Lectins
🧪 Lectin-3931A
Source: Allium sativum
Species: Allium sativum
Tag: Non
Conjugation:
Protein Length:

🧪 Lectin-3988C
Source: Calystega sepiem
Species: Calystega sepiem
Tag: Non
Conjugation:
Protein Length:

🧪 Lectin-4017C
Source: Cicer arietinum
Species: Cicer arietinum
Tag: Non
Conjugation:
Protein Length:

🧪 Lectin-4046D
Source: Dioclea grandiflora
Species: Dioclea grandiflora
Tag: Non
Conjugation:
Protein Length:

🧪 Lectin-4064E
Source: Euonymus europaeus
Species: Euonymus europaeus
Tag: Non
Conjugation:
Protein Length:

🧪 Lectin-4280R
Source: Robinia pseudoacacia
Species: Robinia pseudoacacia
Tag: Non
Conjugation:
Protein Length:

🧪 Lectin-4319S
Source: Succinyl Canavalia ensiformis
Species: Succinyl Canavalia ensiformis
Tag: Non
Conjugation:
Protein Length:

🧪 Lectin-4384V
Source: Vicia graminea
Species: Vicia graminea
Tag: Non
Conjugation:
Protein Length:

Background
Overview
Lectins are carbohydrate-binding proteins that are highly specific for sugar groups that are part of other molecules, thus causing agglutination of particular cells or precipitation of glycoconjugates and polysaccharides.
Lectins have a role in recognition at the cellular and molecular level and play numerous roles in biological recognition phenomena involving cells, carbohydrates, and proteins. Lectins also mediate attachment and binding of bacteria, viruses, and fungi to their intended targets.
The biological properties of lectins are mainly reflected in their interactions with carbohydrates. These proteins are able to recognize and bind to specific sugar chain structures, and this binding is usually non-covalent, relying on non-covalent interactions such as hydrogen bonding, hydrophobic interactions, and van der Waals forces. This binding property of lectins allows them to play a key role in cell recognition, signaling, and cell adhesion.
Categories
- Phytolectins: These lectins are mainly found in plant seeds, such as beans and grains. They are often highly specific, able to recognize and bind to specific monosaccharide or polysaccharide structures.
- Fungal lectins: This class of lectins is widely found in fungi, and they play an important role in the growth and development of fungi.
- Bacterial lectins: Bacterial lectins are involved in bacterial adhesion and biofilm formation, and are related to infection and immune escape mechanisms of pathogens.
- Animal lectins: In animals, lectins are often associated with the immune system, such as selectin and C1q.
Biological Functions
Lectins play a variety of biological roles in nature:
Defense mechanisms: Lectins in plants and animals can act as part of the innate immune system to promote the clearance of pathogens by recognizing specific sugar structures on their surfaces.
Cell signaling: Lectins can affect cell signaling processes such as adhesion, migration and proliferation by interacting with sugar structures on the cell surface.
Nutrient absorption: In some organisms, lectins contribute to the digestion and absorption of sugars in food.
Reproductive process: In plants, lectins may be involved in the interaction between pollen and pistil, affecting the reproductive process of plants.
Cell death: Certain lectins can induce apoptosis, which plays an important role in maintaining tissue homeostasis and immune response.
Inflammatory response: Lectins can be involved in the regulation of inflammatory response by binding to specific cell surface receptors, activating or inhibiting inflammatory signaling pathways.
Cell migration: During development, lectins can guide cells to migrate along specific pathways, which are critical for tissue formation and repair.
Advantages of Lectins
Diversity of action: Lectins can recognize and bind specific sugar molecules, with broad biological activity and specificity, and can be used for research in various fields such as molecular biology, biochemistry, immunology, etc.
Research convenience: Lectins are usually coupled with sugars and are relatively stable when the concentration is low enough. Their structure is also relatively stable, making them easy to extract, separate, and purify in experimental studies.
High safety: Lectins come from nature and are safer compared to other artificially synthesized compounds or drugs, and are easy to synthesize and obtain.
Research Progress
In recent years, the research on lectins has made remarkable progress:
Structure-function relationship: Through techniques such as X-ray crystallography and nuclear magnetic resonance, scientists have resolved the three-dimensional structure of many lectins, which helps to understand their binding specificity and biological function.
Bioengineering: Through genetic engineering and protein engineering, researchers are developing new variants of lectins to improve their specificity and stability and expand their applications in the biomedical field.
Antiagglutinin strategies: Targeting specific sugar structures on the surface of pathogens and tumor cells, scientists are developing inhibitors that can block lectin-mediated signaling as novel therapeutic drugs.

