vWF
-
Official Full Name
von Willebrand factor -
Overview
The glycoprotein encoded by this gene functions as both an antihemophilic factor carrier and a platelet-vessel wall mediator in the blood coagulation system. It is crucial to the hemostasis process. Mutations in this gene or deficiencies in this protein result in von Willebrands disease. An unprocessed pseudogene has been found on chromosome 22. [provided by RefSeq, Jul 2008] -
Synonyms
VWF;von Willebrand factor;VWD;F8VWF;coagulation factor VIII VWF
Recombinant Proteins
- Mouse
- Human
- Pig
- Dog
- Rhesus macaque
- Rat
- Zebrafish
- Bovine
- E.coli
- CHO
- HEK293
- Sf9 Cells
- Mammalian Cells
- Human Plasma
- Yeast
- Baculovirus
- Mammalian cells
- His
- Avi
- Fc
- Non
- Flag
- Myc
Background
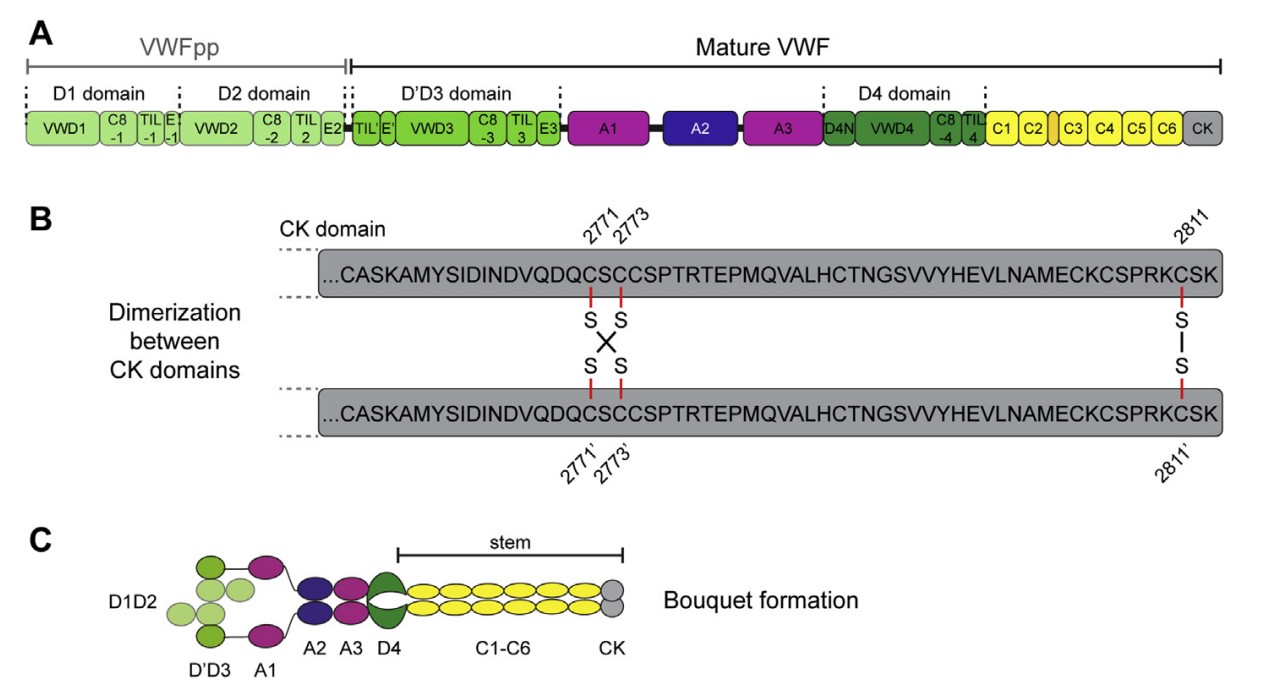
Fig1. VWF domain structure. (A) Schematic representation of VWF's domain structure, (B) dimerizing disulfide bonds between the CK domains of two VWF monomers and (C) dimeric bouquet formation. (Achim Löf, 2017)
What is VWF protein?
