Cell-Free Protein Analysis Services
Cell-free protein analysis is a powerful in vitro platform designed to replicate and study biological reactions outside of living cells. By eliminating the complexities and limitations of cellular environments, it allows for precise, efficient, and scalable protein analytical solutions. At Creative BioMart , we leverage our extensive expertise in protein science and enzymology to develop tailored cell-free biosystems. These systems not only provide exceptional flexibility and accuracy for protein studies but also serve as a cost-effective platform for industrial biomanufacturing and advanced protein characterization.
Introduction to Cell-Free Protein Analysis

Cell-free systems are engineered in vitro environments where biological reactions occur without the interference of cell membranes or cellular metabolism. They employ purified enzymes, coenzymes, and optimized reaction conditions to mimic intracellular processes in a controlled and accessible manner.
Such systems have become invaluable in protein analysis and synthetic biology, offering:
- Simplified Control: Free from membrane barriers and cellular regulation.
- High Yield Potential: Producing significant amounts of target proteins without wasteful by-products.
- Novel Reaction Capabilities: Enabling processes that living organisms or chemical catalysts cannot achieve.
- Robust Performance: Enzyme cocktails that withstand toxic compounds and harsh reaction conditions (e.g., high temperatures, acidic pH, organic solvents, ionic liquids).
At Creative BioMart, we have established advanced expertise in harnessing these advantages for industrial and research applications.
Cell-Free Protein Analysis Services & Capabilities
Creative BioMart provides a full suite of Cell-Free Protein Analysis Services, including but not limited to:
- Development and optimization of customized cell-free biosystems.
- Enzymatic protein synthesis and characterization.
- Analytical testing of protein stability, folding, and activity in cell-free environments.
- Cell-free platforms for ubiquitin-independent proteasomal protein degradation studies.
- Scalable biomanufacturing solutions with low-cost, high-yield advantages.
- Tailored consulting and project support for specialized research needs.
Service Workflow