Research Challenges and Future Directions
Although remarkable progress has been made in the study of lectins, several challenges and future directions remain:
Structure-activity relationship: A deeper understanding of the relationship between the structure of lectins and their biological activity can help design more specific and potent lectin derivatives.
Antiagglutinin drug development: Targeting lectin-mediated pathological processes, effective antiagglutinin drugs have been developed for the treatment of related diseases.
The exact role of lectins in disease: Although lectins are known to play a role in a variety of diseases, their exact mechanism in disease development still needs further study.
Biosynthesis and regulation of lectins: Studying the biosynthesis pathway and regulatory mechanism of lectins is helpful to reveal its function and regulatory network in organisms.
Applications
Lectins have a wide range of applications:

Biological studies: Due to their highly specific nature, lectins are often used as research tools to isolate and identify sugar structures on cell surfaces.
Diagnosis: In the medical field, lectins can be used to detect blood disorders, infectious diseases, and tumor markers.
Food industry: Phytolectins are of concern due to their anti-nutritional properties, which may affect the digestion and absorption of food, so their levels need to be controlled in food processing.
Agriculture: In agriculture, lectins can be used as biopesticides to control pests and pathogens.
Drug delivery: Lectins can be used as part of a targeted drug delivery system to achieve targeted drug release by specifically binding to the sugar structure on the surface of tumor cells.
Biosensors: Using the high specificity of lectins, biosensors can be developed to detect pathogens, toxins or biomarkers.
Cosmetics and personal care: In cosmetics and personal care products, lectins can be used as active ingredients for anti-aging, moisturizing and skin repair.
Case Study
Case Study 1: Active Native Allium sativum Lectin Protein
Dietary food components have the ability to affect immune function; following absorption, specifically orally ingested dietary food containing lectins can systemically modulate the immune cells and affect the response to self- and co-administered food antigens. The mannose-binding lectins from garlic (Allium sativum agglutinins; ASAs) were identified as immunodulatory proteins in vitro. The objective of the present study was to assess the immunogenicity and adjuvanticity of garlic agglutinins and to evaluate whether they have adjuvant properties in vivo for a weak antigen ovalbumin (OVA). Garlic lectins (ASA I and ASA II) were administered by intranasal (50 days duration) and intradermal (14 days duration) routes, and the anti-lectin and anti-OVA immune (IgG) responses in the control and test groups of the BALB/c mice were assessed for humoral immunogenicity. The splenic and thymic indices were evaluated as a measure of immunomodulatory functions. Intradermal administration of ASA I and ASA II had showed a four-fold and two-fold increase in anti-lectin IgG response, respectively, vs. the control on day 14. In the intranasal route, the increases were 3-fold and 2.4-fold for ASA I and ASA II, respectively, on day 50. No decrease in the body weights of animals was noticed; the increases in the spleen and thymus weights, as well as their indices, were significant in the lectin groups.
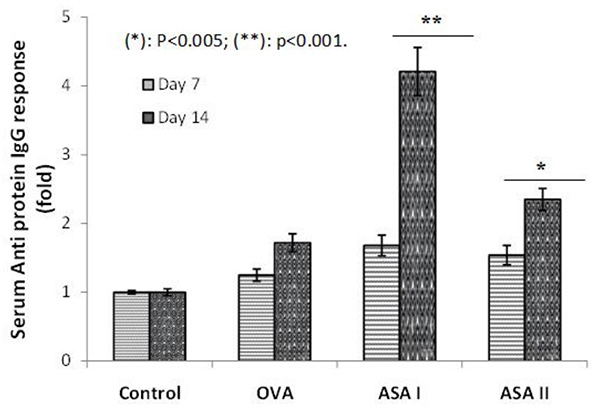
(Shruthishree D Padiyappa, 2022)
Fig1. Serum IgG systemic immune response to ovalbumin (OVA) and garlic lectins (ASA I and II) after intradermal administration in BALB/c mice.
Case Study 2: Active Native Cicer arietinum Lectin Protein
