Eph Receptor
Related Symbol Search List
Immunology Background
Available Resources for Eph Receptor Research
Creative BioMart is your ultimate one-stop shop for all your research needs related to Eph receptors. Our extensive range of carefully developed products and customized services are designed to help you delve deep into the world of Eph receptors and their vital role in various physiological processes.
- Our product line includes top-quality recombinant proteins, protein pre-coupled magnetic beads, cell and tissue lysates, and others.
- We also offer a wealth of resources on Eph receptor molecules, covering essential topics such as involved pathways, protein functions, interacting proteins, related articles, and research areas.
Our Featured Products
| Cat.# | Product name | Species | Source (Host) | Tag |
|---|---|---|---|---|
| EPHA1-3374H | Active Recombinant Human EPHA1 Protein, GST-tagged | Human | Insect Cell | GST |
| EPHA1-5931C | Recombinant Chicken EPHA1 | Chicken | Mammalian Cell | His |
About Eph Receptors
Eph receptors are a family of receptor tyrosine kinases that play important roles in various physiological and developmental processes. They are named after the Ephrin ligands, which are their primary binding partners. The Eph receptor family consists of 14 members (EphA1-EphA8 and EphB1-EphB4), which are divided into two subclasses based on their affinity for specific Ephrin ligands.
Structure of Eph Receptors
Eph receptors are transmembrane proteins that consist of an extracellular domain, a single transmembrane domain, and an intracellular domain with kinase activity. The extracellular domain contains a ligand-binding region, while the cytoplasmic domain contains a tyrosine kinase domain that becomes activated upon ligand binding.
Ephrin Ligands
Eph receptors bind to cell surface ligands called Ephrins. Ephrins are also divided into two subclasses: Ephrin-A and Ephrin-B, which correspond to the subclasses of Eph receptors. Ephrin-A ligands preferentially bind to EphA receptors, while Ephrin-B ligands bind to EphB receptors. Ephrins can exist in both membrane-bound forms (attached to the cell membrane) and soluble forms (released into the extracellular space).
Bidirectional Signaling
The interaction between Eph receptors and Ephrin ligands leads to bidirectional signaling. This means that both the receptor-expressing cell (Eph receptor-bearing cell) and the ligand-expressing cell (Ephrin-bearing cell) can transmit signals upon binding. This bidirectional signaling is critical for the regulation of cell-cell communication and tissue patterning during development.
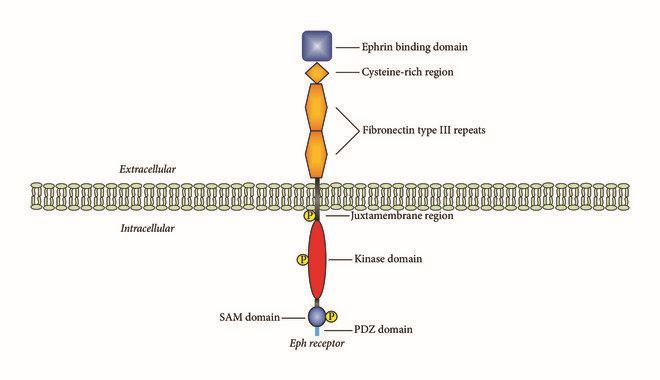 Fig.1 Domains in Eph receptors. (Kou CJ, et al., 2018)
Fig.1 Domains in Eph receptors. (Kou CJ, et al., 2018)
The cytoplasmic and extracellular portions of the receptor are separated by the membrane bilayer. The extracellular region of Eph receptors contains a ligand binding domain, a cysteine-rich domain, and two fibronectin type III repeats. The intracellular region is composed of a tyrosine domain, a sterile α motif (SAM), and a PDZ domain. The domains have been drawn in different shapes and colors, and individual domains are labeled with their designations. Phosphorylated residues are indicated.
Eph-Ephrin Signaling Pathways
Upon binding of Eph receptors to Ephrin ligands, several intracellular signaling pathways are activated. The exact signaling mechanisms can vary depending on the specific Eph receptor and Ephrin involved. However, some common signaling pathways include:
- Tyrosine Phosphorylation: Ligand binding induces autophosphorylation of specific tyrosine residues in the intracellular domain of the receptor. This leads to the activation of the receptor's tyrosine kinase activity and subsequent phosphorylation of downstream signaling molecules.
- Rho GTPase Signaling: Eph receptors can activate Rho GTPases, such as RhoA, Rac1, and Cdc42. These small GTPases regulate cytoskeletal dynamics, cell adhesion, and cell migration, thereby impacting processes like axon guidance, tissue boundary formation, and cell sorting.
- MAPK/ERK Pathway: Eph receptors can activate the MAPK/ERK signaling pathway, which plays a role in cell proliferation, survival, and differentiation. Activation of this pathway contributes to various developmental processes, including neuronal development and tissue morphogenesis.
