DNA Damage and Repair
Related Symbol Search List
- BABAM1
- Bcl-2
- BRCA1
- Catalase
- CDC25A
- CDC25C
- CDC42
- CDKN1A
- CDKN2B
- Chk1
- CHEK2
- ERCC1
- FANCA
- FTO
- MEN1
- MSH2
- MYC
- PRKDC
- PSME3
- RBBP4
- SOD1
- TNFRSF10D
- TRAILR4
- TOPBP1
- p53
- UBE2F
- UBC13
- UBE2N
- UBE2T
- UBE2V1
- UBE2V2
- XPA
Immunology Background
About DNA Damage and Repair
DNA damage and repair is an important biological process in living organisms, which involves damage to DNA molecules that occur within the cell and the mechanisms within the organism that repair these damages. DNA molecules can be damaged for a variety of reasons, including natural environmental factors (e.g., ultraviolet rays, radiation), chemicals, and metabolites produced within the cell. If unrepaired, these damages can lead to mutations in the DNA sequence, or cell death, and may even cause diseases, including cancer.
To combat these potential threats, organisms have evolved a variety of efficient DNA repair mechanisms to repair different types of DNA damage. These repair mechanisms include but are not limited to, base excision repair (BER), nucleotide excision repair (NER), mismatch repair (MMR), homologous recombination (HR), and non-homologous end joining (NHEJ).
The synergistic effect of these mechanisms ensures that DNA damage can be repaired in a timely and effective manner, thus maintaining the genetic stability and viability of cells. The study of DNA damage and repair not only contributes to an in-depth understanding of the basic biological processes in the cells of living organisms but also has important implications for human health and disease treatment.
Functions and Significance of DNA Damage and Repair
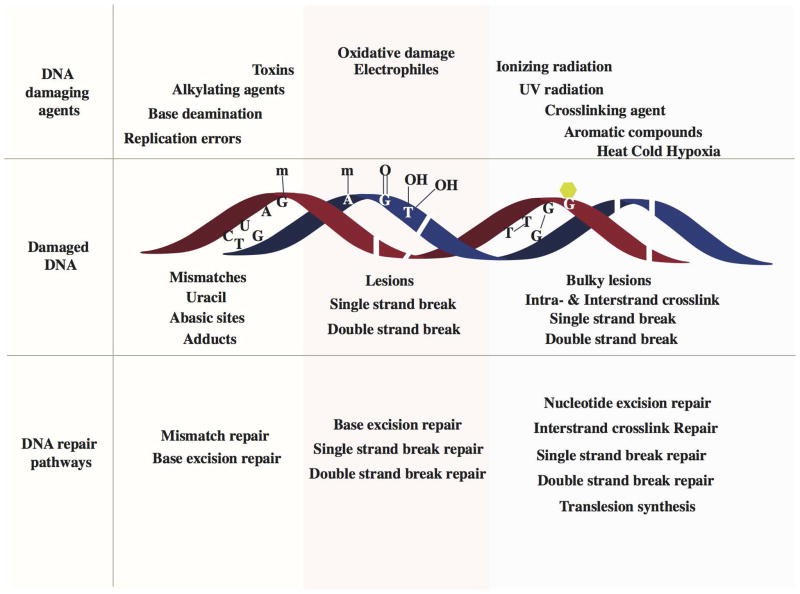 Fig.1 Schematic of various DNA damage-induced DNA repair pathways. (Chatterjee N, et al., 2017)
Fig.1 Schematic of various DNA damage-induced DNA repair pathways. (Chatterjee N, et al., 2017)
A variety of DNA damaging agents can induce DNA damage, which becomes substrate for specific DNA repair pathways. Upper panel shows representative DNA damaging agents: errors from replication, spontaneous base deamination, alkylating agents, toxins, oxidative agents, ionizing radiation, UV radiation, crosslinking agents, aromatic compounds and environmental agents such as heat, cold and hypoxia. Middle panel represents different kinds of damaged DNA: base mismatches (C:T), uracil from deamination of cytosine, an abasic site from the loss of a base from one DNA strand, methylated guanine, methylated adenine, 8-oxo-G lesion, thymine glycols, single strand breaks, double strand breaks, intrastrand cyclobutane thymine dimers and interstrand guanine crosslinks. The lower panel lists the specific DNA repair pathways that are instigated to repair DNA damages: mismatch repair corrects replication errors and other base mismatches; base excision repair removes base adducts, uracil, abasic sites and oxidative lesions; single strand break repair pathways repairs single stranded breaks in the DNA backbone; double strand break repair pathway repair double strand breaks; nucleotide excision repair removes bulky lesions and intrastrand crosslinks; interstrand crosslink repair removes interstrand linkages and translesion synthesis bypasses intrastrand crosslinks and bulky lesions.
Functions and Significance of DNA Damage and Repair
- Maintenance of Genomic Integrity
At the core of DNA damage and repair lies the solemn duty to maintain the sanctity of the genetic material. By diligently detecting and rectifying various forms of DNA damage, these mechanisms shield the genome from the pernicious consequences of mutations. In doing so, they fortify the foundation upon which life's intricate complexities are built.
- Prevention of Disease and Cancer
The consequences of unrepaired DNA damage can be dire, manifesting as genomic instability and providing fertile ground for the inception of diseases, including cancer. DNA repair pathways serve as vigilant guardians, warding off the insidious threats that lurk within the cellular landscape. By faithfully executing their duties, they bestow upon us the gift of protection, mitigating the risk of diseases that haunt humanity.
- Response to DNA-Damaging Agents
The resilience of DNA damage and repair mechanisms is put to the test when faced with the onslaught of external DNA-damaging agents. Chemotherapeutic drugs and radiation therapy, while aiming to eliminate malignant cells, inadvertently inflict collateral damage on healthy DNA. The intricate dance of DNA repair pathways orchestrates a counteroffensive, striving to rectify the damage inflicted by these therapeutic interventions.
- Essentiality in Development and Aging
The delicate balance of DNA damage and repair extends beyond disease prevention, encompassing vital processes such as development and aging. Stem cells, the architects of growth and regeneration, rely on robust DNA repair mechanisms to preserve their pristine genomes. Moreover, the gradual accumulation of DNA damage over time contributes to the aging process, making the study of DNA Damage and Repair an avenue to unravel the mysteries of time's relentless march.
Available Resources for DNA Damage and Repair
The functions and profound importance of DNA damage and repair cannot be overstated. Their vigilant guardianship of genomic integrity, prevention of diseases including cancer, response to DNA-damaging agents, and influence on development and aging. As we navigate the complexities of this field, let us embrace the resources provided by Creative BioMart, forging a path toward transformative discoveries that illuminate the enigmatic mechanisms of DNA damage and repair. Creative BioMart offers a wealth of products and services related to DNA damage and repair. Our extensive product portfolio spans recombinant proteins, cell and tissue lysates, protein pre-coupled magnetic beads, and assay kits, empowering scientists to delve into the intricacies of DNA repair processes. Additionally, our custom services further bolster research endeavors in the ever-expanding domain of DNA damage and repair.
The following DNA damage and repair-related products are displayed, click to view all molecules/targets and research reagents. Please feel free to contact us with any questions or requests.
References:
- Chatterjee N, Walker GC. Mechanisms of DNA damage, repair, and mutagenesis. Environ Mol Mutagen. 2017 Jun;58(5):235-263.
- Huang R, Zhou P K. DNA damage repair: Historical perspectives, mechanistic pathways and clinical translation for targeted cancer therapy[J]. Signal Transduction and Targeted Therapy, 2021, 6(1): 254.