A 35 kDa rabbit erythrocyte agglutinating lectin from the seeds of Cicer arietinum was purified and designated as CAL. The lectin was inhibited by fetuin and N-acetyl-d-galactosamine at a concentration of 20 and 50 mM respectively, but not by simple mono or oligosaccharides. CAL is active between pH 5 and 10 presented thermo stability up to 50 °C and demonstrated DNA damage inhibition at 30 µg concentration. The lectin elicited maximum mitogenic activity towards mice splenocytes at 7.5 µg ml- 1. CAL exerted an inhibitory activity on HIV-1 reverse transcriptase with IC50 of 180 µM. CAL abilities in animal bioassay resulted decreased levels of total triglyceride and creatinine. In vitro and in vivo studies revealed that CAL may constitute an important role impending biomedical applications.
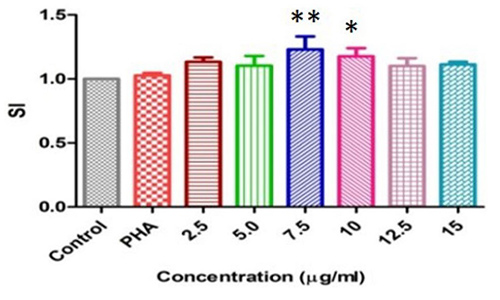
(Ajay Kumar Gautam, 2018)
Fig2. Mitogenic response of mouse splenocytes induced by CAL.
Case Study 3: Active Native Morniga M Lectin Protein
A 35 kDa rabbit erythrocyte agglutinating lectin from the seeds of Cicer arietinum was
Morniga M is a jacalin-related and mannose-specific lectin isolated from the bark of the mulberry (Morus nigra). In order to understand the function and application of this novel lectin, the binding property of Morniga M was studied in detail using an enzyme-linked lectinosorbent assay and lectin-glycan inhibition assay with extended glycan/ligand collection. From the results, it was found that the di-, tri-, and oligomannosyl structural units of N-glycans such as those of the bovine alpha1-acid glycoprotein (gp) and lactoferrin were the most active gps, but not the O-glycans or polysaccharides including mannan from yeast. The binding affinity of Morniga M for ligands can be ranked in decreasing order as follows: gps carrying multiple N-glycans with oligomannosyl residues >> N-glycopeptide with a single trimannosyl core > Tri-Man oligomer [Man alpha1-->6(Man alpha1-->3) Man], Penta-Man oligomer [Man alpha1-->6(Man alpha1-->3)Man alpha1-->6(Man alpha1-->3) Man] > or = Man alpha1-->2, 3 or 6 Man > Man > GlcNAc, Glc >> L-Fuc, Gal, GalNAc (inactive).
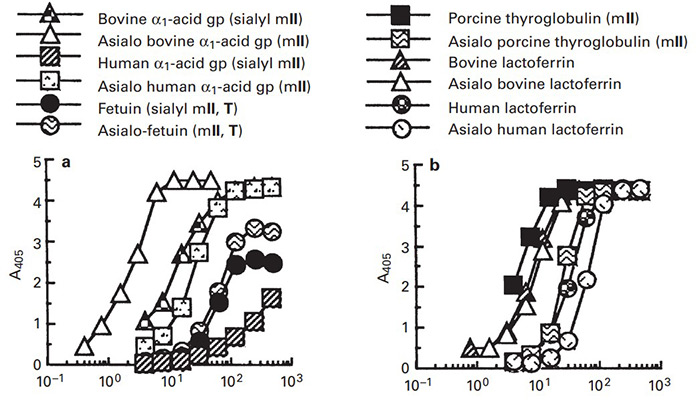
(Albert M Wu, 2004)
Fig3. Binding of Morniga M to microtiter plates coated with various N-linked and O-linked gps and polysaccharides.
Case Study 4: Active Native Aegopodium podagraria Lectin Protein
Lectins from Aegopodium podagraria (APA), Bryonia dioica (BDA), Galanthus nivalis (GNA), Iris hybrid (IRA) and Sambucus nigra (SNAI), and a new lectin-related protein from Sambucus nigra (SNLRP) were studied to ascertain whether they had the properties of ribosome-inactivating proteins (RIP). IRA and SNLRP inhibited protein synthesis by a cell-free system and, at much higher concentrations, by cells and had polynucleotide:adenosine glycosidase activity, thus behaving like non-toxic type 2 (two chain) RIP. APA and SNAI had much less activity, and BDA and GNA did not inhibit protein synthesis.
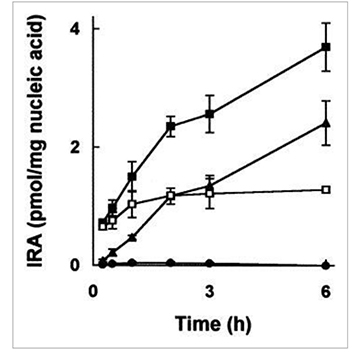
(M G Battelli, 1997)
Fig4. Internalisation of Iris hybrid agglutinin by HeLa cells.
Advantages
- Multiple sources: We offer over 800 types of lectins including bean lectins, adenosine binding lectins, and fungal lectins.
- High quality: We eunsure strict preparation processes to ensure the purity and activity of the products.
- Excellent customer service: We have a professional R & D, after-sales team, with rich experience and expertise, can deeply understand your needs, and provide the appropriate solutions.
- Fast delivery: We will deliver high quality products within the time you expect, with a view to long-term cooperation.
FAQ
-
Q: Can I get professional advice on choosing the right lectin product?
A: Certainly! When it comes to selecting the appropriate lectin product for your needs, there are several factors to consider. Please consult us for details.
-
Q: What are the best conditions for preservation of lectins?
A: Check the recommended storage conditions for the lectin product to ensure it maintains stability and activity over time. Some lectins may require refrigeration or freezing.
Resources
References
- Gautam AK.; et al. Biochemical and functional properties of a lectin purified from the seeds of Cicer arietinum L. 3 Biotech. 2018;8(6):272.
- Battelli MG.; et al. Ribosome-inactivating lectins with polynucleotide:adenosine glycosidase activity. FEBS Lett. 1997;408(3):355-359.