IVD of Chikungunya
🧪 CP-01C
Source: E.coli
Species: Human
Tag: His
Conjugation:
Protein Length: 160 amino acids

🧪 CHIKVgp2-25C
Source: Insect Cells
Species: Chikungunya
Tag: Non
Conjugation:
Protein Length: amino acids 1-415

🧪 CHIKVgp2-27C
Source: Insect Cells
Species: Chikungunya
Tag: Non
Conjugation:
Protein Length: amino acids 1-415

🧪 CHIKVE1-823C
Source: Insect Cells
Species: Chikungunya virus
Tag: Non
Conjugation:
Protein Length:

🧪 CHIKVE1-727C
Source: Insect Cells
Species: Chikungunya
Tag: Non
Conjugation:
Protein Length:

🧪 E2-336C
Source: Insect Cells
Species: CHIKV
Tag: His
Conjugation:
Protein Length: Ser326-Gln666

🧪 nsp4-4619C
Source: E.coli
Species: CHIKV
Tag: His
Conjugation:
Protein Length: 2228-2474aa

🧪 CHIKV-2767P
Source: HEK293
Species: Pan-species
Tag: His
Conjugation:
Protein Length: Tyr810-His1248

🧪 CHIKV-2754P
Source: E.coli
Species: Pan-species
Tag: His
Conjugation:
Protein Length: Tyr810-Ile1222

🧪 E2-2427C
Source: Insect Cells
Species: CHIKV
Tag: His
Conjugation:
Protein Length: Ser326-Gln666