VWF (von Willebrand factor) gene is a protein coding gene which situated on the short arm of chromosome 12 at locus 12p13. This gene encodes a glycoprotein involved in hemostasis. The encoded preproprotein is proteolytically processed following assembly into large multimeric complexes. These complexes function in the adhesion of platelets to sites of vascular injury and the transport of various proteins in the blood. The VWF protein is consisted of 2813 amino acids and its molecular mass is approximately 309.3 kDa.
What is the function of VWF protein?
This protein is important in the maintenance of hemostasis, it promotes adhesion of platelets to the sites of vascular injury by forming a molecular bridge between sub-endothelial collagen matrix and platelet-surface receptor complex GPIb-IX-V. Also it acts as a chaperone for coagulation factor VIII, delivering it to the site of injury, stabilizing its heterodimeric structure and protecting it from premature clearance from plasma.
VWF Related Signaling Pathway
VWF is a glycoprotein associated with blood clotting disorders and plays a role in the clotting process. VWF binds to the GPIb-IX-V complex on platelets and promotes platelet adhesion and agglutination, thus initiating the signaling pathway for thrombosis. It can also bind to platelet membrane glycoprotein (GP) complexes and subcutaneous collagen, and this interaction mediates platelet adhesion at the site of vascular injury. It is also involved in reticulum formation and thrombosis.
VWF Related Diseases
VWF protein abnormalities can cause a variety of diseases, von Willebrand disease (VWD) is a group of hereditary bleeding disorders that can be divided into three types based on their severity and the degree of functional deficiency of VWF. When VWF function may also be affected, resulting in bleeding tendencies that can cause thrombocytopenic purpura (ITP). It can also cause von Willebrand Disease Type A. When the VWF protein is abnormal, patients may experience symptoms such as easy bruising and difficulty in hemostasis.
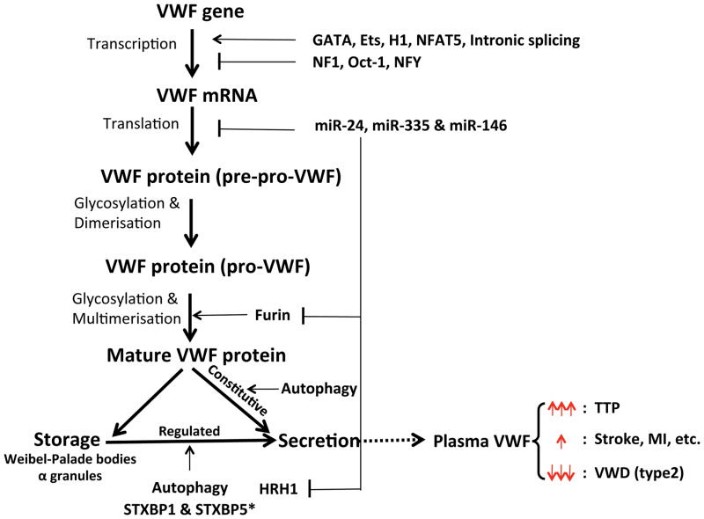
Fig2. Regulation of VWF expression & secretion and diseases. (Yaozu Xiang, 2016)
Bioapplications of VWF
In patients with von Willebrand disease, supplementation of exogenous VWF proteins or coagulation factors may be required to maintain normal clotting function. Because VWD is a genetic disorder, VWF genetic testing and counseling are important for genetic risk assessment and family planning in patients with familial VWD. Intraoperative or daily monitoring of the protein level to avoid the risk of bleeding.
Case Study
Case study 1: Jirawat Khanongnoi, 2018
Snake venom-metalloproteinases (SVMPs) are the primary factors that disturb hemostasis and cause hemorrhage in the venomous snake bitten subjects. Kaouthiagin is a unique SVMP that binds and cleaves von Willebrand factor (vWF) at a specific peptide bond leading to inhibition of platelet aggregation, which enhances the hemorrhage. Kaouthiagin is a low abundant venom component of Thai cobra (Naja kaouthia); thus, most horse-derived antivenins used for cobra bite treatment do not contain adequate anti-kaouthiagin. This study aimed to produce human single-chain antibody variable fragments (HuscFvs) that bind to and interfere with kaouthiagin activity for further clinical use.
