STAT
Related Symbol Search List
Immunology Background
About STAT
STAT (signal transduction and transcription activation) proteins a family of potential cytoplasmic transcription factors, originally described as key mediators of interferon signaling, are now recognized as signaling factors downstream of many cytokines, growth factors, and hormones. They are responsible for a range of cellular activities, including the regulation of growth, survival, differentiation, motility, and immune responses.
Seven mammalian STAT family members have been identified: STAT1, STAT2, STAT3, STAT4, STAT5 (STAT5A and STAT5B), and STAT6. All seven STAT proteins share a common structural structure. proteins share a common structural motif consisting of an N-terminal domain followed by a coiled-coil, DNA-binding domain, linker, Src homology 2 (SH2), and a C-terminal transcriptional domain. Much research has focused on elucidating the roles each of these domains plays in regulating different STAT isoforms. Much research has focused on elucidating the roles each of these domains plays in regulating different STAT isoforms. Both the N-terminal and SH2 domains mediate homo or heterodimer formation, while the coiled-coil domain functions partially as a nuclear localization signal (NLS). localization signal (NLS). Transcriptional activity and DNA association are determined by the transactivation and DNA-binding domains, respectively.
Understanding the biology and function of STAT proteins is important in various fields such as immunology, cancer research, and developmental biology.
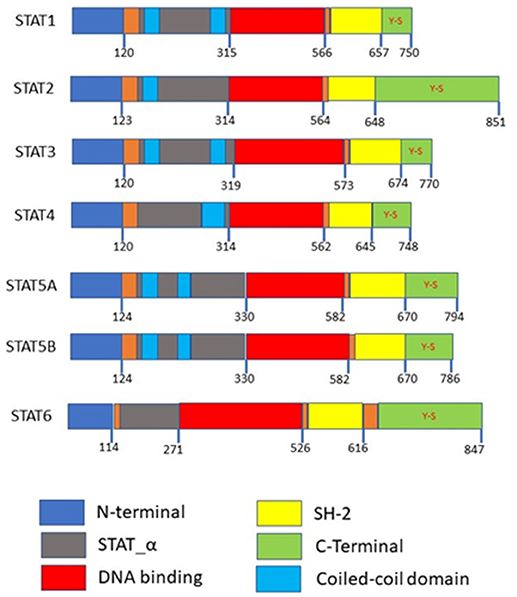 Fig.1 Domain structures of the STAT family members. N-terminal is required for protein interactions, SH-2 domains are required for dimerization, both the N-terminal and SH-2 domains mediated homo or heterodimer formation. Coiled-coil domains are required as a nuclear localization signal for activation. Y-S within the C-terminal are the phosphorylation sites. (Loh C Y, et al., 2019)
Fig.1 Domain structures of the STAT family members. N-terminal is required for protein interactions, SH-2 domains are required for dimerization, both the N-terminal and SH-2 domains mediated homo or heterodimer formation. Coiled-coil domains are required as a nuclear localization signal for activation. Y-S within the C-terminal are the phosphorylation sites. (Loh C Y, et al., 2019)
Mechanism of Action of STAT
The activation of STAT proteins is triggered by the activation of specific cell surface receptors, such as cytokine receptors and growth factor receptors. Upon ligand binding, these receptors activate associated Janus kinases (Jaks), which phosphorylate specific tyrosine residues on the cytoplasmic domain of the receptors. This creates docking sites for STAT proteins, which are recruited to the receptor complex. The Jaks then phosphorylate the recruited STAT proteins, leading to their dimerization. Dimerized STAT proteins translocate to the nucleus, where they bind to specific DNA sequences known as STAT response elements (SREs), resulting in the regulation of target gene expression.
Functions of STAT
STAT proteins have diverse functions in different cellular processes. Here are some examples of their functions:
- Immune Response
STAT proteins play a crucial role in immune responses. They regulate the production of cytokines, chemokines, and other immune-related molecules, and also participate in the differentiation and activation of immune cells. For example, STAT3 is involved in the differentiation of Th17 cells, while STAT5 regulates the development and function of regulatory T cells.