Advantages of Partnering with Us for Cell-Free Protein Studies
- Proven Expertise: Decades of experience in protein science and cell-free systems.
- Custom Solutions: Tailor-made workflows to meet specific research or industrial requirements.
- High Efficiency: Faster reaction rates compared to microbial systems, ensuring time savings.
- Robust Performance: Systems designed to tolerate toxic compounds and extreme conditions.
- Cost-Effective Platforms: Low-cost alternatives to traditional biomanufacturing with superior yields.
- Comprehensive Support: From initial consultation to final data interpretation, we provide end-to-end service.
Case Studies: Applications of Cell-Free Protein Analysis
Case 1: Cell-free multi-enzyme catalysis for sustainable fumarate production
Hou et al., 2023. doi:10.1016/j.synbio.2023.03.004
Fumarate is an important value-added chemical with applications across food, medicine, materials, and agriculture. To address increasing demand and the need for sustainable production, researchers designed a novel in vitro cell-free multi-enzyme catalytic pathway for fumarate synthesis. Using three enzymes—acetyl-CoA synthase, malate synthase, and fumarase from E. coli —the system efficiently converted low-cost substrates acetate and glyoxylate into fumarate. The process incorporated recyclable coenzyme A, and optimization of enzymatic conditions achieved a 0.34 mM fumarate yield with a 34% conversion rate after 20 hours. This study demonstrates a promising alternative to traditional petrochemical-based fumarate production.
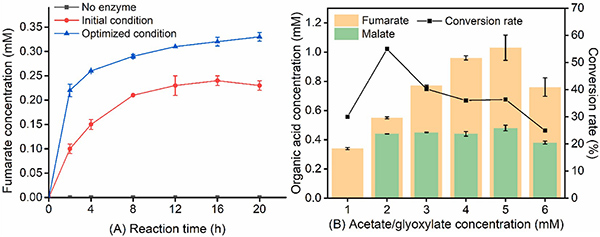
Figure 1. Fumarate production under optimized conditions. (A), Comparison of initial and optimized conditions for fumarate production. (B), Effect of substrate concentration on the conversion rate. (Hou et al., 2023)
Case 2: Affinity microfluidics enables high-throughput protein degradation analysis in cell-free extracts
Brio et al., 2022. doi:10.1038/s42003-022-04103-3
Protein degradation via the ubiquitin-proteasome pathway is central to cellular regulation, but traditional in vitro degradation assays are time-consuming, low-throughput, and labor-intensive. To overcome these limitations, protein degradation on chip (pDOC), a MITOMI-based microfluidic technology, was developed for high-throughput analysis in cell-free extracts. pDOC enables hundreds of microchambers to simultaneously assay protein degradation using minimal reagents under diverse physiochemical conditions. This multiplex platform is sensitive, rapid, and scalable, offering a powerful alternative to conventional gel-based methods. With its efficiency and precision, pDOC holds significant potential for biomedical and translational research focused on regulated proteolysis.
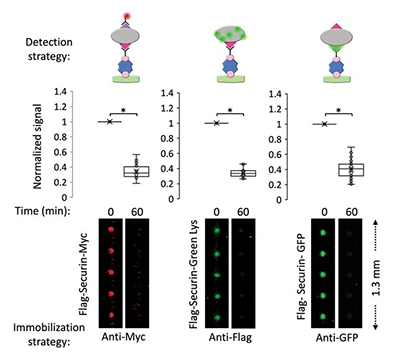
Figure 2. Degradation of Flag-Securin-Myc, Flag-Securin-GFP, and Green-Lys-labeled Flag-Securin (IVT products) in NDB mitotic extracts was assayed in tube for 1 h. On-chip analysis was performed in multiple ways: 1) Flag-Securin-Myc was immobilized by anti-Myc antibodies and detected by anti-Flag Cy5-conjugated antibodies; 2) Flag-Securin-GFP was immobilized and detected via the GFP tag; and 3) Green-Lys-labeled Flag-Securin was immobilized via anti-Flag antibodies and detected by the Green-Lys signal. Anti-Myc/Flag/GFP antibodies are biotinylated. (Brio et al., 2022)
Researcher Feedback on Our Protein Analysis Services
“We engaged Creative BioMart to explore ubiquitin-independent proteasomal degradation for one of our oncology targets. Their cell-free platform provided clear, reproducible data that helped us understand the degradation mechanism within weeks, not months. The insights we gained significantly accelerated our preclinical decision-making, and their responsive team ensured smooth communication throughout the project.”
— R&D Director | Mid-Sized Pharmaceutical Company
“Our fermentation-based systems could not withstand the acidic pH and high temperatures required for our biocatalysis project. Creative BioMart developed a customized cell-free enzymatic system that performed consistently under these extreme conditions. The resulting high-yield protein synthesis opened new pathways for scale-up, saving us both time and production costs.”
— Senior Scientist | Industrial Biotech Firm
“As part of a synthetic biology grant, we needed rapid screening of enzyme variants in a non-cellular environment. Creative BioMart’s cell-free protein analysis allowed us to test dozens of variants without the delays of cell culture. Their detailed reports and technical support gave our team confidence to present robust data to funding agencies.”
— Principal Investigator | Academic Research Institute
“Before committing to large-scale microbial fermentation, we wanted a low-cost method to assess protein yield potential. Creative BioMart’s cell-free biosystem delivered high-quality data that allowed us to make an informed investment decision. Their efficiency, transparency, and technical depth made them an invaluable partner in our feasibility study.”
— VP of Process Development | Global Biomanufacturing Company
FAQs About Cell-Free Protein Analysis Services
-
Q: What are the main advantages of using cell-free protein analysis over traditional cell-based systems?
A: Cell-free systems offer precise control over reaction conditions, faster reaction rates, and higher yields without generating unwanted by-products or cell mass. They also tolerate harsh environments (e.g., high temperature, low pH, organic solvents) and toxic compounds that microbial systems cannot withstand, making them ideal for specialized protein studies and industrial applications. -
Q: Can you handle challenging protein projects, such as degradation studies or work under extreme conditions?
A: Yes. Creative BioMart has extensive expertise in developing customized enzyme cocktails that can operate under extreme conditions. We also provide specialized services for ubiquitin-independent proteasomal protein degradation, delivering reliable insights where traditional systems often fail. -
Q: How scalable are your cell-free biosystems for industrial applications?
A: Our cell-free platforms are designed with scalability in mind. They can be used for early-stage feasibility studies, pilot-scale protein synthesis, and even as cost-effective alternatives to microbial fermentation for large-scale protein production. -
Q: What does your typical service workflow look like?
A: Our workflow includes consultation to understand your goals, tailored system design, construction of customized cell-free biosystems, comprehensive protein analysis, delivery of validated reports, and ongoing support. This ensures both scientific rigor and flexibility throughout your project. -
Q: How do you ensure data reliability and reproducibility?
A: We use highly purified enzymes and optimized coenzyme systems to minimize variability. Our team follows stringent quality control measures, providing reproducible results backed by detailed datasets and clear interpretation. -
Q: Why should I choose you for cell-free protein analysis?
A: We combine decades of protein science expertise with state-of-the-art cell-free technology, offering tailored solutions, faster turnaround times, cost-effective platforms, robust performance under diverse conditions, and end-to-end project support.
Resources
Related Services
- Protein Analytical Service
- Biopharmaceutical Solution
- Protein Characterization
- Cell-free Display Platform
- Cell-Free In Vitro Protein Expression
Related Products
References:
- Brio L, Wasserman D, Michaely-Barbiro E, Barazany-Gal G, Gerber D, Tzur A. Affinity microfluidics enables high-throughput protein degradation analysis in cell-free extracts. Commun Biol. 2022;5(1):1147. doi:10.1038/s42003-022-04103-3
- Hou C, Tian L, Lian G, Fan LH, Li ZJ. Conversion of acetate and glyoxylate to fumarate by a cell-free synthetic enzymatic biosystem. Synthetic and Systems Biotechnology. 2023;8(2):235-241. doi:10.1016/j.synbio.2023.03.004
Contact us or send an email at for project quotations and more detailed information.
Quick Links
-

Papers’ PMID to Obtain Coupon
Submit Now -

Refer Friends & New Lab Start-up Promotions

