Mouse Protein
🧪 CBLN2-2783M
Source: HEK293
Species: Mouse
Tag: His
Conjugation:
Protein Length: 52-225 aa
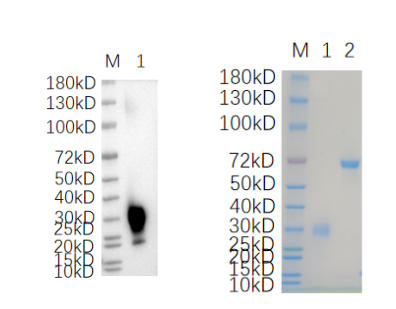
🧪 DOK5-4764M
Source: HEK293
Species: Mouse
Tag: His
Conjugation:
Protein Length: 1-306 aa
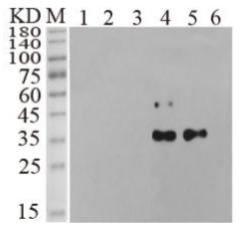
🧪 Egf -634M
Source: Mouse Submaxillary Gland
Species: Mouse
Tag: Non
Conjugation:
Protein Length:

🧪 A2m-367M
Source: Mouse Serum
Species: Mouse
Tag: Non
Conjugation:
Protein Length:
.jpg)

🧪 MBP-6949M
Source: Mouse Central Nervous System
Species: Mouse
Tag: Non
Conjugation:
Protein Length:

🧪 Egf-634M
Source: Mouse Submaxillary Gland
Species: Mouse
Tag: Non
Conjugation:
Protein Length:

🧪 Alb-7992M
Source: Mouse Serum
Species: Mouse
Tag: Non
Conjugation:
Protein Length:
🧪 Ngf-298M
Source: Mouse Submaxillary Gland
Species: Mouse
Tag: Non
Conjugation:
Protein Length:









