Slit Ligands and ROBO Receptors
Related Symbol Search List
Immunology Background
Background
Slit Ligands
Slit ligands are a family of secreted proteins that play critical roles in guiding axons during neural development, as well as in various other biological processes. They are primarily expressed during embryonic development and are known for their repulsive effects on growing axons. The identification of Slit ligands was first associated with their function in the fruit fly (Drosophila melanogaster), where they were discovered to help regulate neuronal pathways.
Key Features of Slit Ligands
- Structure: Slit proteins are large glycoproteins that exist in multiple isoforms. They contain several functional domains, including EF-hand domains and Leucine-rich repeats (LRRs), which are important for their interactions with receptors.
- Function: Slit ligands provide guidance cues for axons, helping to organize the developing nervous system. They play roles not only in repulsion but also in cell migration, tissue patterning, and organogenesis.
- Expression Patterns: Slit ligands are expressed in various tissues, contributing to distinct developmental processes beyond the nervous system, including vascular development and the regulation of epithelial structures.
ROBO Receptors
ROBO (Roundabout) receptors are a family of membrane-bound receptors that interact with Slit ligands. The ROBO family includes several members (ROBO1, ROBO2, ROBO3, and ROBO4), each with specific expression patterns and functions in the body.
Key Features of ROBO Receptors
- Structure: ROBO receptors are characterized by their Ig-like domains and fibronectin type III repeats, which are involved in ligand binding. The extracellular domain allows for the interaction with Slit ligands, while the intracellular domain influences signaling pathways involved in cellular responses.
- Function: When Slit ligands bind to ROBO receptors, they initiate a repulsive signaling response that helps guide axons away from Slit sources. This is crucial for establishing precise neural circuits. ROBO receptors also have roles in cell migration and the maintenance of axonal pathways.
- Signaling Pathways: ROBO receptors activate several intracellular signaling pathways, including those involving Rho GTPases, which modulate the cytoskeleton and ultimately dictate cell behavior.