- Phosphoinositide 3-kinase (PI3K) Pathway: Eph receptors can activate PI3K, leading to the activation of downstream effectors such as Akt. The PI3K pathway regulates cell survival, growth, and motility.
Functions of Eph Receptors
Recent research in the field of Eph receptors has made significant progress in understanding their roles in various aspects of development, tissue homeostasis, and disease pathogenesis. Here are some notable advances and emerging insights:
- Cancer: Eph receptors and their ligands have been implicated in cancer progression and metastasis. Recent studies have identified dysregulated Eph receptor signaling in multiple cancer types. For example, EphA2 and EphB4 receptors have been associated with tumor growth, invasion, and angiogenesis in breast, colorectal, and lung cancers. Targeting Eph receptors and their downstream signaling pathways is being explored as a potential therapeutic strategy in cancer treatment.
- Neurological Disorders: Eph receptors play crucial roles in neuronal development, synaptic plasticity, and axon guidance. Dysfunction of Eph receptor signaling has been implicated in neurological disorders such as Alzheimer's disease, Parkinson's disease, and autism spectrum disorders. Recent research has uncovered the involvement of Eph receptors in synaptic dysfunction, neuronal degeneration, and cognitive impairment. Modulating Eph receptor signaling holds promise as a potential therapeutic approach for these disorders.
- Cardiovascular Diseases: Eph receptors have been implicated in various cardiovascular conditions. Studies have highlighted the role of Eph receptors in angiogenesis, blood vessel development, and cardiac remodeling. Dysregulated Eph receptor signaling has been associated with conditions such as atherosclerosis, hypertension, and cardiac hypertrophy. Targeting Eph receptors and their ligands may offer potential therapeutic interventions for cardiovascular diseases.
- Immune System Regulation: Eph receptors and Ephrins play roles in immune cell interactions and regulation of the immune response. Recent studies have demonstrated their involvement in immune cell migration, T cell activation, and inflammation. Dysregulated Eph receptor signaling has been linked to autoimmune diseases, such as multiple sclerosis and rheumatoid arthritis. Understanding the precise mechanisms underlying Eph receptor-mediated immune modulation could lead to the development of targeted immunotherapies.
- Tissue Regeneration and Repair: Eph receptors have been implicated in tissue regeneration and wound healing processes. Recent research has shown their involvement in cell migration, proliferation, and tissue reorganization during tissue repair. Manipulating Eph receptor signaling pathways may hold therapeutic potential for enhancing tissue regeneration and promoting wound healing.
- Therapeutic Targeting: Given the involvement of Eph receptors in various diseases, efforts are underway to develop targeted therapies that modulate Eph receptor signaling. This includes the development of small molecule inhibitors, antibodies, and engineered Eph receptor-based therapeutics. Preclinical studies and early-phase clinical trials are exploring the efficacy of these approaches in cancer treatment and other disease contexts.
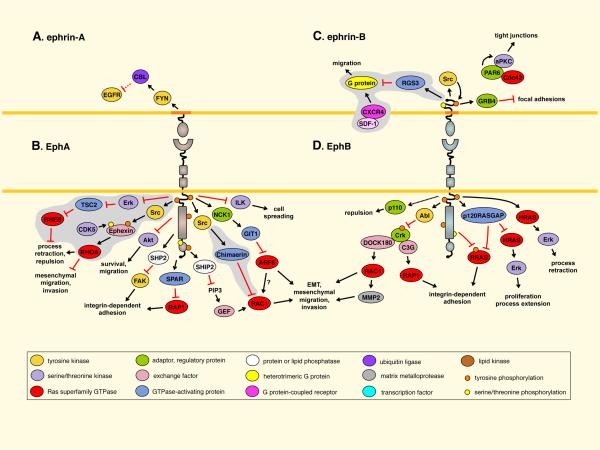 Fig.2 Tumor suppression through bidirectional signaling. (Pasquale EB, et al., 2010)
Fig.2 Tumor suppression through bidirectional signaling. (Pasquale EB, et al., 2010)
Overall, recent research advances have shed light on the multifaceted roles of Eph receptors in disease onset and progression. Understanding the precise mechanisms of Eph receptor signaling and its interactions with other signaling pathways is crucial for developing therapeutic strategies that target these receptors effectively. Further research in this field holds promise for improving our understanding of disease pathogenesis and developing innovative treatments for a range of conditions.
If you have any questions, requirements, or cooperation intentions, please feel free to contact us. We very much look forward to working with you and helping you achieve research and commercial success.
Related References
- Kou CJ, Kandpal RP. Differential expression patterns of Eph receptors and Ephrin ligands in human cancers. Biomed Res Int. 2018;2018:7390104. Published 2018 Feb 28.
- Pasquale EB. Eph receptors and ephrins in cancer: bidirectional signalling and beyond. Nat Rev Cancer. 2010;10(3):165-180.

