Immunocytochemistry protocol
Immunocytochemistry (ICC) is a widely used technique to study the ex
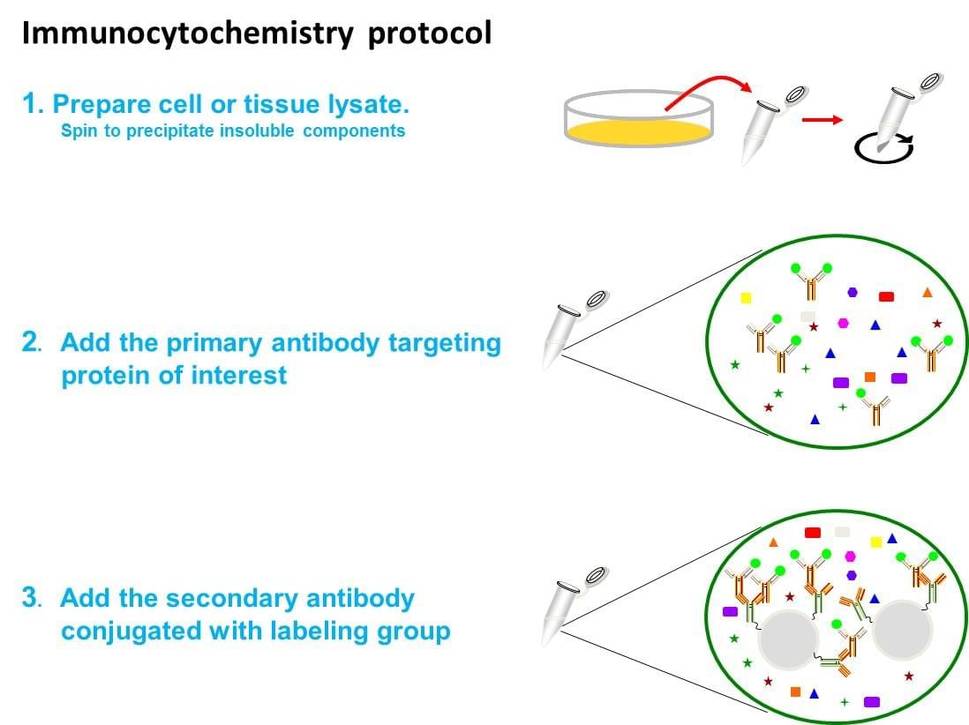
Fig. The schematic diagram for immunocytochemistry.
Immunocytochemistry protocol
Materials
Primary antibody
Secondary antibody
PBS
Triton-X100
Serum (depends on the host of the secondary antibody)
Hoechst 33258 or DAPI
Cell culture
- For immunocytochemistry, cells are grown on coverslips and cells should not reach over 80% confluency as recommendation.
Cell fixation
- Remove the medium and wash cells once with PBS (pH 7.4).
- Cells are fixed with 4% p -formaldehyde in PBS for 20 min at room temperature.
- Wash cells twice with PBS (pH 7.4).
- Incubate cells with 0.1% Triton-X100 for 5 min Permeabilize cell membrane.
- Wash cells twice with PBS (pH 7.4).
Staining
- Block nonspecific binding sites in PBS for 1 h with 10% serum which depends on the host of the secondary antibody.
- Primary antibodies (1-2 ug/ml) and secondary antibodies (5-10 ug/ml) are diluted in 0.1% serum in PBS. Incubate cell with primary antibody at 37 °C for 2 h or at 4 °C overnight.
- Wash with PBS 3 times for 15 min each.
- Incubate cell with secondary antibody at 37 °C for 1 h protecting from light.
- Wash with PBS 3 times for 15 min each.
- Nuclei are stained with Hoechst 33258 or DAPI in PBS for 15 min at room temperature.
- Coverslips are then mounted with medium for fluorescent staining.
- Once the medium dried, the slides are imaged with confocal fluorescence microscopy.