The interplay between Slit ligands and ROBO receptors is fundamental to many developmental processes, particularly in the nervous system. By providing guidance signals that influence axon growth and cell migration, these molecules ensure the proper formation of neural circuits and tissue organization. Understanding the molecular mechanisms of Slit-ROBO interactions can illuminate their roles in development and disease, providing insights into potential therapeutic strategies for neural injuries and congenital disorders.
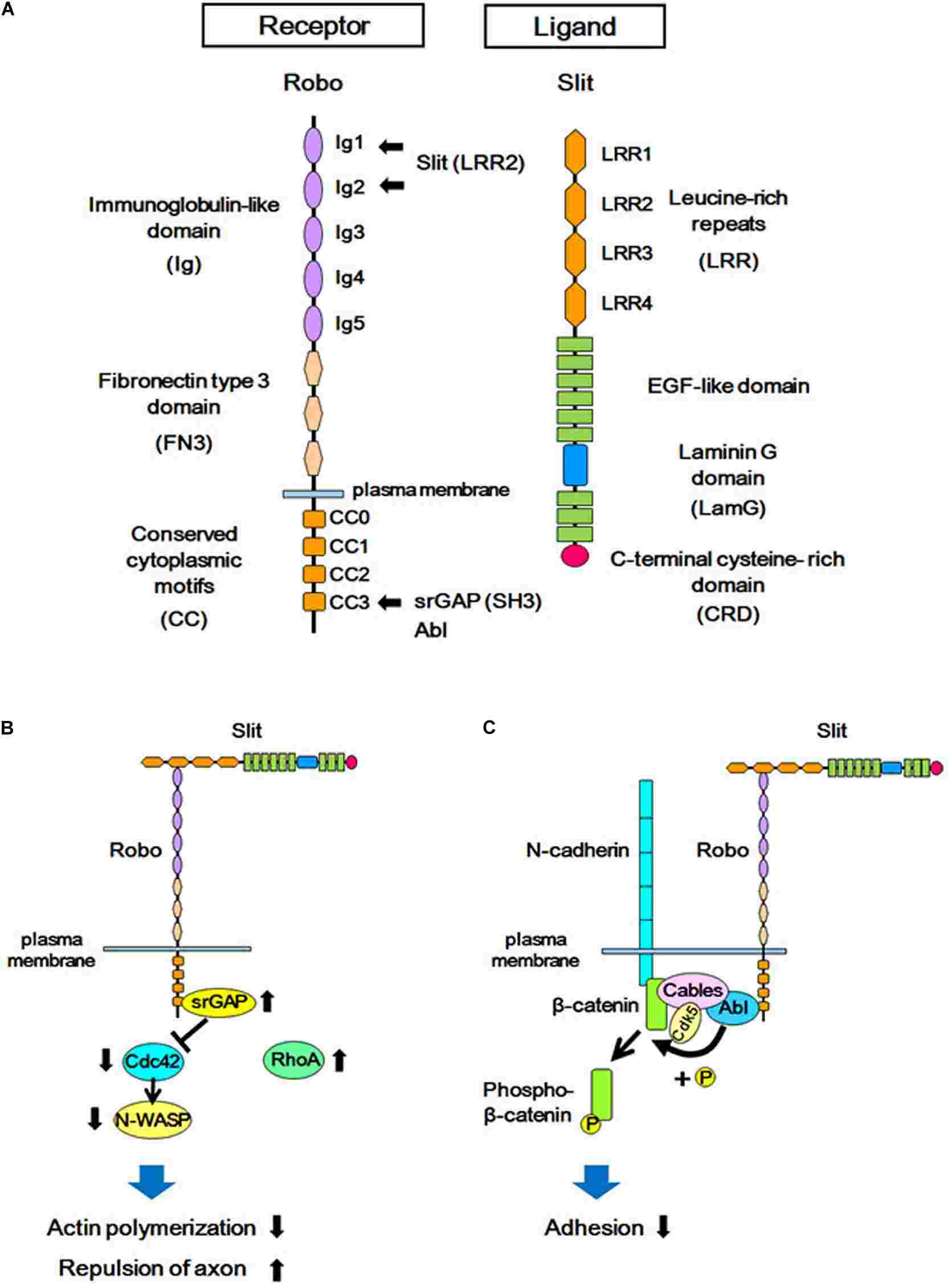 Fig.1 Structures of Slit/Robo, and the Slit-Robo signaling pathway. (Gonda Y, et al., 2020)
Fig.1 Structures of Slit/Robo, and the Slit-Robo signaling pathway. (Gonda Y, et al., 2020)Signaling Mechanisms of Slit Ligands and ROBO Receptors
Binding Dynamics
- Slit proteins bind to ROBO receptors in a manner that initiates intracellular signaling. The Slit-ROBO interaction is primarily mediated through the extracellular domains of ROBO.
- ROBO2, in particular, is a key mediator of Slit-induced repulsion of growth cones.
Intracellular Pathways
- Cytoskeletal Regulation: The binding of Slit to ROBO results in the activation of Rho family GTPases (e.g., RhoA, Rac1), which regulate cytoskeletal dynamics.
- Inhibition of Cell Adhesion: ROBO signaling can decrease integrin-mediated adhesion, allowing for enhanced cell mobility.
- Activation of Phospholipase C (PLC): Slit-ROBO interaction can activate PLC, leading to an increase in intracellular calcium, affecting downstream signaling events.
Signaling Outcomes
- Repulsion and Guidance: The primary outcome of Slit-ROBO signaling is the repulsive guidance of neurons, ensuring correct axonal targeting.
- Cell Migration: Beyond neuronal development, Slit-ROBO signaling influences the migration and positioning of various cell types, including immune cells and mesenchymal cells.
Regulation of Gene Expression
- ROBO receptors can also influence gene expression through downstream signaling pathways, coordinating developmental processes and cellular responses.
Integration with Other Signaling Pathways
- The signaling pathways activated by Slit-ROBO interactions are often integrated with other signaling cascades to coordinate complex cellular responses. Cross-talk with pathways such as Wnt, Notch, and growth factor signaling pathways can further modulate the cellular outcomes of Slit-ROBO signaling.
The Slit ligands and ROBO receptors represent a vital signaling pathway in various biological processes beyond their initial role in axon guidance. Understanding the mechanisms of Slit-ROBO interactions provides insights into the complexities of cellular communication during development and their implications in diseases, highlighting their potential as targets for therapeutic interventions in conditions such as cancer and neurodegenerative disorders.
