long-term use of rifaximin in HE require the search for alternative therapies. In this context, the aim of this study was to evaluate the safety and efficacy of nitazoxanide (NTZ) versus rifaximine (RFX) in preventing recurrence of HE and to assess its impact on quality of life. This prospective, randomized, double-blind controlled study included 60 patients randomly assigned to receive rifaximin 550 mg twice daily (group 1; N = 30) or nidazolide 500 mg twice daily (2 groups; N = 30) for 24 weeks. During the study period, patients were monitored for neurological symptoms, mental status, and work performance. Serum levels of HE triggers (ammonia, TNF-α, and octopamine) were assessed. Patients' health-related quality of life was also assessed.
The results showed that six months after treatment, patients treated with NTZ showed statistically significant improvements in chess scores and mental state. In addition, NTZ showed statistically significant reductions in serum levels of ammonia (as detected by the kit), TNF-α, and octopamine compared with rifaximine. In terms of quality of life, the Chronic Liver Disease Questionnaire (CLDQ) scores improved in the NTZ group. Both groups had mild, manageable side effects.
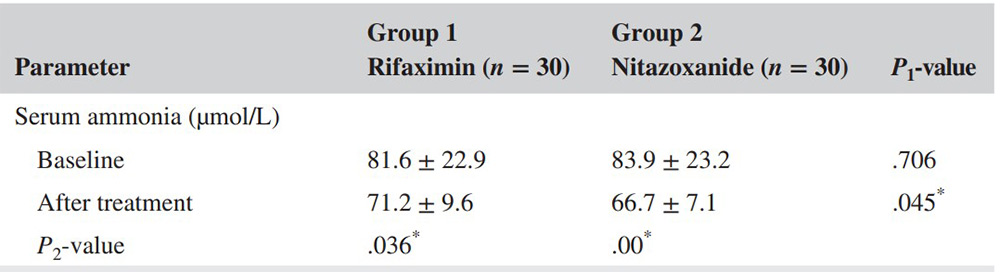
(Khadija A M Glal, 2021)
Fig4. During the current study, the nitazoxanide group showed statistically significant reduction in serum ammonia level as compared to rifaximin group.
Advantages
- Multiple types: We offer thousands of kits in a variety of formats and provide all necessary reagents, instructions, plates and analysis protocols to help you quickly obtain accurate and reproducible results.
- Easy and fast operation: The kits are designed to simplify the experimental process, making the operation more intuitive and user-friendly, thus reducing the complexity and time consumption of experimental operations.
- Continuous innovation: We continue to invest in research and development, constantly introduce new products and improve existing products to adapt to market needs and technological advances.
FAQ
-
Q: Whether it can guide the correct interpretation of test results?
A: Certainly! In our kits, you can obatain detailed analysis protocols to Carry out an experiment. If you still have questions after reading the protocols, contact us at your convenience.
-
Q: What are the precautions for the preservation of test reagents?
A: In the kits, we have tips about the storage conditions of the reagents, cold storage or dark storage, etc., please follow the instructions.
Resources
References
- Ding Q, Zhu H. Upregulation of PSMB8 and cathepsins in the human brains of dementia with Lewy bodies. Neurosci Lett. 2018;678:131-137.
- Müller-Calleja N.; et al. Lipid presentation by the protein C receptor links coagulation with autoimmunity. Science. 2021;371(6534):eabc0956.
- Gu W.; et al. An Aminotransferase Is Responsible for the Deamination of the N-Terminal Leucine and Required for Formation of Oxazolone Ring A in Methanobactin of Methylosinus trichosporium OB3b. Appl Environ Microbiol. 2016;83(1):e02619-16.
- Glal KAM.; et al. Nitazoxanide versus rifaximin in preventing the recurrence of hepatic encephalopathy: A randomized double-blind controlled trial. J Hepatobiliary Pancreat Sci. 2021;28(10):812-824.