IVD of Olea europaea Allergy
🧪 Olee 7-3730C
Source: E.coli
Species: Olive
Tag: His&KSI
Conjugation:
Protein Length: 1-21aa

Olea europaea
In the Mediterranean region, where olive trees are widely planted, olive hay fever has become an important health problem. Olive pollen, an immunoglobulin E (IgE)-mediated allergen, is one of the most important causes of respiratory allergies due to the large amounts of pollen released by olive trees into the atmosphere. Currently, in vitro diagnostics (IVD) identifies the most clinically relevant sensitizing allergen for olive pollen allergy, Ole e 1, by detecting specific IgE antibodies in patients using specific reagents or serological methods.

Main Steps of IVD for Olea europaea Allergy
- Immunological Test
The main methods are ELISA, immunoblotting, radio allergen absorption test (RAST), etc. Detect IgE antibodies or cytokines in serum, focusing on total IgE determination and specific IgE determination. The amount of specific IgE antibodies can help identify allergens.
- Skin Prick Test
In order to determine whether the patient is allergic to olive pollen, drop an olive pollen extract on his/her skin, then gently prick it with a needle and observe the skin's reaction.
Creative BioMart provides high-quality recombinant Olea europaea allergen proteins used for IVD, including ELISA, lateral flow assay, western blot, and other immunoassays.
Highlights of Our Products
- High specificity and sensitivity. Our products exhibit greater specificity since they are meticulously designed and created to target known, specific allergen proteins.
- High purity, no contamination with non-specific allergens.
- Easy to scale up production. A large number of specific proteins can be obtained through process amplification, without being limited by the content of natural extracts and materials.
- Higher safety and repeatability.
Our Outstanding Advantages
- A comprehensive range of IVD products to meet the different needs of customers and provide customers with comprehensive scientific research support.
- We can provide first-class services through a skilled technical team combined with advanced scientific research equipment and technology.
- Pay attention to service quality, provide customers with timely and accurate IVD-related services, and ensure that customers can get the best scientific research experience.
Clinical Related Information
Major Allergen:
The major allergen of Olea europaea is ole e 1, a protein found in olive tree pollen.
Olive oil, on the other hand, is made from the fruit of the olive tree and usually does not contain significant amounts of pollen. Therefore, most people who are allergic to olive tree pollen can consume olive oil without experiencing allergic reactions.
Cause and Symptoms:
Cause: The allergy is caused by inhalation of pollen from the olive tree, which is highly allergenic. The pollen is prevalent in areas where olive trees are abundant, especially during the flowering season.
Symptoms:
- Sneezing
- Runny or stuffy nose
- Itchy or watery eyes
- Coughing
- Wheezing
- Shortness of breath
- Asthma attacks in severe cases
- Skin rashes (less common)
Case Study
Case 1: Somoza ML, Pérez-Sánchez N, Torres-Rojas I, et al. Sensitisation to Pollen Allergens in Children and Adolescents of Different Ancestry Born and Living in the Same Area. J Asthma Allergy. 2022 Sep 26;15:1359-1367. doi: 10.2147/JAA.S370279. PMID: 36189188; PMCID: PMC9525024.
The study aimed to describe the pattern of sensitisation and allergy in children and adolescents of Spanish versus Moroccan ancestry but born in the same rural area of Spain. A detailed questionnaire was completed, and skin prick tests were performed to assess reactions to the most prevalent pollen allergens (O. europaea, P. pratense, S. kali, C. arizonica, P. acerifolia, A. vulgaris and P. judaica) plus molecular components Ole e 1 and Ole e 7.
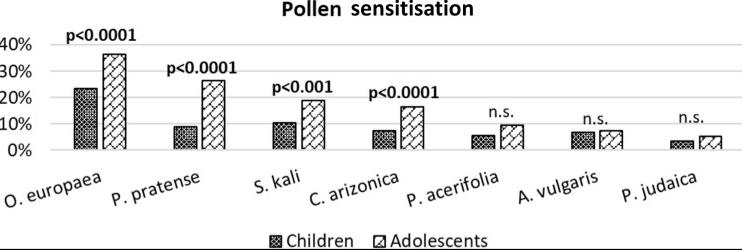 Fig2. Pollen sensitisation in children vs adolescents.
Fig2. Pollen sensitisation in children vs adolescents.Case 2: Manzanares B, González R, Serrano P, Navas A, Alonso C, Fernandez L, Jurado A, Moreno-Aguilar C. Back to basics: likelihood ratios for olive and grass pollen specific IgE in seasonal allergic rhinitis. Front Allergy. 2023 Oct 4;4:1241650. doi: 10.3389/falgy.2023.1241650. PMID: 37859976; PMCID: PMC10582635.
Specific IgE (sIgE) is merely a sensitization marker that cannot be used for allergy diagnosis if there are no associated clinical symptoms. This study aimed to calculate cut-offs for sIgE, allowing us to effectively diagnose olive or grass pollen allergy and select allergenic immunotherapy (AIT) candidate patients in a region under high olive and grass allergenic pressure.
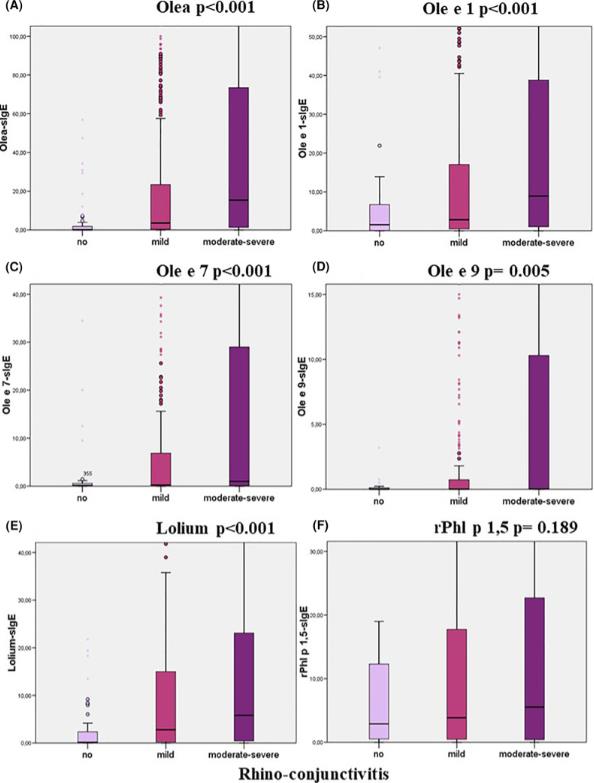 Fig3. Relationship between specific IgE levels and the degree of rhino-conjunctivitis categorised according to the ARIA guidelines (Kruskal–Wallis test).
Fig3. Relationship between specific IgE levels and the degree of rhino-conjunctivitis categorised according to the ARIA guidelines (Kruskal–Wallis test).