Common Slit Ligands and ROBO Receptors
Slit proteins (Slit1, Slit2, Slit3) and Robo receptors (ROBO1, ROBO2, ROBO3, ROBO4) are key components of the guidance system in the nervous system, playing crucial roles in directing neuronal growth and determining cellular behavior. Here's a summary of each:
| Type | Molecule | Details |
|---|---|---|
| Slit proteins | SLIT1 |
Function: Involved in repulsive guidance of migrating neurons and axons. It helps in neural crest cell migration and also plays a role in preventing axons from crossing inappropriate midline areas. Expression: Primarily expressed in the midline of the developing nervous system. |
| SLIT2 |
Function: Similar to Slit1, promotes repulsive guidance and influences axon trajectories. It is involved in the development of the spinal cord and forebrain. Role in Development: Helps in establishing boundaries between different tissue types. |
|
| SLIT3 |
Function: Less studied than Slit1 and Slit2 but also involved in axonal guidance and may have roles in angiogenesis and immune responses. Expression Patterns: Found in various tissues, suggesting functions beyond the nervous system. |
|
| ROBO Receptors | ROBO1 |
Function: Primarily mediates the repulsive effects of Slit proteins on growing axons. Critical for anterior-posterior pathway guidance in axons. Role in Neural Development: Essential for midline crossing of axons in the developing spinal cord. |
| ROBO2 |
Function: Similar to ROBO1, involved in guidance cues but can also act in conjunction with other receptors to modulate neuronal guidance. Involvement in Overlapping Pathways: Can form heterodimers with other Robo receptors. |
|
| ROBO3 |
Function: Distinct from ROBO1 and ROBO2, it has a unique role in allowing specific axons to cross the midline during development. Contribution to Midline Crossing: Known to be involved in the development of commissural axons and also has roles in the central nervous system. |
|
| ROBO4 |
Function: Primarily associated with vascular development and endothelial cell migration, rather than strictly in neuronal guidance. Role in Angiogenesis: Involved in blood vessel formation and maintaining vascular integrity. |
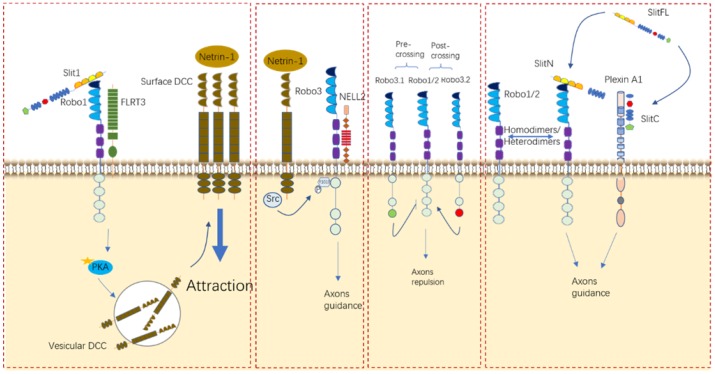 Fig.2 Roles of Slit/Robo in neurocyte navigation. (Tong M, et al., 2019)
Fig.2 Roles of Slit/Robo in neurocyte navigation. (Tong M, et al., 2019)
Robo receptors can interact with other receptors, such as FLRT3, DCC, and NELL2, and form homodimers and/or heterodimers to elicit their functions in different contexts.
The Role of Slit Ligands and ROBO Receptors in Varies Physiological Processes
Slit ligands and ROBO receptors play critical roles in various physiological processes, primarily involved in axon guidance, cell migration, and tissue patterning. Here's an overview of their roles in different physiological contexts:
Neuronal Development
- Axon Guidance: Slit ligands (Slit1, Slit2, Slit3) interact with ROBO receptors (ROBO1, ROBO2, ROBO3, ROBO4) to regulate the growth and directional guidance of axons during neural development. This interaction helps neurons navigate to their appropriate targets.
- Repulsive Signals: The Slit-ROBO pathway provides repulsive signals for developing axons, ensuring they avoid certain regions and extend towards their destined locations.
- Example: In zebrafish, Slit/Robo interactions are essential for the establishment of the spinal cord's neuronal architecture.
Vascular Development
- Angiogenesis: ROBO receptors, particularly ROBO4, are implicated in endothelial cell behavior during angiogenesis. Slit signaling helps regulate blood vessel development and maintenance.
- Vascular Integrity: Slit-ROBO interactions contribute to maintaining the integrity and function of blood vessels, promoting proper endothelial cell junction formation.
- Example: Slit2 influences endothelial cell migration and tube formation, crucial for developing the vascular system.
