What is COMP Protein
The Cartilage Oligomeric Matrix Protein (COMP) is a complex protein that is primarily associated with the extracellular matrix of cartilage. Research has formulated numerous narratives to better understand this protein by examining its structure, function, related diseases, and applications.
COMP protein was first discovered in 1976 during a study on the biochemistry of cartilage in St. Bartholomew's Hospital Medical College, London. However, the full gene encoding for COMP was cloned successfully in 1990.
With the location of the gene locus transpiring at the 19p13.1 on the short arm of chromosome 19, the gene covers an area of about 35 kilobases and comprises of 19 exons. The process of forming mature mRNA demands that exon 1 to 2 should be spliced out, during which they form important structural amino acids necessary for the protein structure of COMP.
The structural elucidation of COMP explicates its existence as a homopentameric protein composed of identical subunits. Each subunit entails a coiled-coil domain generating an oligomerization site, four epidermal growth factor (EGF)-like domains, and a large C-terminal globular domain. This terminal facilitates interaction with other ECM components like collagen and fibronectin.
Function of COMP protein
The pivotal function of COMP is predominantly associated with the mechanical properties of cartilage. REP, an extracellular matrix protein that interacts with both fibrillar collagens and thin filaments, is regulated by COMP. This regulation renders COMP veritably integral in maintaining the integrity of the cartilage extracellular matrix. Additionally, COMP is crucial for angiogenesis, the process of forming new blood vessels, due to its bindings with collagen types I, II, and IX.
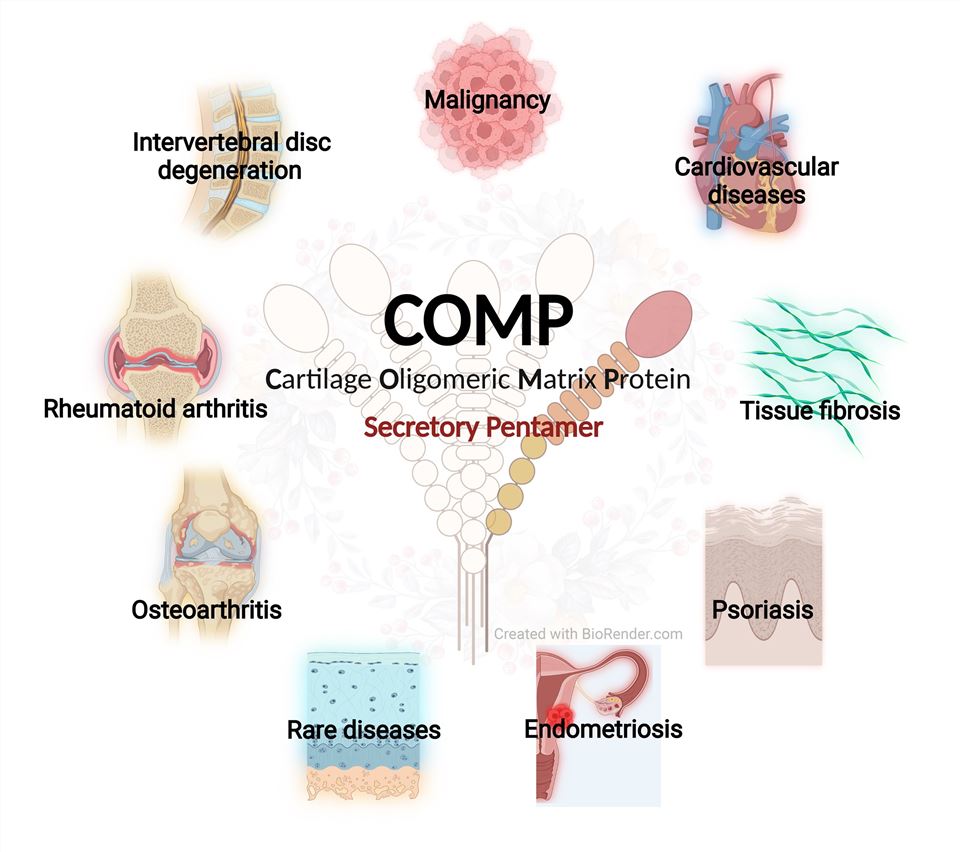
Fig1. COMP Functions (Cui, J., & Zhang, J. 2021)
COMP protein related signal pathway
The COMP protein also plays a vital role in the transforming growth factor-beta (TGF-β) signaling pathway, a cellular mechanism integral for cell proliferation, differentiation, and growth. COMP interacts with TGF-β and enhances TGF-β signaling by facilitating TGF-β latency via binding to latent TGF-β Binding Proteins.
COMP protein related diseases
Insights into COMP-related diseases provide a better understanding of the significance of COMP protein. Its dysfunctions can be clinically associated with two key groups of diseases - Pseudoachondroplasia (PSACH) and Multiple Epiphyseal Dysplasia (MED). These disorders are genetically defined as COMP mutations, resulting in skeletal dysplasia, generally categorized by disproportionate short stature and early onset osteoarthritis. The understanding of COMP-related diseases provides pivotal insights into the molecular pathogenesis of these health conditions, aiding in the development of potential therapeutic strategies.
COMP protein's applications in biomedical
The critical examination of COMP protein is not just limited to the realm of molecular genetics and disease diagnosis, it extends into the domain of biomedical application as well. COMP is used as a biomarker in biochemical assays for the diagnosis and prognosis of diseases like osteoarthritis and rheumatoid arthritis. The elevated levels of COMP can be indicative of cartilage breakdown in these arthritic conditions. Consequently, monitoring these levels could provide important information about disease activity and efficacy of treatment in disease management.
Moreover, the gene therapy approaches using COMP cDNA have also been explored for their potential use in correcting skeletal dysplasia caused by COMP mutations. Likewise, the introduction of COMP in tissue engineering matrices has shown promising results in formulating advanced strategies for cartilage repair and formation.
In conclusion, COMP protein, being a major non-collagenous component of cartilage, plays a significant role in maintaining cartilage integrity and contributing to various biological processes. The understanding of COMP and its association with diseases could lead to effective diagnosis and treatment strategies. Furthermore, leveraging the applications of COMP in the biomedical field can revolutionize the way we address an array of health conditions associated with cartilage and bone. Future research is warranted for more sophisticated and comprehensive comprehension of COMP that can foster broader biomedical implications.
Our Featured Products
| Cat.No. | Product Name | Species | Source (Host) | Tag |
|---|---|---|---|---|
| COMP-11451H | Recombinant Human COMP, GST-tagged | Human | E.coli | GST |
| COMP-26251TH | Recombinant Human COMP, FLAG-tagged | Human | HEK293 | Flag |
| COMP-3183H | Recombinant Human COMP protein, His-tagged | Human | E.coli | His |
| COMP-3659H | Recombinant Human COMP protein, FLAG-tagged | Human | HEK293 | Flag |
| COMP-1879M | Recombinant Mouse COMP Protein, His (Fc)-Avi-tagged | Mouse | HEK293 | His (Fc)-Avi |
| Comp-921M | Recombinant Mouse Comp Protein, MYC/DDK-tagged | Mouse | HEK293T | MYC/DDK |
| COMP-1185R | Recombinant Rat COMP Protein, His (Fc)-Avi-tagged | Rat | HEK293 | His (Fc)-Avi |
Reference
- Cui, J., & Zhang, J. (2021). Cartilage Oligomeric Matrix Protein, Diseases, and Therapeutic Opportunities. International Journal of Molecular Sciences, 23(16), 9253. https://doi.org/10.3390/ijms23169253