Kaouthiagin was purified from N. kaouthia-holovenom by a single-step gel-filtration chromatography. The selected phages were used to infect Escherichia coli bacteria. Soluble HuscFvs expressed by three phage-transformed-E. coli clones interfered with cobra kaouthiagin binding to human vWF. omputerized simulation indicated that HuscFv of two phage-transformed E. coli clones formed contact interface with kaouthiagin residues at or near catalytic site and effectively inhibited fibrinogenolytic activity of the kaouthiagin.

Fig1. Western blotting patterns of the SDS-PAGE-separated fractions 44–56 probed with 1 μg/mL of recombinant human von Willebrand factor (vWF).

Case study 2: Yingxue Qi, 2018
Platelet glycoprotein Ibα (GPIbα) extracellular domain, which is part of the receptor complex GPIb-IX-V, plays an important role in tumor metastasis. However, the mechanism through which GPIbα participates in the metastatic process remains unclear. The researchers established a series of screening models and obtained rat anti-mouse GPIbα monoclonal antibodies (mAb) 1D12 and 2B4 that demonstrated potential value in suppressing cancer metastasis. 1D12 and 2B4 affected the von Willebrand factor (vWF)-GPIbα interaction via binding to GPIbα aa 41-50 and aa 277-290 respectively, which markedly inhibited the interaction among platelets, tumor cells, and endothelial cells in vitro, and reduced the mean number of surface nodules in the experimental and spontaneous metastasis models in vivo.
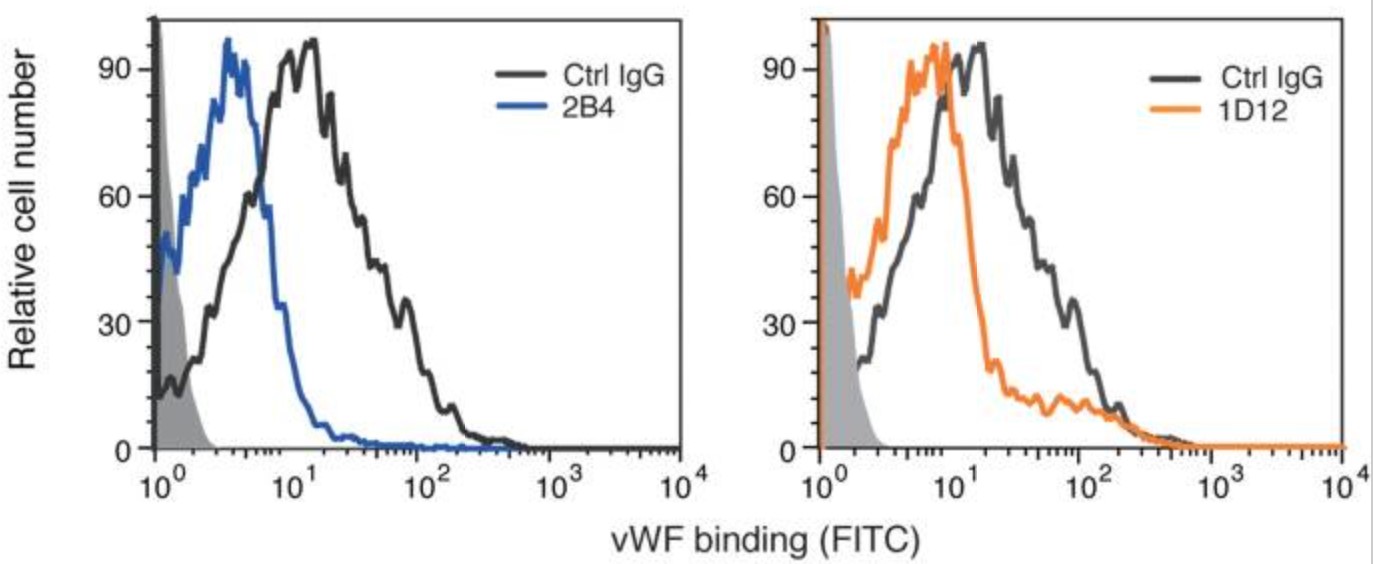
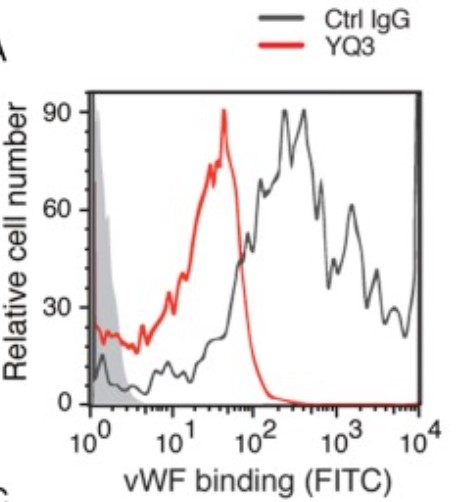
Fig3. The vWF binding was inhibited by 2B4 and 1D12 and detected by flow cytometry. Binding of vWF was detected with FITC-conjugated mouse vWF IgG by flow cytometry and quantitated by mean fluorescence intensity.