Immune Response
- Cell Migration: ROBO receptors are involved in the migration of immune cells. Slit ligands can modulate the movement of leukocytes, influencing their recruitment to sites of inflammation. For example, during embryogenesis, Slit2 can modulate the movement of mesenchymal cells, affecting tissue organization.
- T-cell Development: The Slit-ROBO pathway is also important in guiding T-cell progenitors to the thymus, enabling proper immune system maturation.
Organ Development
- Patterning: Slit and ROBO signaling influence the patterning of various organs during embryonic development. They help in establishing spatial organization in tissues.
- Limb Formation: In limb development, Slit acts to control the growth and positioning of developing structures, assisting in proper limb patterning.
Cancer Biology
- Tumor Suppression: Evidence suggests that Slit-ROBO signaling can act as a tumor suppressor by inhibiting cancer cell migration and invasion. Loss of this pathway may lead to enhanced tumor aggressiveness.
- Metastasis Regulation: Altered Slit-ROBO signaling can affect the migration of cancer cells, potentially influencing the metastatic spread of tumors.
Central Nervous System Functions
- Synaptic Plasticity: Slit-Robo signaling may play roles in synaptic plasticity, a critical process for learning and memory, by influencing how neurons connect and communicate.
Slit ligands and ROBO receptors are fundamental in various physiological processes, notably in development, immune response, and potentially in pathological conditions like cancer. Understanding their mechanisms provides insights into developmental biology, neurobiology, and therapeutic opportunities in disease contexts.
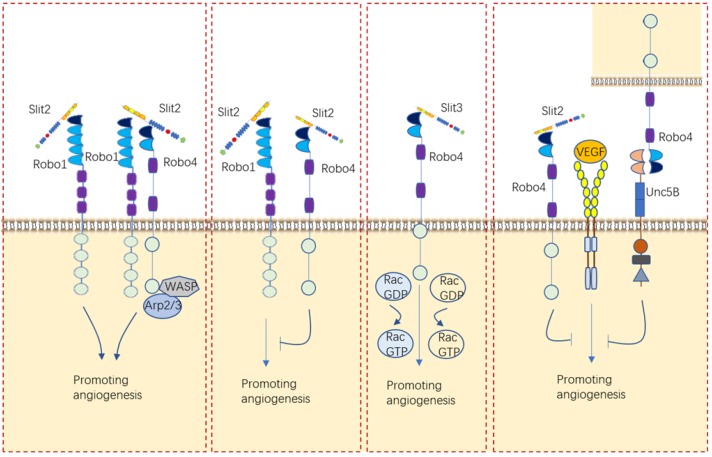 Fig.3 Roles of Slit/Robo in angiogenesis. (Tong M, et al., 2019)
Fig.3 Roles of Slit/Robo in angiogenesis. (Tong M, et al., 2019)
Slits/Robos can alternatively bind with Robo1 or Robo4 or cooperate with VEGF to balance vascular vessel development.
The Impact of Slit Ligands and ROBO Receptors in Disease Pathology
Slit ligands and ROBO receptors play significant roles in various diseases due to their involvement in cellular migration, signaling, and tissue development. Here is a summary of their implications in disease pathology:
Cancer
- Metastasis: The dysregulation of Slit-ROBO signaling is closely linked to cancer metastasis, where abnormal expression or activity of Slit ligands and ROBO receptors can drive cancer cell migration and invasion. Notably, in breast cancer, heightened expression of ROBO1 correlates with increased metastasis, facilitating the spread of cancer to distant sites.
- Tumor Microenvironment: Across various cancers, including glioblastoma (GBM), the interplay between Slit ligands and ROBO receptors significantly influences the tumor microenvironment. This interaction impacts immune responses, tumor vessel function, and the effectiveness of chemotherapy and immunotherapy. In GBM, elevated SLIT2 expression is associated with a poor prognosis, with SLIT2 signaling through ROBO receptors playing a pivotal role in shaping the tumor microenvironment and treatment outcomes.
Neurological Disorders
- Neuropathies: Mutations in genes encoding Slit proteins or ROBO receptors are implicated in certain neuropathies and neurodevelopmental disorders. Disruption of Slit-ROBO signaling can perturb axon guidance, leading to neuronal miswiring and functional impairments. For instance, mutations in the ROBO3 gene are linked to horizontal gaze palsy with progressive scoliosis (HGPPS), a rare neurological condition characterized by impaired eye movement coordination and spinal curvature abnormalities.
