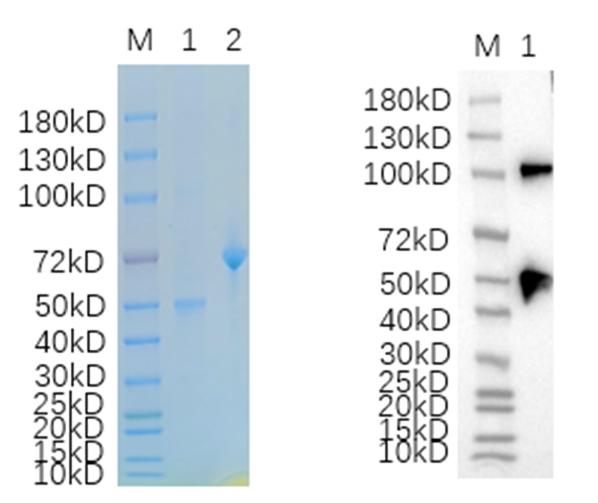
IVD of Herpes Simplex Virus Type 2
🧪 VP13 & VP14-458V
Source: E.coli
Species: HSV2
Tag: Non
Conjugation:
Protein Length: 1-280 a.a.

Herpes Simplex Virus Type 2 (HSV-2)
Genital herpes is a sexually transmitted disease caused by herpes simplex virus type 2 (HSV-2). It can be transmitted through sexual contact or perinatal infection of newborns in the uterus or birth canal and has a particularly serious impact on women. After the HSV-2 virus infects the human body, it will lie dormant in the skin, mucous membranes, and nervous tissue. During a period of reduced immunity, latent viruses can become active and cause recurrent infections. An individual infected with HSV-2 is also more likely to be infected with and transmit immunodeficiency virus (HIV).
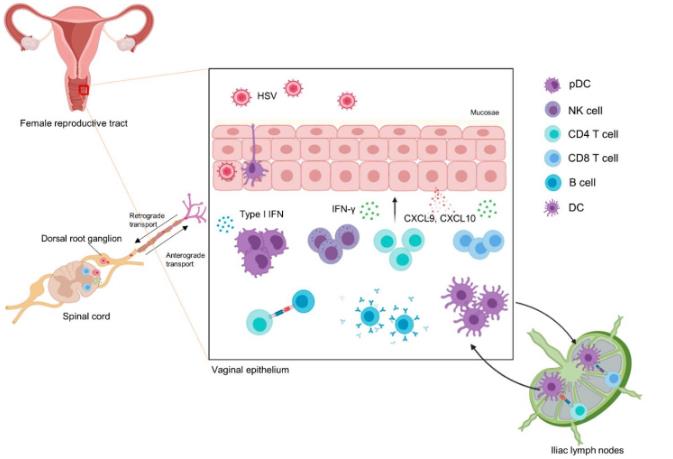 Figure 1. Anti-HSV immune response when HSV enters the female human body, epithelial cells recognize HSV in a PRR-dependent or PRR-independent manner. (Kim H C, et al., 2020)
Figure 1. Anti-HSV immune response when HSV enters the female human body, epithelial cells recognize HSV in a PRR-dependent or PRR-independent manner. (Kim H C, et al., 2020)Main Methods of IVD for Herpes Simplex Virus Type 2
- Serological testing. Infection can be determined by detecting HSV-2 antibodies in serum.
- PCR test. Through the use of PCR reactions, the genetic material of the HSV-2 can be detected in specimens collected from lesions or body fluids, such as cerebrospinal fluid.
- Perform in vitro cell culture. The collected specimens are inoculated into specific cells, and the presence of virus proliferation is observed through cell culture to determine whether the virus is infected with HSV-2.
- Cellular immunohistochemistry, immunofluorescence technology, and in situ hybridization technology.
Creative BioMart can provide high-quality recombinant HSV-2 proteins for IVD, including ELISA, lateral flow assays, western blots, and other immunoassays.
Highlights of Our Products
- High purity and uniformity. The protein purity is above 90% and verified by SDS-PAGE.
- Completed biological functions and efficient activity.
- A wide range of immune tests are available, such as ELISA, lateral flow, WB, and others.
- Easy to store and transport, conducive to large-scale production and use of vaccines.
- High batch-to-batch consistency and low variability.
Our Outstanding Advantages
- Viral proteins are continuously improved and come in a wide variety to meet different research and development needs.
- With a professional R&D team and advanced laboratory facilities, we can conduct a variety of production research and development.
