PKC Isoforms
Related Symbol Search List
Immunology Background
About PKC Isoforms
Protein kinase C (PKC) is a calcium-dependent, lipid cofactor-sensitive protein kinase belonging to the serine/threonine family of protein kinases, which plays an essential role in cellular signaling pathways.PKC isoforms are different forms of PKC proteins, each with different characteristics and functions. These are 11 isoforms encoded by 9 genes and are divided into 3 categories - classical (cPKCs-α, βI, βII, γ), novel (nPKCs-δ, ϵ, η, θ), and atypical (aPKCs-ζ, ι/λ).
Conventional (cPKCs) - require DAG, Ca2+, and phospholipid for activation
- PKC-α (PRKCA)
- PKC-β1 (PRKCB)
- PKC-β2 (PRKCB)
- PKC-γ (PRKCG)
Novel (nPKC) - require DAG but not Ca2+ for activation
- PKC-δ (PRKCD)
- PKC-ε (PRKCE)
- PKC-η (PRKCH)
- PKC-θ (PRKCQ)
Atypical (aPKCs) - require neither Ca2+ nor DAG for activation (require phosphatidyl serine)
- PKC-ι (PRKCI)
- PKC-ζ (PRKCZ)
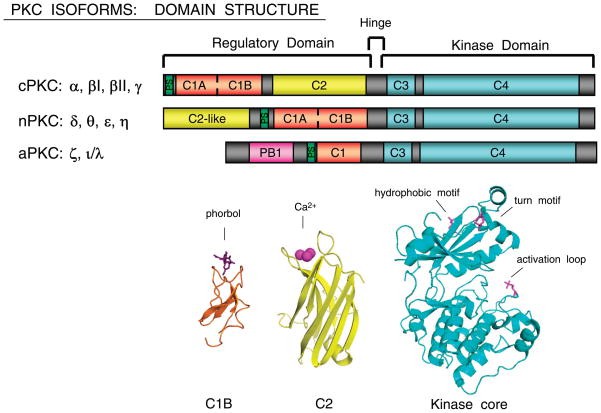 Fig.1 Domain structure of protein kinase C (PKC) isoforms. (Steinberg SF, 2008)
Fig.1 Domain structure of protein kinase C (PKC) isoforms. (Steinberg SF, 2008)
Mechanism of Action of PKC Isoforms
PKC isoforms are activated by different mechanisms, primarily involving lipid second messengers and calcium ions.
- The conventional PKCs (α, βI, βII, and γ) require both diacylglycerol (DAG) and calcium ions for activation. They possess a C1 domain that interacts with DAG, allowing membrane translocation and subsequent activation.
- Novel PKCs (δ, ε, η, and θ) are activated by DAG but do not require calcium ions. They contain a C1 domain that selectively binds DAG.
- Atypical PKCs (ζ and ι/λ) do not require DAG or calcium ions for activation. Instead, they are regulated by protein-protein interactions and phosphorylation events.
Functions of PKC Isoforms
According to existing studies, PKC isoforms play different roles in signaling transduction and cellular processes, performing complex regulatory roles.
- Conventional PKCs (cPKCs) are involved in cell proliferation, differentiation, and survival.
Signaling Transduction: cPKC phosphorylates various substrates, including transcription factors, receptor proteins, and cytoskeletal proteins, to regulate cell growth, differentiation, and survival.
Cellular Processes: cPKCs play roles in immune response, hormone secretion, synaptic plasticity, and cardiac function.
- Novel PKCs (nPKCs) regulate cellular processes related to cell growth, migration, and apoptosis.
Signaling Transduction: nPKCs phosphorylate specific substrates involved in cell adhesion, cytoskeletal rearrangement, and apoptosis regulation.
Cellular Processes: nPKCs contribute to immune responses, neuronal development, and cancer progression.
- Atypical PKCs (aPKCs) are involved in cell polarity, migration, and insulin signaling.
Signaling Transduction: aPKCs regulate protein-protein interactions and phosphorylation events critical for cell polarity determination and migration. migration.
Cellular Processes: aPKCs play roles in epithelial cell polarization, neuronal development, and insulin-stimulated glucose uptake.
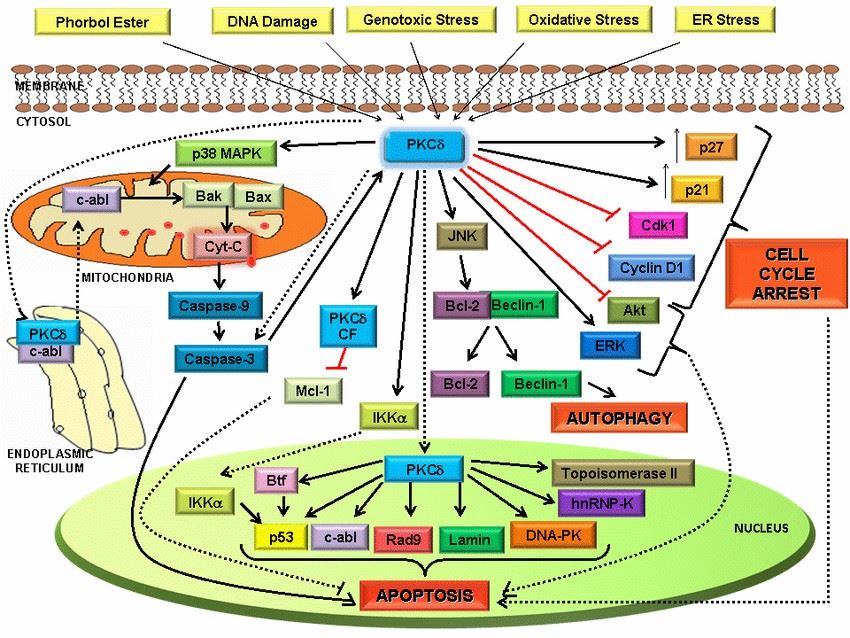 Fig.2 PKC as a tumor suppressor. (Basu A, et al., 2010)
Fig.2 PKC as a tumor suppressor. (Basu A, et al., 2010)
Available Resources for PKC Isoforms
PKC isoforms play a critical role in cell signaling and regulation. Traditional, novel and atypical isoforms exhibit different mechanisms and functions that affect a variety of cellular processes, and Creative BioMart offers a wide selection of products to help researchers study the functions and mechanisms of PKC isoforms in a variety of biological studies, including recombinant proteins, cell and tissue lysates, and protein pre-coupled magnetic beads, as well as customizable services and other resources. The following PKC isoforms are displayed, click to view all related molecules/targets and research reagents. For further information or to purchase products, please contact us. We are committed to providing the highest quality resources and support for your research to help you succeed.
References:
- Steinberg SF. Structural basis of protein kinase C isoform function. Physiol Rev. 2008;88(4):1341-1378
- Üsküdar Zeynep Kamil Vocational, Highs T A .The molecular functions of protein kinase C (PKC) isoforms[J]. November 30, 2018
- Basu A, Pal D. Two faces of protein kinase Cδ: the contrasting roles of PKCδ in cell survival and cell death. Scientific World Journal. 2010;10:2272-2284.