- Alzheimer's Disease: Studies suggest that Slit-ROBO signaling pathways play a role in Alzheimer's disease pathology, potentially influencing neuronal connectivity and synaptic plasticity. Disruptions in these pathways may contribute to synaptic dysfunction and neuronal loss in Alzheimer's disease, impacting cognitive function.
Cardiovascular Diseases
- Angiogenesis: Dysregulated Slit-ROBO signaling can influence angiogenesis, a process relevant in cardiovascular diseases such as atherosclerosis and ischemic heart disease. Changes in the expression of Slit ligands or the activity of ROBO receptors may affect blood vessel formation and remodeling.
- Vascular Permeability: Altered Slit-ROBO signaling has been associated with vascular permeability disorders, contributing to conditions like edema and inflammation.
Immune Disorders
- Inflammatory Responses: Slit-ROBO signaling plays a crucial role in immune cell migration and responses. Dysregulation of this pathway can impact inflammatory processes, potentially contributing to autoimmune diseases and chronic inflammation.
- Cancer Immunotherapy: Targeting Slit-ROBO signaling in the tumor microenvironment shows promise in enhancing the efficacy of cancer immunotherapy by modulating immune cell behavior and responses.
Other Diseases
- Wound Healing: Slit-ROBO signaling affects wound healing processes by regulating cell migration and tissue repair mechanisms.
- Organ Dysfunction: Disruptions in Slit-ROBO interactions may contribute to organ dysfunction and developmental abnormalities across different systems.
Understanding the multifaceted roles of Slit ligands and ROBO receptors in diverse diseases underscores their significance as potential therapeutic targets and highlights the need for further research to unravel the complexities of these signaling pathways in disease pathology.
Case Study
Case 1: Basha S, Jin-Smith B, Sun C, Pi L. The SLIT/ROBO pathway in liver fibrosis and cancer. Biomolecules. 2023; 13(5):785.