- A complete IVD protein platform can provide customized services to meet different scientific research needs.
- High-quality service, high-level experiments, and reliable analysis.
- IVD proteins can be used to test for a variety of diseases and conditions, making them valuable tools for diagnosing and monitoring health.
Other Applications of HSV-2
1. Diagnostic Tools:
Serological Testing: HSV-2 glycoproteins, such as gG-2, are commonly used in serological assays to detect HSV-2 specific antibodies in patients' blood. This helps differentiate between HSV-1 and HSV-2 infections.
PCR and Immunoassays: Proteins from HSV-2 can be targets in molecular assays like PCR and immunoassays to detect viral DNA or proteins directly from clinical samples.
2. Vaccine Development:
Subunit Vaccines: HSV-2 proteins, particularly viral glycoproteins, are candidates for subunit vaccines. Injecting purified viral proteins can elicit an immune response without requiring a live virus.
Vector-based Vaccines: HSV-2 proteins are expressed in viral vectors to stimulate an immune response.
3. Antiviral Drug Screening:
Target Identification: HSV-2 proteins are essential for identifying potential antiviral targets. For example, viral enzymes like DNA polymerase and proteases can be inhibited by small molecules.
High-Throughput Screening (HTS): Recombinant HSV-2 proteins are used in HTS assays to identify novel antiviral compounds.
Basic Research:
4. Viral Replication and Pathogenesis:
Understanding the role of HSV-2 proteins in viral replication, entry, and immune evasion helps elucidate the virus's life cycle and pathogenesis.
Protein-Protein Interactions: Studying interactions between HSV-2 proteins and host proteins can reveal mechanisms of viral infection and potential therapeutic targets.
5. Therapeutic Targets:
Immunotherapeutics: HSV-2 proteins are used to develop therapies that harness the immune system, such as monoclonal antibodies against viral glycoproteins.
Gene Editing: Technologies like CRISPR-Cas9 can be guided to target HSV-2 genes, using knowledge of viral proteins to disrupt viral replication.
6. Immune Response Studies:
T-cell Response Profiling: HSV-2 proteins help in studying the T-cell responses in infected individuals, which is crucial for understanding immunity and vaccine development.
Antibody Response: Analyzing how the body's antibodies react to HSV-2 proteins aids in designing better therapeutics and vaccines.
7. Biomarker Discovery:
Disease Progression: Specific HSV-2 proteins can serve as biomarkers for disease progression and therapeutic response in infected individuals.
Immune Evasion: Studying how HSV-2 proteins help the virus evade the host immune system can lead to the discovery of new therapeutic targets.
Case Study
Case 1: Ren F, Narita R, Rashidi AS, Fruhwürth S, Gao Z, Bak RO, Thomsen MK, Verjans GM, Reinert LS, Paludan SR. ER stress induces caspase-2-tBID-GSDME-dependent cell death in neurons lytically infected with herpes simplex virus type 2. EMBO J. 2023 Oct 4;42(19):e113118. doi: 10.15252/embj.2022113118. Epub 2023 Aug 30. PMID: 37646198; PMCID: PMC10548179.
Neurotropic viruses, including herpes simplex virus (HSV) types 1 and 2, have the capacity to infect neurons and can cause severe diseases. This is associated with neuronal cell death, which may contribute to morbidity or even mortality if the infection is not controlled. However, the mechanistic details of HSV-induced neuronal cell death remain enigmatic. Here, we report that lytic HSV-2 infection of human neuron-like SH-SY5Y cells and primary human and murine brain cells leads to cell death mediated by gasdermin E (GSDME).