Quality Guarantee
High Purity
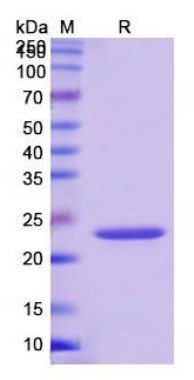
Fig1. SDS-PAGE (VWF-6569H) (PROTOCOL for western blot)
.
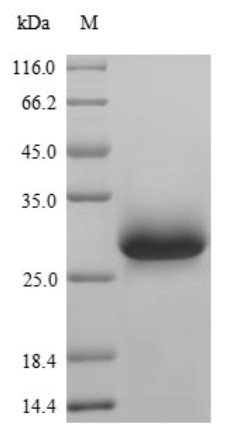
Fig2. SDS-PAGE (VWF-2463M) (PROTOCOL for western blot)
Involved Pathway
vWF involved in several pathways and played different roles in them. We selected most pathways vWF participated on our site, such as PIK-Akt signaling pathway,Focal adhesion,ECM-receptor interaction, which may be useful for your reference. Also, other proteins which involved in the same pathway with vWF were listed below. Creative BioMart supplied nearly all the proteins listed, you can search them on our site.
| Pathway Name | Pathway Related Protein |
|---|---|
| Platelet activation | PIK3CB,ARHGAP35,VAMP8,PIK3CG,GUCY1A3,PTGIR,PIK3R5,PRKACG,SYK,SRC |
| Focal adhesion | CAV1,THBS4,VASPA,THBS3A,VEGFC,ITGAV,CDC42L2,FYN,FLT4,PDPK1B |
| Complement and coagulation cascades | GM5077,F2R,C7,CR1,C1QB,SERPINC1,C8G,F2,F5,C4A |
| ECM-receptor interaction | Itga10&Itgb1,ITGB8,COL1A2,COL3A1,COL6A2,COL4A5,COL5A2,HSPG2,TNW,COL5A1 |
| PIK-Akt signaling pathway | COL4A1,ITGB6,IKBKB,TNR,GNB3,PPP2R5A,PIK3CG,ITGA3,THBS3,Fgf15 |
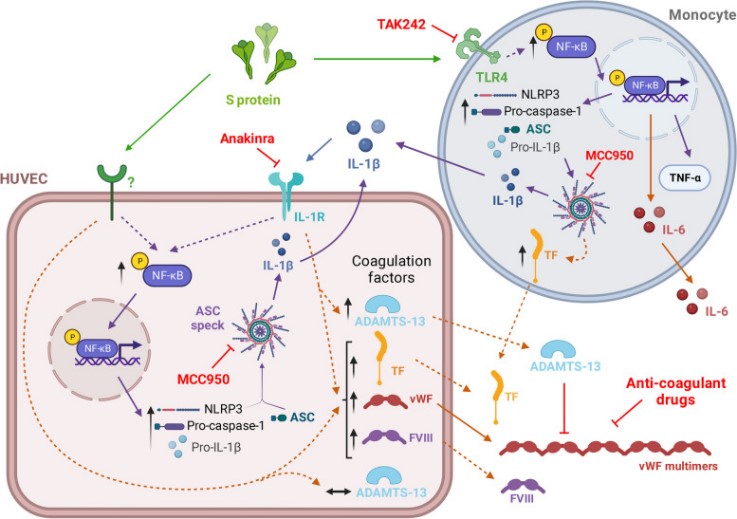
Fig1. Diagram of cellular pathways activated by SARS-CoV-2 S protein and the targets for pharmacological interference. (Alicia Villacampa, 2024)
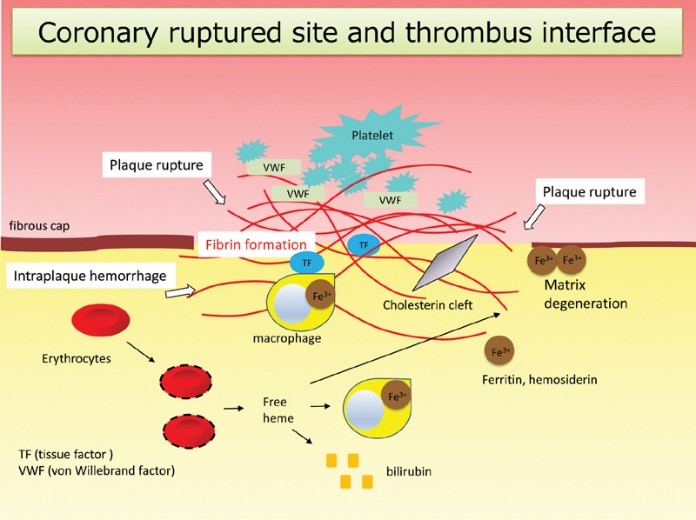
Fig2. Coronary ruptured site and thrombus interface. (Atsushi Yamashita, 2021)
Protein Function
vWF has several biochemical functions, for example, chaperone binding,collagen binding,glycoprotein binding. Some of the functions are cooperated with other proteins, some of the functions could acted by vWF itself. We selected most functions vWF had, and list some proteins which have the same functions with vWF. You can find most of the proteins on our site.
