PI 3-Kinases (Phosphoinositide 3-Kinases)
Related Symbol Search List
Immunology Background
About PI 3-Kinases (Phosphoinositide 3-Kinases)
Phosphoinositide 3-kinases (PI3Ks), also called phosphatidylinositol 3-kinases, are lipid kinases that play a central role in the regulation of cell cycle, apoptosis, DNA repair, senescence, angiogenesis, cellular metabolism, and motility. They act as intermediate signaling molecules and are most well-known for their roles in the PI3K/AKT/mTOR signaling pathway. PI3Ks transmit signals from the cell surface to the cytoplasm by generating second messengers phosphorylated phosphatidylinositols which in turn activate multiple effector kinase pathways, including BTK, AKT, PKC, NF-kappa-B, and JNK/SAPK pathways, and ultimately result in survival and growth of normal cells.
The PI3K family can be divided into four distinct classes based on primary structure, regulation, and in vitro lipid substrate specificity: class I, class II, class III, and class IV. Of these, class I PI 3-Kinases are the most commonly studied category and include four isoforms: PI3Kα, PI3Kβ, PI3Kγ, and PI3Kδ. They have similar structures and contain a catalytic subunit and one or more regulatory subunits.
PI 3-Kinases activate downstream signaling pathways by phosphorylating the hydroxyl group at the 3-position of phosphatidylinositides (phosphoinositides) to produce the important secondary signaling molecule phosphatidylinositol triphosphate (PIP3).
Mechanism of Action of PI 3-Kinases (Phosphoinositide 3-Kinases)
The mechanism of action of PI 3-Kinases involves multiple steps and signaling molecules, the following are its main mechanisms of action:
- Activation and Phosphorylation
When cells are exposed to external stimuli (e.g., binding of growth factors), PI 3-Kinases are activated and localized to the cell membrane. The activated PI 3-kinases enzyme catalyzes the phosphorylation of the 3-hydroxyl group of the substrate phosphatidylinositol 4,5-bisphosphate (PIP2), generating phosphatidylinositol 3,4,5-trisphosphate (PIP3).
- Effector Protein Binding of PIP3
PIP3 is enriched at the cell membrane and acts as a signaling molecule that recruits and binds several downstream effector proteins (the most important of which is Akt (protein kinase B)).PIP3 recruits Akt to the cell membrane and causes it to be activated by phosphorylation at specific sites.
- Akt Signaling Pathway
Activated Akt phosphorylates multiple target proteins, thereby regulating multiple cellular processes. It regulates cell proliferation, survival, apoptosis, and cytoskeletal reorganization by phosphorylating and regulating cell cycle regulatory proteins, apoptosis-related proteins, cytoskeletal proteins, and transcription factors.
- mTOR Signaling Pathway
The activated PI 3-Kinases/Akt signaling pathway also regulates cell metabolism and growth by inhibiting the negative regulatory effects of the mTOR signaling pathway. mTOR is an important protein kinase involved in the regulation of cellular protein synthesis, lipid metabolism, and cell proliferation.
