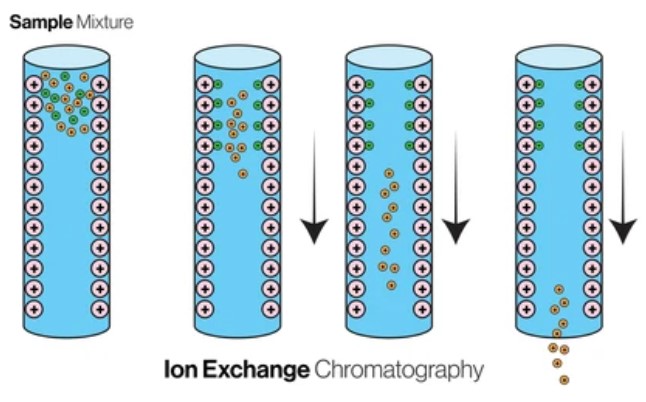
Ion Exchange Chromatography
🧪 diethylaminoethyl-004C
Source:
Species:
Tag: Non
Conjugation:
Protein Length:
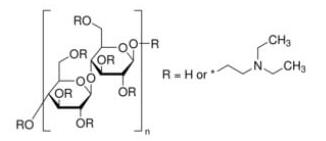
Background
Overview
Ion Exchange Chromatography (IEC) is a chromatography technology based on the principle of ion exchange, which is widely used in the separation and purification of biological macromolecules such as proteins, nucleic acids, peptides and so on.
The basic principle of ion-exchange chromatography is to use the electrostatic attraction between the charge groups on the ion-exchange resin and the ions in the sample. Ion exchange resins are usually composed of high polymer with ionizable functional groups on the surface, such as negatively charged groups such as sulfuric acid group (-SO3H) and carboxylic group (-COOH), or positively charged groups such as amino group (-NH3+).
In the ion exchange process, the ions in the sample will undergo a reversible exchange reaction with the ions on the resin. By changing the ionic strength and pH value of the mobile phase or adding specific competitive ions, the binding strength of the resin and the ions in the sample can be adjusted, so as to achieve the elution and separation of the target molecules.
Types of Ion Exchange Resins
Ion Exchange resins are key materials in Ion Exchange Chromatography (IEC). They are divided into two main categories based on the type of ions that can be exchanged and their charge properties: cation exchange resins and anion-exchange resins. Each resin has its specific structure and function and is suitable for different separation needs.
Cation exchange resin
Cation exchange resins contain negatively charged active groups that can exchange cations in solution. These resins are usually used under acidic conditions because they have a high exchange capacity under such conditions. Common types of cation exchange resins include:
- Sulfuric acid resin: contains sulfuric acid group (-SO3H) as the active group, suitable for the exchange of various cations, especially at low pH conditions.
- Carboxylic resin: Containing carboxyl group (-COOH) as the active group, suitable for cation exchange under neutral or alkaline conditions.
- Phosphoric acid type resin: containing phosphoric acid group (-PO3H2) as the active group, with high selectivity, often used for the separation of specific cations.
Anion exchange resin
Anion exchange resins contain positively charged active groups that are able to exchange anions in solution. These resins are usually used under alkaline conditions because they have a high exchange capacity under such conditions. Common types of anion exchange resins include:
- Quaternary ammonium resin: contains quaternary ammonium salt group (-NR4+, where R can be hydrogen or organic group) as the active group, suitable for the exchange of a variety of anions.
- Amino resin: Containing an amino group (-NH3+) as the active group, suitable for anion exchange under neutral or acidic conditions.
- Imidazole-type resin: containing imidazole rings as active groups, with high selectivity, often used for the separation of specific anions.
Other properties of resin
In addition to charge type, ion exchange resins can also be classified according to the following characteristics:
The crosslinking degree of resin affects its physical stability and exchange ability. The resin with high crosslinking degree is harder and has better mechanical stability, but the exchange rate may be slower; Low crosslinked resins are softer and exchange faster, but have poor mechanical stability.
The particle size and pore structure of resin affect its ability to screen molecules and exchange efficiency. Resins with small particles and large pore structures provide higher resolution and faster exchange rates, but may increase column pressure.
Different ion exchange resins have different selectivity for specific ions, which allows them to be used for specific types of ion separation.