Liver fibrosis is a common result of chronic liver injuries and can progress to cirrhosis and liver cancer. Recent advancements in liver cancer research have identified signaling pathways involved in disease progression. The SLIT family of proteins, including SLIT1, SLIT2, and SLIT3, interact with ROBO receptors to regulate cell interactions. This pathway plays a role in neural targeting, axon guidance, and neuronal migration in the nervous system. Studies have shown that SLIT and ROBO signaling levels vary in different tumor cells, affecting tumor development and progression. The SLIT and ROBO pathway has been associated with liver fibrosis and cancer development. Research has explored the expression of SLIT and ROBO proteins in normal adult livers, hepatocellular carcinoma, and cholangiocarcinoma. Potential therapeutics targeting this pathway may hold promise for anti-fibrosis and anti-cancer treatments.
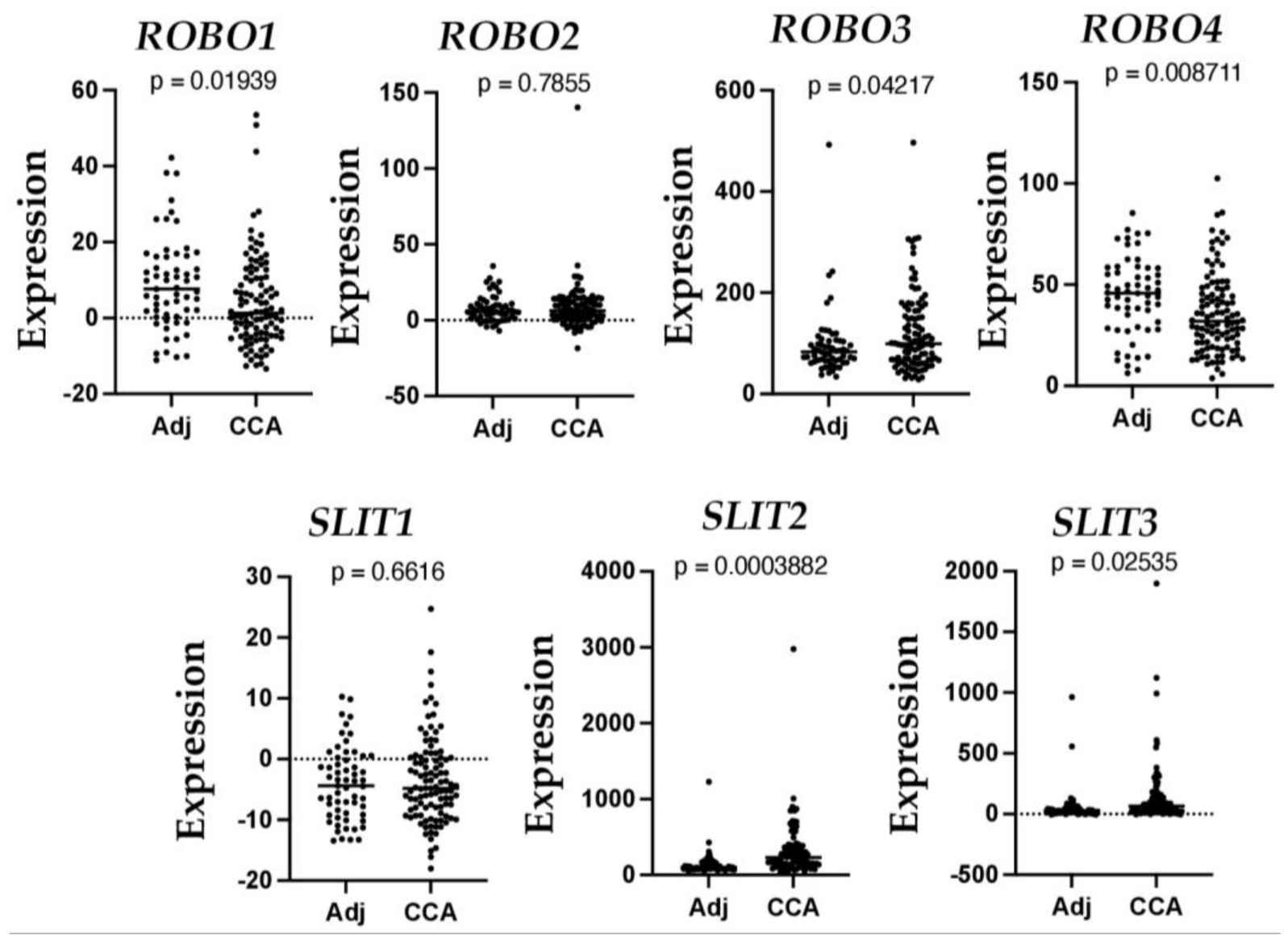 Fig.1 Altered mRNA levels of SLIT and ROBO in human CCA.
Fig.1 Altered mRNA levels of SLIT and ROBO in human CCA.Case 2: Geraldo LH, Xu Y, Jacob L, et al. SLIT2/ROBO signaling in tumor-associated microglia and macrophages drives glioblastoma immunosuppression and vascular dysmorphia. J Clin Invest. 2021;131(16):e141083.
SLIT2, a secreted polypeptide, guides the migration of cells expressing ROBO1 and ROBO2 receptors. The effects of SLIT2/ROBO signaling in gliomas were investigated in this study. In individuals with glioblastoma (GBM), SLIT2 expression levels escalated with malignant progression, correlating with diminished survival rates and immunosuppression. Knockdown of SLIT2 in mouse glioma cells and patient-derived GBM xenografts resulted in reduced tumor growth and, in conjunction with immunotherapy, extended survival periods. Suppression of SLIT2 in tumor cells inhibited macrophage invasion, instigating a cytotoxic gene expression profile that ameliorated tumor vessel function and bolstered the effectiveness of chemotherapy and immunotherapy. Mechanistically, SLIT2 facilitated microglia/macrophage chemotaxis and tumor-supportive polarization through ROBO1 and ROBO2-mediated PI3Kγ activation. Deletion of macrophage Robo1 and Robo2 and systemic delivery of a SLIT2 trap mirrored the effects of SLIT2 knockdown on tumor growth and the tumor microenvironment (TME). This revealed SLIT2 signaling via macrophage ROBOs as a novel regulator of the GBM microenvironment and a potential immunotherapeutic target for brain tumors.