- Development and Differentiation
STAT proteins are involved in various aspects of development and differentiation processes. They participate in cell fate determination, tissue-specific gene expression, and organogenesis. For instance, STAT1 and STAT3 are important for the differentiation and function of various cell types during embryonic development.
- Inflammation
STAT proteins are implicated in the regulation of inflammatory responses. They mediate the signaling of pro-inflammatory cytokines, such as interleukins (ILs) and interferons (IFNs), leading to the production of inflammatory mediators and the modulation of immune cell function.
- Cancer
Dysregulation of STAT signaling has been observed in various types of cancers. Aberrant activation of STAT proteins can promote cell survival, proliferation, and invasion, contributing to tumor development and progression. Therefore, STAT proteins have emerged as potential targets for cancer therapy.
Available Resources for STAT
Understanding the role of STAT proteins in cellular signaling is crucial for unraveling the complexities of various biological processes and developing targeted therapies. At Creative BioMart, we offer a comprehensive range of research tools and services to support studies related to STAT, including recombinant proteins, antibodies, cell & tissue lysates, protein pre-coupled magnetic beads, and custom assay development. The following STATs are displayed, click to view all related molecules/targets and research reagents. Please feel free to contact us with any questions or requests.
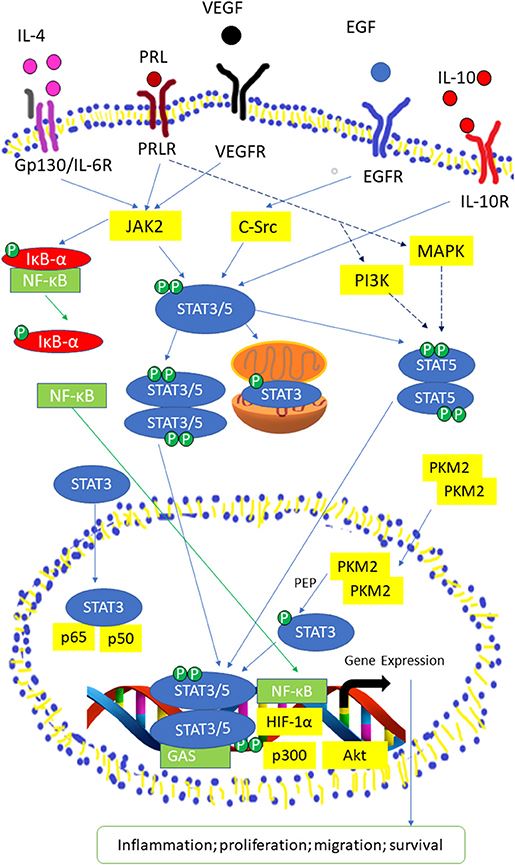 Fig.2 Activated STAT3/5 signaling pathway by IL-6 receptor, Prolactin (PRL) receptor, VEGF Receptor, EGF Receptor, and IL-10 receptor. (Loh C Y, et al., 2019)
Fig.2 Activated STAT3/5 signaling pathway by IL-6 receptor, Prolactin (PRL) receptor, VEGF Receptor, EGF Receptor, and IL-10 receptor. (Loh C Y, et al., 2019)
References:
- Loh C Y, Arya A, Naema A F, et al. signal transduction and transcription activation (STATs) proteins in cancer and inflammation: functions and therapeutic implication[J]. Frontiers in oncology, 2019, 9: 48.
- Li Y J, Zhang C, Martincuks A, et al. STAT proteins in cancer: orchestration of metabolism[J]. Nature Reviews Cancer, 2023, 23(3): 115-134.
- Turkson J, Jove R. STAT proteins: novel molecular targets for cancer drug discovery[J]. Oncogene, 2000, 19(56): 6613-6626.