Advantages of IEC
Ion exchange chromatography has the advantages of high resolution and high throughput, making it one of the most commonly used chromatography methods in protein purification. In addition, there are many other advantages that make it widely used.
High selectivity. Ion-exchange chromatography enables highly selective separation based on charge interactions between molecules. By selecting the appropriate ion exchange resin and adjusting the mobile phase conditions, it is possible to effectively separate specific ions or molecules.
High separation efficiency. Ion exchange chromatography has a high separation efficiency and can achieve the separation of multiple components in complex samples in a single operation. This is especially useful for purifying target molecules from mixed samples.
Repeatability. The operating conditions of ion-exchange chromatography (e.g. resin type, mobile phase pH and ion strength, elution method, etc.) can be precisely controlled to ensure repeatability of each experiment.
Scalability. Ion-exchange chromatography can be extended from small-scale experimental separation to large-scale production processes. By adjusting the column volume and flow rate, it can adapt to different throughput requirements.

Applications
- Purification of proteins and peptides. Proteins are major molecules that perform many functions in living organisms, and their purification is a key step in biochemical research and biopharmaceutical production. Ion exchange chromatography can effectively separate proteins according to their charge differences and achieve highly purified protein preparation. It is of great significance to study the structure and function of proteins and to develop new drugs.
- Preparation of antibodies. Antibodies are proteins produced by the immune system to recognize and neutralize foreign pathogens. In the field of biopharmaceuticals, the purification of specific antibodies is essential for the development of therapeutic antibody drugs. Ion exchange chromatography provides a highly efficient method to purify and concentrate antibodies.
- Separation of nucleic acids. Nucleic acids (such as DNA and RNA) are carriers of genetic information. In molecular biology research, ion exchange chromatography can be used to purify specific nucleic acid fragments, which is essential for experiments such as gene cloning, sequencing, and expression analysis.
- Purification of polysaccharides. Polysaccharide is a kind of complex carbohydrate with many biological functions. In the field of food industry and biomedicine, the purification of polysaccharides is of great value for studying their biological activities and developing polysaccharide based drugs.
- Analysis of biological samples. Ion exchange chromatography is also commonly used for the analysis of biological samples, such as the quantitative and qualitative analysis of specific molecules in blood, urine, or other biological fluids. This is of great significance for diagnosis and treatment monitoring of diseases.

Case Study
Case Study 1: CM Sepharose Fast Flow
Bacteriocins have antimicrobial activities against food-spoiling bacteria and food-borne pathogens. Paracin 1.7, a bacteriocin synthesized by Lactobacillus paracasei HD1-7 isolated from Chinese sauerkraut juice, was studied. Following partial purification with ammonium sulfate precipitation, CM Sepharose Fast Flow, and Sephadex G-10 chromatography, the molecular weight of Paracin 1.7 was about 10 kDa based on Tricine-SDS-PAGE results. A 2.87 fold purified bacteriocin was produced, reaching a final yield of 39.93% and the specific activity of 1.56 × 10(3) AU/mg. The N-terminal amino acid sequence of Paracin 1.7 was VSNTFFA, and the LC/LTQ results revealed that the N-terminal amino acid sequence was similar to that of ABC-type oligopeptide transport system protein and N-acetylmuramoyl-L-alanine amidase. Paracin 1.7 was sensitive to protease K, had antimicrobial activities at a broad pH range (3.0-8.0), and was heat resistant (121 °C for 20 min).

(Jingping Ge, 2016)
Fig1. Antibacterial activities of precipitate and supernatant with different saturation of ammonium sulfate.
Case Study 2: DEAE Cellulose Resin
Ion exchange chromatography is a method that uses the different surface charges of trypanosomes and blood cells to separate them. This makes it possible to use molecular and immunological methods to diagnose or study these protozoans. DEAE-cellulose resin is commonly used to perform this method. The goal of this study was to compare three novel chromatographic resins designated as PURIFICA™ (Y-C2N®, Y-HONOH®, and Y-CNC3®). The resins were evaluated based on their ability to isolate the parasite, purification time, examination of parasite viability and morphology, and trypanosome recovery potential after passing through the columns. In terms of the evaluated parameters, there was no significant difference between DEAE-cellulose and the three tested resins in most experiments. However, PURIFICA™ (Y-C2N®, Y-HONOH®, and Y-CNC3®) resins are less expensive and easier to prepare than DEAE-Cellulose, making them an alternative for the purification of Trypanosoma evansi.