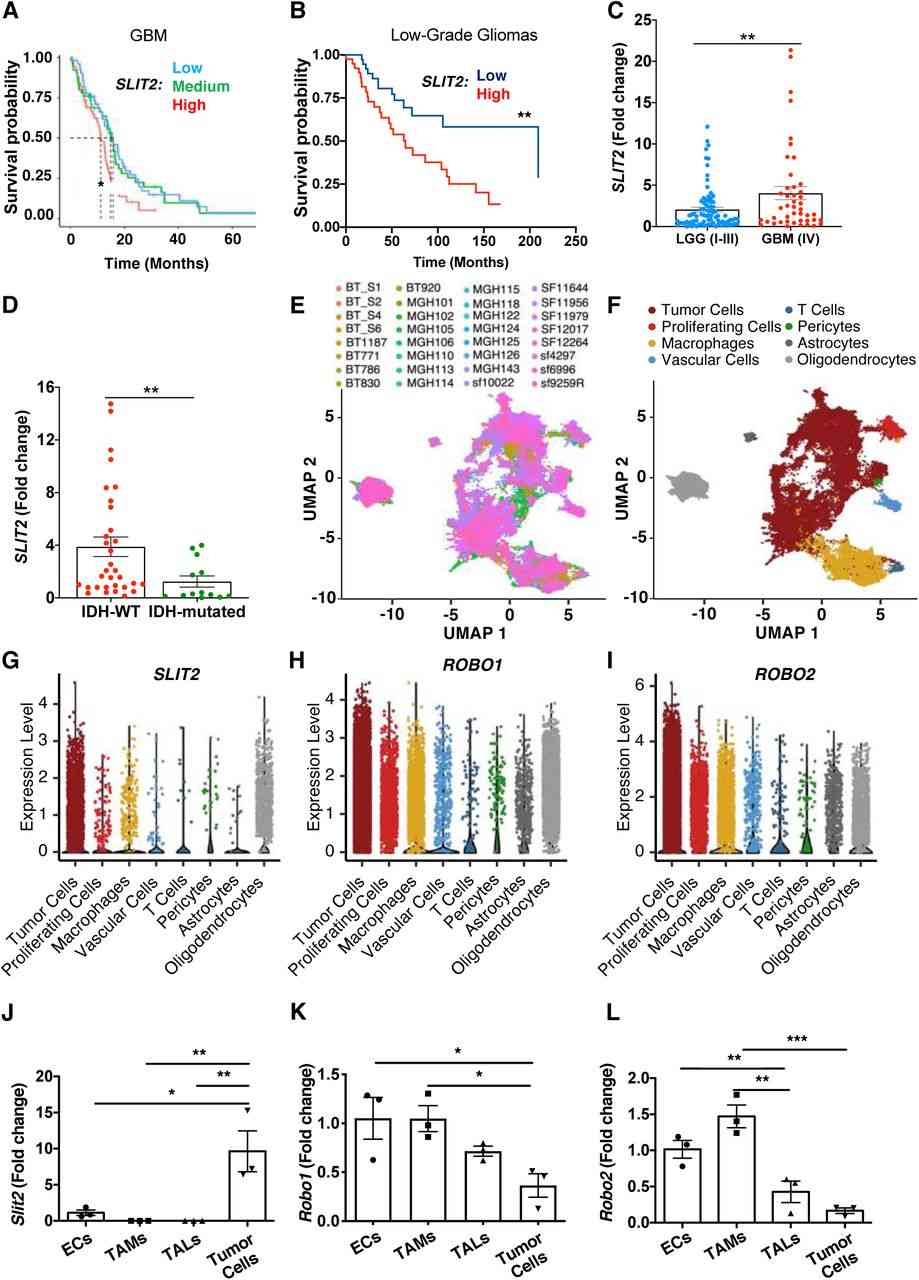 Fig.2 Slit2 expression correlates with glioma aggressiveness and poor patient prognosis.
Fig.2 Slit2 expression correlates with glioma aggressiveness and poor patient prognosis.Related References
- Chédotal A. Slits and their receptors. Adv Exp Med Biol. 2007;621:65-80.
- Gonda Y, Namba T, Hanashima C. Beyond Axon Guidance: Roles of Slit-Robo signaling in neocortical formation. Front Cell Dev Biol. 2020;8:607415.
- Tong M, Jun T, Nie Y, Hao J, Fan D. The role of the Slit/Robo signaling pathway. J Cancer. 2019;10(12):2694-2705.
- Basha S, Jin-Smith B, Sun C, Pi L. The SLIT/ROBO pathway in liver fibrosis and cancer. Biomolecules. 2023;13(5):785.

