Western blot protocol
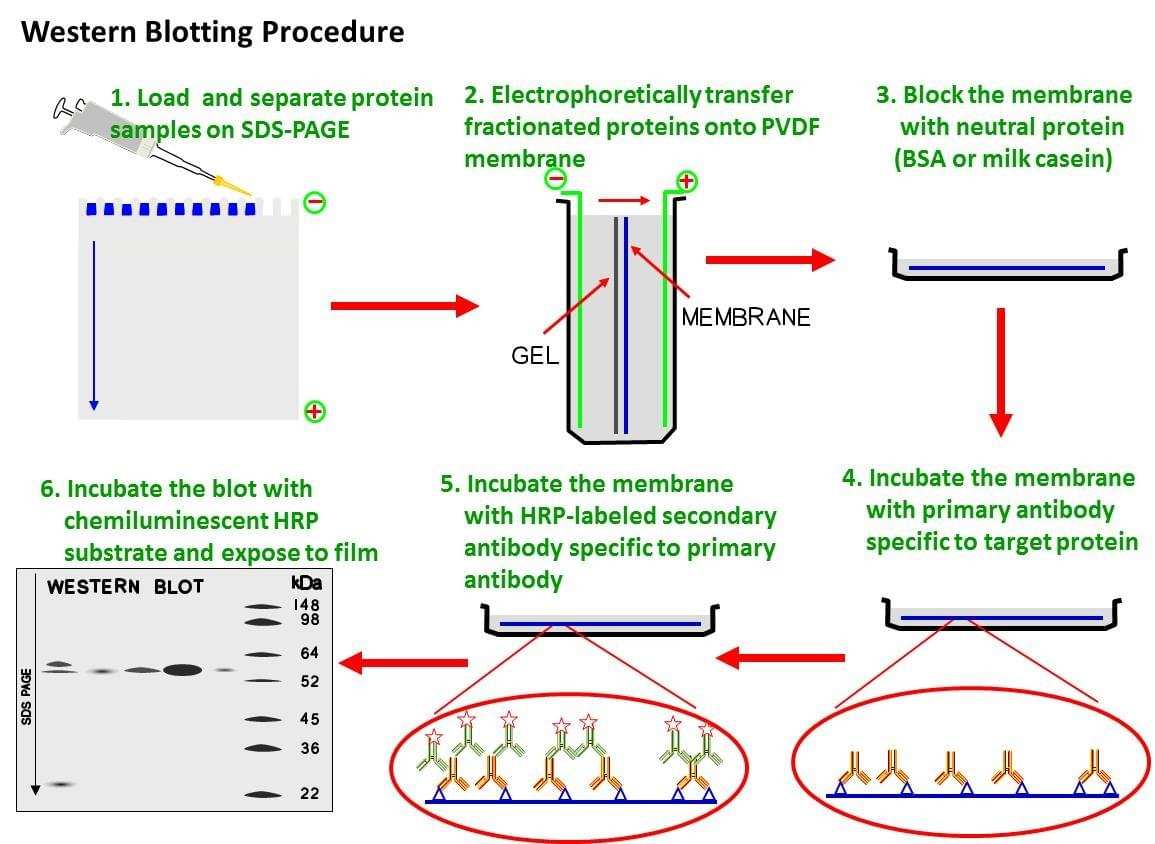 Fig. The schematic diagram of western bolt protocol.
Fig. The schematic diagram of western bolt protocol.
Western blot protocol procedure
Sample prepare
Usually, samples are resuspended or mixed in 20-50 µl of 1X sample buffer and all the mixtures are incubated at 90°C for five minutes or 50°C for 15 minutes to break down the interactions between peptides.
Protein Concentration Determination
The Bradford assay method (Bio-Rad Protein Assay) is utilized to determine the concentration of proteins in each sample. The reading obtained is compared to a standard curve that allowed for a relative measurement of protein concentration. The standard curve is produced by using varying concentrations of bovine serum albumin (BSA).
SDS-PAGE
SDS-PAGE is performed using the Mini-PROTEAN III electrophoresis system (Bio-Rad).
- Samples are loaded and fractionated on 8, 10, or 12% polyacrylamide gels along with a protein ladder.
- Gels are electrophoresed between 100 and 150 V in running buffer (3% Tris-HCl, 14.4% glycine, 1% SDS) until the bromophenol blue dye reaches the bottom of the gel.
- Gels are either electrotransferred to PVDF membranes.
Western Blot Analysis
- The Mini Trans-Blot Electrophoretic Transfer Cell system (Bio-Rad) is used for western blot experiments.
- Proteins fractionated by SDS-PAGE are electrotransferred onto polyvinylidene difluoride (PVDF) membranes. The transfer is carried out in transfer buffer (192 mM glycine, 20% v/v methanol, 25 mM Tris-HCl, pH 8.3). The protein transfer is carried out at 100 V for 1 hour at 4°C with buffer recirculation. All of the remaining steps are carried out at room temperature.
- After transfer, the membranes are blocked with a BSA blocking solution (5% BSA, 150 mM NaCl, 0.05% Tween 20, 20 mM Tris-HCl, pH 7.5) or a milk blocking solution (5% powdered milk, 150 mM NaCl, 0.05% Tween 20, 20 mM Tris-HCl, pH 7.5).
- The membranes are incubated in appropriate dilutions of primary antibody in TBST (150 mM NaCl, 0.05% Tween 20, 20 mM Tris-HCl, pH 7.5) for 1 hour at room temperature or overnight at 4°C, with carefully shaking. The primary antibody is used following the manufacturer’s advice.
- The membranes are washed 3 times with TBST for 10 minutes remove free primary antibody.
- For colorimetric detection, the secondary antibody alkaline, phosphataseconjugated α-rabbit IgG, is incubated with the membrane at appropriate dilution in TBST for 1 hour at room temperature.
- The membrane is washed with TBST as before to remove any free secondary antibody.
- An additional 30 second wash with TBS is carried out.
- The antibodies are visualized by adding the color development substrates, Nitro blue tetrazolium (NBT) and 5-bromo-4-chloro-3-indolylphosphate (BCIP), in alkaline phosphatase buffer (100 mM NaCl, 5 mM MgCl2, 100 mM Tris-HCl, pH 9.5). The reactions are terminated by washing the membranes in water.
Chemiluminescence detection
- For chemiluminescence detection, the secondary antibody, horseradish peroxidase-conjugated α-mouse IgG, is incubated with the membrane at a 1:10,000 dilution in TBST for 1 hour.
- The membrane is washed with TBST as before to remove any free secondary antibody.
- An additional 30 second wash with TBS is carried out.
- The antibodies are visualized by mixing the chemiluminescence development substrates, Peroxide Buffer and Luminol/Enhancer Solution (Pierce), in a 1:1 ratio and incubating this mixture on the membrane for 5 minutes.
- The membrane is immediately scanned using an imager following manufacturer’s instructions.