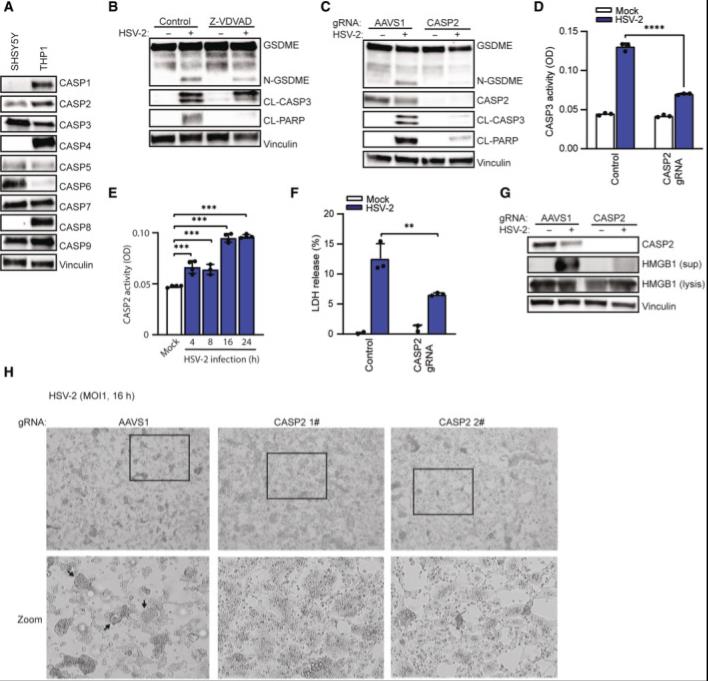 Figure 2. HSV-2-induced pyroptotic cell death in SH-SY5Y cells depends on CASP2
Figure 2. HSV-2-induced pyroptotic cell death in SH-SY5Y cells depends on CASP2A Lysates from SH-SY5Y and THP1 cells were immunoblotted for a panel of caspases and vinculin.
B SH-SY5Y cells were pretreated with CASP2 inhibitor Z-VDVAD (20 μM) for 1 h and infected with HSV-2 (MOI = 1). Lysates were isolated 16 hpi for immunoblot analysis.
C, D SH-SY5Y cells were transfected with CASP2 gRNA-Cas9 RNPs or AAVS1 gRNA-Cas9 RNPs and infected with HSV-2. Lysates were isolated 16 h post-infection and assayed for CASP3 activity (D, ELISA) and immunoblotted for GSDME, CASP2, cleaved caspase 3 (CL-CASP3), cleaved PARP (CL-PARP), and vinculin (C).
E SH-SY5Y cells were infected with HSV-2 (MOI = 1) for the indicated time points and lysates were analyzed for CASP2 activity.
F, G SH-SY5Y cells were infected with HSV-2. Supernatant and lysates were isolated for measurement of LDH (F, 22 h) and identification of HMGB1 (G, 16 h).
H SH-SY5Y cells transfected with AAVS1 or CASP2 gRNA-Cas9 RNPs and infected with HSV-2 for 16 h and examined for morphological changes by microscopy. Boxes indicate areas highlighted in the zoomed images. Arrows indicate ballooning or syncytial cells.
Case 2: Bourne N, Keith CA, Miller AL, Pyles RB, Cohen G, Milligan GN. Boosting of vaginal HSV-2-specific B and T cell responses by intravaginal therapeutic immunization results in diminished recurrent HSV-2 disease. J Virol. 2023 Sep 28;97(9):e0066923. doi: 10.1128/jvi.00669-23. Epub 2023 Sep 1. PMID: 37655939; PMCID: PMC10537585.
Boosting herpes simplex virus (HSV)-specific immunity in the genital tissues of HSV-positive individuals to increase control of HSV-2 recurrent disease and virus shedding is an important goal of therapeutic immunization and would impact HSV-2 transmission. Experimental therapeutic HSV-2 vaccines delivered by a parenteral route have resulted in decreased recurrent disease in experimental animals. Researchers used a guinea pig model of HSV-2 infection to test if HSV-specific antibody and cell-mediated responses in the vaginal mucosa would be more effectively increased by intravaginal (Ivag) therapeutic immunization compared to parenteral immunization.
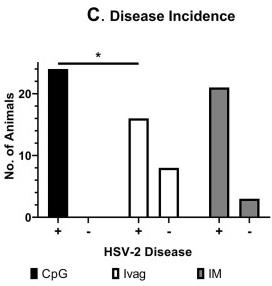 Fig3. Fig 3 Decreased recurrent disease, but not HSV-2 shedding, in Ivag therapeutically immunized guinea pigs. (C) Incidence of recurrent disease. Data show the number of animals experiencing recurrent lesions versus animals in which recurrent lesions were not detected for each immunization group on days 28–63 PI (*P < 0.004, Fisher's exact test).
Fig3. Fig 3 Decreased recurrent disease, but not HSV-2 shedding, in Ivag therapeutically immunized guinea pigs. (C) Incidence of recurrent disease. Data show the number of animals experiencing recurrent lesions versus animals in which recurrent lesions were not detected for each immunization group on days 28–63 PI (*P < 0.004, Fisher's exact test).Case 3: Koelle DM, Norberg P, Fitzgibbon MP, Russell RM, Greninger AL, Huang ML, Stensland L, Jing L, Magaret AS, Diem K, Selke S, Xie H, Celum C, Lingappa JR, Jerome KR, Wald A, Johnston C. Worldwide circulation of HSV-2 × HSV-1 recombinant strains. Sci Rep. 2017 Mar 13;7:44084. doi: 10.1038/srep44084. PMID: 28287142; PMCID: PMC5347006.