(Júlia Marques, 2023)
Fig2. SSC-A/FSC-A-DEAE-Cellulose purified parasites.
Case Study 3: Q Agarose fast flow
This work describes a novel extracellular lipolytic carboxylester hydrolase named FAL, with lipase and phospholipase A1 (PLA1) activity, from a newly isolated filamentous fungus Ascomycota CBS strain, identified as Fusarium annulatum Bunigcourt. FAL was purified to about 62-fold using ammonium sulphate precipitation, Superdex® 200 Increase gel filtration and Q-Sepharose Fast Flow columns, with a total yield of 21%. The specific activity of FAL was found to be 3500 U/mg at pH 9 and 40°C and 5000 U/mg at pH 11 and 45°C, on emulsions of triocanoin and egg yolk phosphatidylcholine, respectively. SDS-PAGE and zymography analysis estimated the molecular weight of FAL to be 33 kDa. FAL was shown to be a PLA1 with a regioselectivity to the sn-1 position of surface-coated phospholipids esterified with α-eleostearic acid. The analysis of washing performance showed that it has the capability to efficiently eliminate oil-stains.
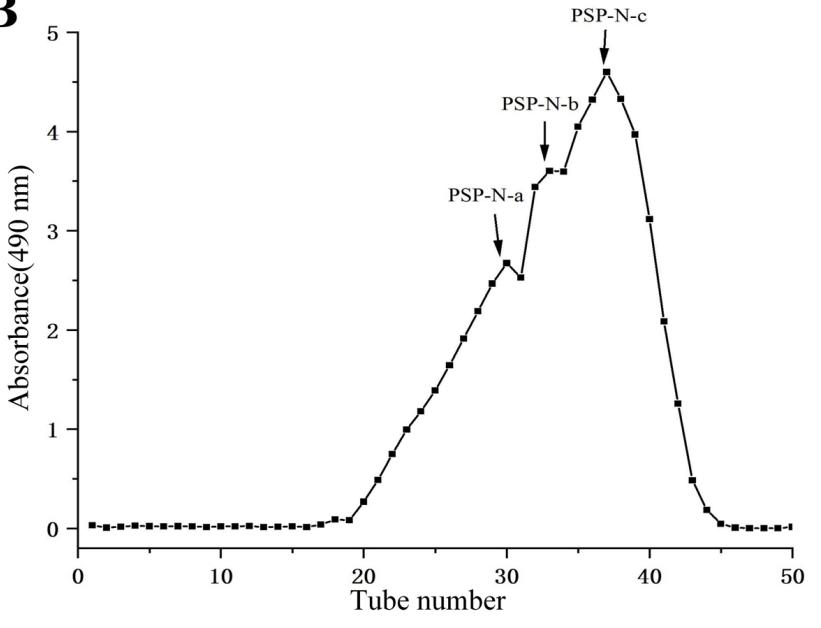
(Ahlem Dab, 2023)
Fig3. Chromatogram profile of FAL purification on a HiTrap™ Q-Sepharose FF column.
Advantages
- Product diversity. We can offer many types of ion exchange chromatography columns, including cation exchange columns and anion exchange columns, to suit the purification needs of different types of proteins or other biomacromolecules.
- Application support. We not only provide products, but also may provide relevant technical support and application guidance to help customers optimize purification processes, improve purification efficiency and product purity.
- Customized services. We can provide customized fillers and services to meet the requirements of different biological processes according to the specific needs of customers.
Creative BioMart can provide various types of ion exchange chromatography related products to meet customer’s different research goals. You can also let us know if you have any other customized requirements. Please contact us for more product details.
References
- Ge J.; et al. Purification and Partial Characterization of a Novel Bacteriocin Synthesized by Lactobacillus paracasei HD1-7 Isolated from Chinese Sauerkraut Juice. Sci Rep. 2016;6:19366.
- Marques J.; et al. Comparative study of three novel ion exchange resins with DEAE-cellulose for the purification of Trypanosoma evansi. Anal Biochem. 2023;676:115226.
- Dab A.; et al. Biochemical characterization of an alkaline and detergent-stable Lipase from Fusarium annulatum Bugnicourt strain CBS associated with olive tree dieback. PLoS One. 2023;18(5):e0286091.