| Function | Related Protein |
|---|---|
| collagen binding | Itga10&Itgb1,GP5,PDGFB,MMP13,ANTXR1,ACHE,LRRC15,CTSL1,Pdgfa&Pdgfb,SERPINH1B |
| integrin binding | FN1A,FN1,LAMB1,ACTN1,ECM2,TSPAN4,IGF1,ITGB1BP1,CTGFB,ITGB5 |
| protein N-terminus binding | MAP2K1,TP53,YWHAQ,CHMP6,RPA2,UNC13A,SLC6A3,PPP1CC,PIAS3,NPAT |
| immunoglobulin binding | cgr2b,HRG |
| protein homodimerization activity | PFKM,TLR13,NFS1,ADAM10,HNF1B,NLRC4,CD247,PDCD6,RCHY1,MAP3K10 |
| chaperone binding | AHSA1,DNAJA2,HSPA5,DNAJB4,ATP1A3,TSC1,SLC25A17,TBCD,BAG3,DNAJA2L |
| identical protein binding | PPP1R13L,LRRK2,CLDN6,DACT3,DESI1,MCIN,UGP2,ALDH4A1,KCNRG,PARK7 |
| protein binding | GLE1,DCAF8,C9orf9,LRRC6,SNAP47,POLD3,FBXO33,COX4I1,PPFIA4,LANCL1 |
| glycoprotein binding | TGFB1,THBS1,Serpina1b,SERPINA1E,BBC3,TFRC,IGF2R,LACRT,CDH1,FLNA |
Interacting Protein
vWF has direct interactions with proteins and molecules. Those interactions were detected by several methods such as yeast two hybrid, co-IP, pull-down and so on. We selected proteins and molecules interacted with vWF here. Most of them are supplied by our site. Hope this information will be useful for your research of vWF.
GP1BA;ADAMTS13
Resources
Research Area
Related Services
Related Products
References
- Timm, A; Hillarp, A; et al. Comparison of automated von Willebrand factor activity assays. THROMBOSIS RESEARCH 135:684-691(2015).
- Tomlinson, RE; Silva, MJ; et al. HIF-1 alpha regulates bone formation after osteogenic mechanical loading. BONE 73:98-104(2015).