Homo sapiens harbor two distinct, medically significant species of simplexviruses, herpes simplex virus (HSV)-1 and HSV-2, with estimated divergence 6-8 million years ago (MYA). Unexpectedly, we found that circulating HSV-2 strains can contain HSV-1 DNA segments in three distinct genes. Using over 150 genital swabs from North and South America and Africa, researchers detected recombinants worldwide.
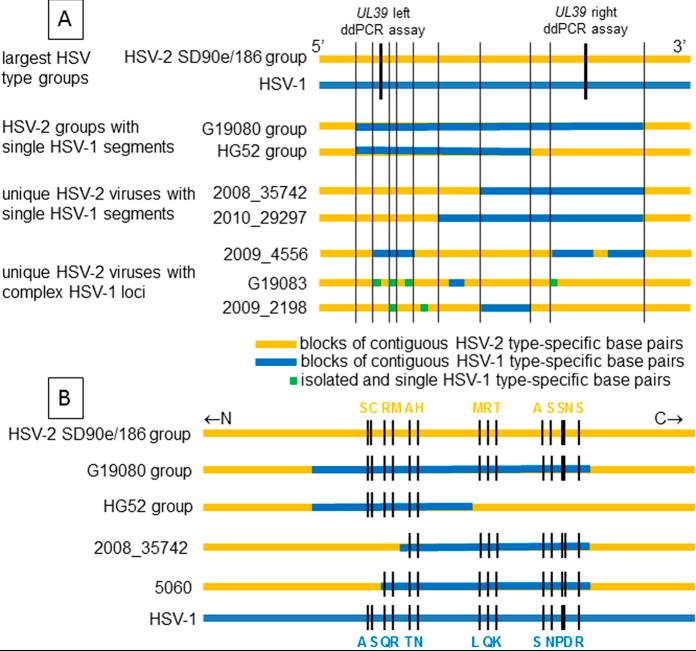 Fig4. HSV UL39 variants in circulating strains. Horizontal lines represent C-terminal UL39 sequences to approximate scale. Group and virus names at left. (A) Genotypes. At top, the largest HSV-2 clade similar to strains SD90e and 186 (top) is yellow and the HSV-1 group is blue. Within IRV, blue bars represent zones of 4 or more contiguous HSV-1 variant SNPs. Green spots are isolated SNPs containing one HSV-1 variant nucleotide. Thin vertical black lines represent short crossover zones indistinguishable between HSV genotypes. Thick vertical black lines show approximate locations of type-specific ddPCR genotyping assays. (B) Amino acid variations in selected groups and viruses. Strains SD90e and 186 (top) are yellow and HSV-1 group (bottom) is blue. Blue bars in IRV represent zones of contiguous SNPS with HSV-1 variant nucleotides. Short black lines represent locations of color coded amino acid differences between strains.
Fig4. HSV UL39 variants in circulating strains. Horizontal lines represent C-terminal UL39 sequences to approximate scale. Group and virus names at left. (A) Genotypes. At top, the largest HSV-2 clade similar to strains SD90e and 186 (top) is yellow and the HSV-1 group is blue. Within IRV, blue bars represent zones of 4 or more contiguous HSV-1 variant SNPs. Green spots are isolated SNPs containing one HSV-1 variant nucleotide. Thin vertical black lines represent short crossover zones indistinguishable between HSV genotypes. Thick vertical black lines show approximate locations of type-specific ddPCR genotyping assays. (B) Amino acid variations in selected groups and viruses. Strains SD90e and 186 (top) are yellow and HSV-1 group (bottom) is blue. Blue bars in IRV represent zones of contiguous SNPS with HSV-1 variant nucleotides. Short black lines represent locations of color coded amino acid differences between strains.In addition, Creative BioMart also offers a series of viral proteins and protein-related services to provide customers with high-quality, low-cost active recombinant proteins to meet different needs and assist in preclinical drug development.
Reference
- Kim H C, Lee H K. (2020). Vaccines against genital herpes: Where are we?[J]. Vaccines. 8(3): 420.