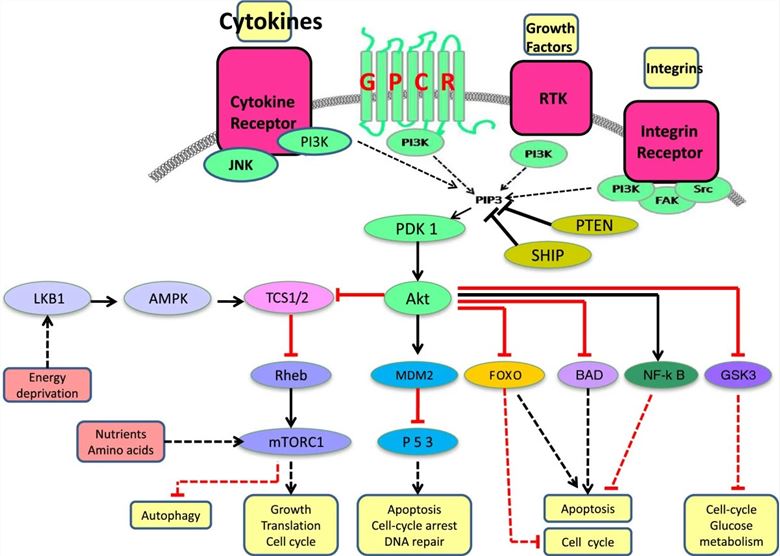 Fig.2 Schematic representation of the PI3K signaling pathway. (Akinleye A, et al., 2013)
Fig.2 Schematic representation of the PI3K signaling pathway. (Akinleye A, et al., 2013)
PI3Ks represent a family of lipid kinases that lie upstream of complex, intricate, interconnected intracellular signaling networks. They transduce signals from transmembrane receptors such as RTKs and G-protein coupled receptors (GPCRs) to the cytoplasm through production of phosphorylated lipids to regulate key cellular processes including proliferation, differentiation, senescence, motility, and survival. Four major extracellular signals, growth factors, cytokines, hormones/chemokines, and integrins, activate PI3K, which transmit the signals through appropriate pathways to control diverse cellular processes, including cell cycle, apoptosis, DNA repair, senescence, angiogenesis, cellular metabolism, autophagy, and motility. The multiple effector kinase pathways activated by PI3K are highlighted in the figure.
Functions of PI 3-Kinases (Phosphoinositide 3-Kinases)
PI 3-Kinases play a variety of functions in the cell and are involved in several biological processes. The following are the main functions of PI 3-Kinases:
- Cell Proliferation and Growth
PI 3-Kinases are involved in the regulation of cell proliferation and growth processes. Activated PI 3-Kinases produce the secondary signaling molecule PIP3 that recruits and activates Akt protein kinase, which in turn affects cell cycle regulation, cell division, and cell growth. This is essential for maintaining normal cell proliferation and development.
- Cell Survival and Apoptosis
PI 3-Kinases have an impact on cell survival by regulating cell survival and apoptotic processes. Activated PI 3-Kinases/Akt signaling pathway inhibits the activity of apoptosis-associated proteins, thereby promoting cell survival. This has an important role in processes such as cell development, tissue repair, and immune response.
- Cell migration and Invasion
PI 3-Kinases are involved in the regulation of cell migration and invasion capacity. They affect cell mobility and invasiveness by activating downstream effector proteins, such as Akt and Rho family GTPases, which regulate cytoskeletal reorganization, cell-matrix interactions, and intercellular junctions. This is critical for physiological and pathological processes such as embryonic development, cancer metastasis, and inflammatory responses.
- Metabolic Regulation
PI 3-Kinases are involved in the regulation of cellular metabolic processes. The activated PI 3-Kinases/Akt/mTOR signaling pathway regulates glucose uptake, lipid synthesis, protein synthesis, and energy metabolism. This is important for maintaining the metabolic homeostasis of cells and adapting to the external environment.
- Immunomodulation
PI 3-Kinases play an important regulatory role in immune cells. They are involved in the regulation of immune cell proliferation, migration, activation, and cell death, etc. Dysregulation of the PI 3-Kinases signaling pathway is closely related to the occurrence and development of immune system-related diseases.
Available Resources for PI 3-Kinases (Phosphoinositide 3-Kinases)
PI 3-Kinases are essential components of cell signaling networks and play important roles in cell growth, differentiation, and survival. Due to their key roles in a variety of diseases such as cancer, inflammation, and metabolic disorders, PI 3-Kinases have become important targets for drug discovery. Creative BioMart offers a variety of PI 3-Kinases-related research products, such as recombinant proteins, cell and tissue lysates, and protein pre-coupled magnetic beads, as well as customizable services and other resources to support your research in the field of PI 3-Kinases or PI 3-Kinases/Akt/mTOR signaling pathway. The following PI 3-Kinases are displayed, click to view all related molecules/targets and research reagents. For further information or to purchase products, please contact us. We are committed to providing the highest quality resources and support for your research to help you succeed.
Reference:
- Akinleye A, Avvaru P, Furqan M, Song Y, Liu D. Phosphatidylinositol 3-kinase (PI3K) inhibitors as cancer therapeutics. J Hematol Oncol. 2013;6(1):88. Published 2013 Nov 22.