Chikungunya Fever
Chikungunya fever is an arboviral disease caused by chikungunya virus (CHIKV) and transmitted by Aedes mosquitoes. It is common in tropical and subtropical regions. Symptoms of chikungunya mainly include fever and headache, often accompanied by joint pain and lower back pain. In addition, CHIKV may cause meningoencephalitis and other central nervous system diseases in newborns. Currently, laboratories can perform in vitro diagnostics (IVD) of CHIKV infection by detecting virus-specific IgM/IgG antibodies and viral RNA.
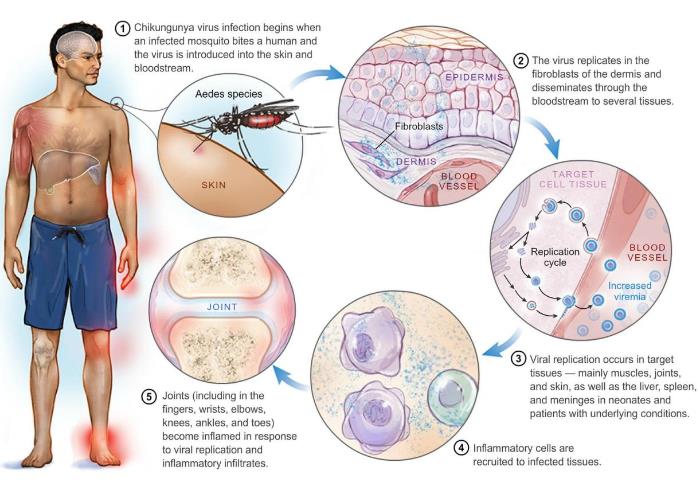 Figure 1. Successive steps of chikungunya virus infection in humans, based on human clinical data and animal experiments. (Couderc T, et al., 2015)
Figure 1. Successive steps of chikungunya virus infection in humans, based on human clinical data and animal experiments. (Couderc T, et al., 2015)Main Steps of IVD for Chikungunya
- Antigen and antibody testing. Serum samples from patients in the acute phase of the disease were collected and tested using ELISA, immunochromatography, and immunofluorescence antibody assay (IFA).
- Nucleic acid amplification testing. Blood samples from patients can be collected and RT-PCR and other methods can be used to detect the nucleic acid of the virus to diagnose CHIKV infection.
- Virus isolation and culture. Samples such as serum, blood cells, or saliva are collected from patients in the acute stage of illness, inoculated into sensitive cells, and the virus is isolated and cultured.
Creative BioMart provides high-quality recombinant Chikungunya protein used for IVD, including ELISA, lateral flow assays, western blots, and other immunoassays.
Highlights of Our Products
- High sensitivity, high specificity, and high purity.
- It is widely used and suitable for downstream immunological experiments.
- Easy to store and transport, conducive to large-scale production and use of vaccines.
- Outstanding success rate and fast development speed.
Our Outstanding Advantages
- Viral proteins are continuously improved and come in a wide variety to meet different research and development needs.
- With a professional R&D team and advanced laboratory facilities, we can conduct a variety of production research and development.
- A complete IVD protein platform can provide customized services to meet different scientific research needs.
- High-quality service, high-level experiments, and reliable analysis.
- IVD proteins can be used to test for a variety of diseases and conditions, making them valuable tools for diagnosing and monitoring health.
Applications
Vaccine Development: CHIKV proteins, particularly the envelope proteins (E1 and E2), are crucial targets for vaccine development. These proteins are used to develop subunit vaccines, virus-like particle (VLP) vaccines, and mRNA vaccines that aim to elicit an immune response without causing disease.
Diagnostic Tools: CHIKV proteins are used in diagnostic assays to detect the presence of antibodies against the virus in patient serum. Enzyme-linked immunosorbent assays (ELISAs) and rapid diagnostic tests (RDTs) often utilize recombinant CHIKV proteins to improve the accuracy and specificity of chikungunya diagnostics.
Antiviral Drug Development: Understanding the structure and function of CHIKV proteins, especially the non-structural proteins (nsP1 to nsP4), can aid in the development of antiviral drugs. These proteins play key roles in viral replication and transcription, making them prime targets for small molecule inhibitors.
Monoclonal Antibody Production: CHIKV proteins are also used to generate monoclonal antibodies, which can be used both for therapeutic purposes and as research tools. These antibodies can neutralize the virus, study its biology, and even help in passive immunotherapy.
Structural Biology Studies: The structural analysis of CHIKV proteins using techniques like X-ray crystallography and cryo-electron microscopy (cryo-EM) provides insights into their three-dimensional structures. This information is crucial for understanding virus assembly, entry, and replication mechanisms.
Molecular Biology Research: CHIKV proteins are used as tools in molecular biology to understand the virus-host interactions, viral life cycle, and immune evasion strategies. These studies can provide broader insights into viral pathogenesis and host immune responses.
Gene Therapy Vectors: Modified versions of chikungunya virus proteins can potentially be used in gene therapy as delivery vectors due to their ability to package and deliver genetic material into host cells.
Autoimmune and Inflammatory Disease Research: Since CHIKV infection can lead to chronic joint inflammation similar to autoimmune diseases, studying the virus and its proteins can provide insights into the mechanisms of inflammatory diseases, potentially leading to novel therapeutic approaches.
Case Study
Case 1: Alvarez-García L, Sánchez-García FJ, Vázquez-Pichardo M, Moreno-Altamirano MM. Chikungunya Virus, Metabolism, and Circadian Rhythmicity Interplay in Phagocytic Cells. Metabolites. 2023 Nov 11;13(11):1143. doi: 10.3390/metabo13111143. PMID: 37999239; PMCID: PMC10672914.
Chikungunya virus (CHIKV) is transmitted to humans by mosquitoes of the genus Aedes, causing the chikungunya fever disease, associated with inflammation and severe articular incapacitating pain. in host-pathogen interactions, CHIKV might have the ability to alter the function of immune cells, partly by rewiring the tricarboxylic acid cycle. Some viral evasion mechanisms depend on the metabolic reprogramming of immune cells, and the cell metabolism is intertwined with circadian rhythmicity; thus, a circadian immunovirometabolism axis may influence viral pathogenicity.
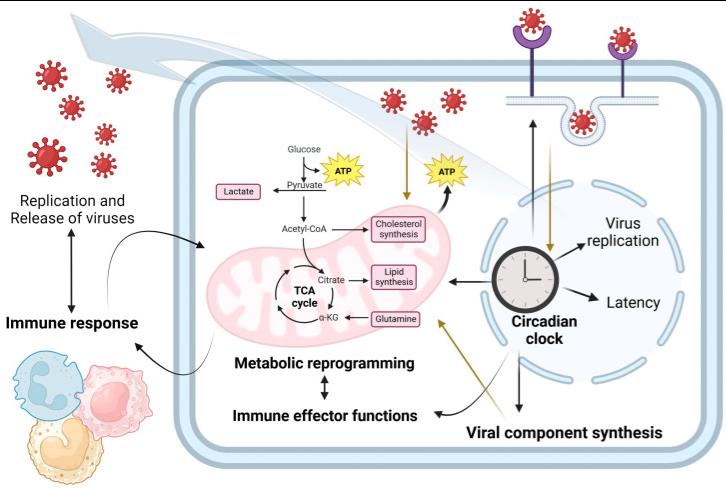 Fig2. Circadian immunovirometabolism: connections between the cell clock, metabolic reprogramming, and the immune response in the context of viral infection. The cell clock has an important role in viral replication, at multiple stages of the viral replication cycle including the regulation of entry receptors and the anabolic pathways required for viral particle synthesis. In addition, infection can disrupt circadian rhythmicity and viruses might induce metabolic changes in infected cells to favor their replication while avoiding immune antiviral responses.
Fig2. Circadian immunovirometabolism: connections between the cell clock, metabolic reprogramming, and the immune response in the context of viral infection. The cell clock has an important role in viral replication, at multiple stages of the viral replication cycle including the regulation of entry receptors and the anabolic pathways required for viral particle synthesis. In addition, infection can disrupt circadian rhythmicity and viruses might induce metabolic changes in infected cells to favor their replication while avoiding immune antiviral responses.Case 2: Yin P, Davenport BJ, Wan JJ, Kim AS, Diamond MS, Ware BC, Tong K, Couderc T, Lecuit M, Lai JR, Morrison TE, Kielian M. Chikungunya virus cell-to-cell transmission is mediated by intercellular extensions in vitro and in vivo. Nat Microbiol. 2023 Sep;8(9):1653-1667. doi: 10.1038/s41564-023-01449-0. Epub 2023 Aug 17. PMID: 37591996; PMCID: PMC10956380.
The mechanistic role of these intercellular extensions in CHIKV infection was unclear. Here the authors developed a co-culture system and flow cytometry methods to quantitatively evaluate transmission of CHIKV from infected to uninfected cells in the presence of neutralizing antibody.
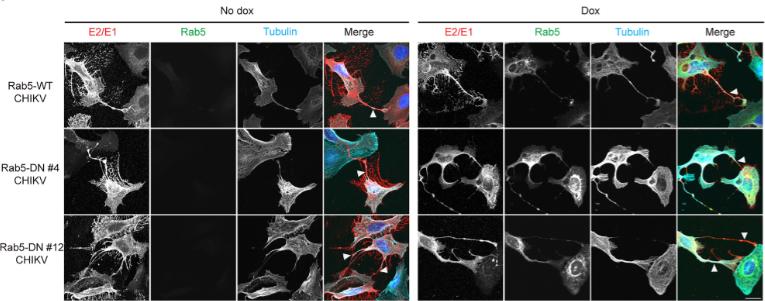 Fig3. Characterization of inducible Rab5 U-2 OS cell lines.
Fig3. Characterization of inducible Rab5 U-2 OS cell lines.Case 3: Law MCY, Zhang K, Tan YB, Nguyen TM, Luo D. Chikungunya virus nonstructural protein 1 is a versatile RNA capping and decapping enzyme. J Biol Chem. 2023 Dec;299(12):105415. doi: 10.1016/j.jbc.2023.105415. Epub 2023 Oct 31. PMID: 37918803; PMCID: PMC10687048.
Chikungunya virus (CHIKV) nonstructural protein 1 (nsP1) contains both the N7-guanine methyltransferase and guanylyltransferase activities and catalyzes the 5' end cap formation of viral RNAs. To further understand its catalytic activity and role in virus-host interaction, researchers demonstrate that purified recombinant CHIKV nsP1 can reverse the guanylyl transfer reaction and remove the m7GMP from a variety of capped RNA substrates including host mRNAs.
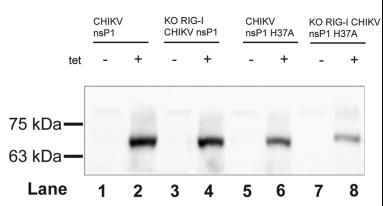 Fig4. CHIKV nsP1 enzymatic activity activates interferon mediated by RIG-I in tet-inducible expression of CHIKV nsP1 and mutant in A549 Dual and RIG-I KO cells. Western blot detection of CHIKV nsP1 in tet-inducible A549 Dual and RIG-I KO cells with and without tetracycline (1 μg/ml) at 72 h. CHIKV, chikungunya virus; nsP, nonstructural protein; RIG-I, retinoic acid–inducible gene I.
Fig4. CHIKV nsP1 enzymatic activity activates interferon mediated by RIG-I in tet-inducible expression of CHIKV nsP1 and mutant in A549 Dual and RIG-I KO cells. Western blot detection of CHIKV nsP1 in tet-inducible A549 Dual and RIG-I KO cells with and without tetracycline (1 μg/ml) at 72 h. CHIKV, chikungunya virus; nsP, nonstructural protein; RIG-I, retinoic acid–inducible gene I.Reference
- Couderc T, Lecuit M. (2015). Chikungunya virus pathogenesis: From bedside to bench[J]. Antiviral research. 121: 120-131.






