Cell Biology: The Importance of PEFs in Cell Mitosis
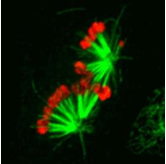
The picture shows the expression of the chromokinesin NOD (red) stabilizes aberrant interactions between kinetochores and spindle microtubules (green).
Cell division helps to maintain the even distribution genome of two daughter cells in which every chromosome need to properly interact with spindle. However, the interaction errors are difficult to avoid since every of the 46 chromosomes must to locate perfectly whenever a cell divides and commonly seen. Some mitosis experts said the error rate could over 80 and even 90 percent.
Some errors in cell division might lead to apoptosis or human diseases. But the mechanism was not totally clear. Thanks to a lately study, cell biologists revealed new details about a molecular surveillance system that helps detect and correct errors in cell division.
The key to flawless cell division is to correct dangerous interactions before the cell splits in two.
Using fruit fly tissue culture cells, researchers developed a way to watch and record images of the key players in cell division including microtubule filaments that form the mitotic spindle and sites called kinetochores that mediate chromosome-microtubule interactions. They also examined the contribution of a force generated by polar ejection force (PEF), which is thought to help handle the chromosome structure—line up the chromosomes in the middle of the spindle for division. In normal cell division, chromosomes line up in the center, where two copies of each chromosome are held together with "molecular glue" until signaled to dissolve the glue and divide.
For the first time, they directly tested and quantified how PEF exactly influences tension at kinetochores and affects error correction in mitosis. They conduct their experiments inside living cells. And then each chromosome copy is then pulled to opposite poles of the cell, escorted in what looks like a taffy pull away from the center as two new daughter cells are formed.
During the split, molecular engines pull the copies apart along microtubule tracks that take an active role in the process that includes shortening microtubules by large, flexible scaffold-like protein structures called kinetochores that assemble on every chromosome during division.
Overall, this well-orchestrated process prevents serious problems such as aneuploidy, that is, too many chromosomes in daughter cells. Aneuploidy in somatic or body cells leads to cell death and is a hallmark of most cancer cells. But in eggs or sperm, it leads to serious birth defects and miscarriages.
In properly aligned division, microtubules from opposite spindle poles tug chromosome copies toward opposite poles, but they stick together with molecular glue until the proper moment. This creates tension at the kinetochores and stabilizes their interactions with microtubules. However, if attachments are bad, or syntelic, both copies attach to the same pole, leading to chromosome mis-segregation and aneuploidy if uncorrected. Cells have a surveillance mechanism that allows them to wait for every chromosome to properly align before divvying up the chromosomes as the study showed.
To study this at the molecular level, researchers designed experiments to trick the cellular machinery into the overexpression of the molecular engine that produces PEF. Surprisingly, this caused a dramatic increase in a type of bad kinetochore-microtubule interaction called syntelic attachments. They also fluorescently tag chromosomes, microtubules, kinetochores and the molecular engine kinesin with different colors to visualize interactions in real time using a special microscope to image single molecules. Quantifying the amount of fluorescence of the force-producing molecular engine, they were able to assess the relative strength of the PEF in cells.
During this process, they saw the detection pathway preventing the molecular glue from dissolving until every chromosome is correctly aligned. The delay gives the cell time to correct errors. Thus they propose that these bad syntelic attachments are normally very short-lived because they are not under proper tension. However, when we experimentally elevate PEF, tension is introduced at attachments that do not typically come under tension, essentially tricking the cell into thinking these chromosomes are properly aligned.
In cells with elevated PEFs, the correction pathway is overridden, the detection mechanism is silenced and the result is disastrous because it leads to severe aneuploidy. This research has taught us about how an important molecular engine generates the PEF and how this force affects the accuracy of cell division.
Article Link: Cell Biology: The Importance of PEFs in Cell Mitosis

