NOD-like Receptors and the Inflammasome
Related Symbol Search List
- TLR3
- TLR4
- FOS
- NFKBIB
- IKBKE
- JUN
- JUND
- MAPK11
- MAPK12
- MAPK13
- MAPK14
- MAPK8
- JNK2
- NFKBIA
- REL
- RELA
- CASP1
- CASP4
- IL18
- IL1B
- IL1R1
- IL1RAP
- IRAK4
- MAP2K3
- MAP2K4
- MKK6
- MAP3K7
- MYD88
- NOD1
- PANX1
- PYCARD
- RIPK1
- RALBP1
- TANK
- TICAM2
- TLR1
- TLR10
- TLR2
- TLR5
- TLR6
- TLR7
- TLR9
Immunology Background
About NOD-like Receptors and the Inflammasome
NOD-like receptors (NLRs) are a family of intracellular pattern recognition receptors (PRRs) that play a crucial role in the innate immune system. It contains three main domains: a central nucleotide-binding and oligomerization domain (NOD/NACHT), a C-terminal leucine-rich repeat (LRR) domain involved in ligand sensing, and an N-terminal effector domain. The effector domain determines the downstream signaling and functional outcomes of NLR activation. NLRs are expressed in various cell types, including immune cells such as macrophages, dendritic cells, and neutrophils.
Some NLRs, such as NLRP1, NLRP3, and NLRC4, can form multiprotein complexes called inflammasomes. Inflammasomes are assembled in response to specific stimuli and serve as platforms for the activation of caspase-1, a protease enzyme. Caspase-1 activation leads to the processing and release of pro-inflammatory cytokines IL-1β and IL-18, promoting inflammation and immune responses. Inflammasomes consist of an NLR, the adaptor protein ASC, and the effector molecule pro-caspase-1. Once activated, these inflammasomes facilitate the cleavage and activation of caspase-1, which in turn mediates the cleavage of the pro-inflammatory cytokines IL-1β and IL-18 into their active and secreted forms. Activated caspase-1 also drives the cleavage of gasdermin D, which triggers an inflammatory form of cell death known as pyroptosis. Several NLRs are also known to possess non-canonical, inflammasome-independent functions, regulating a variety of signaling pathways.
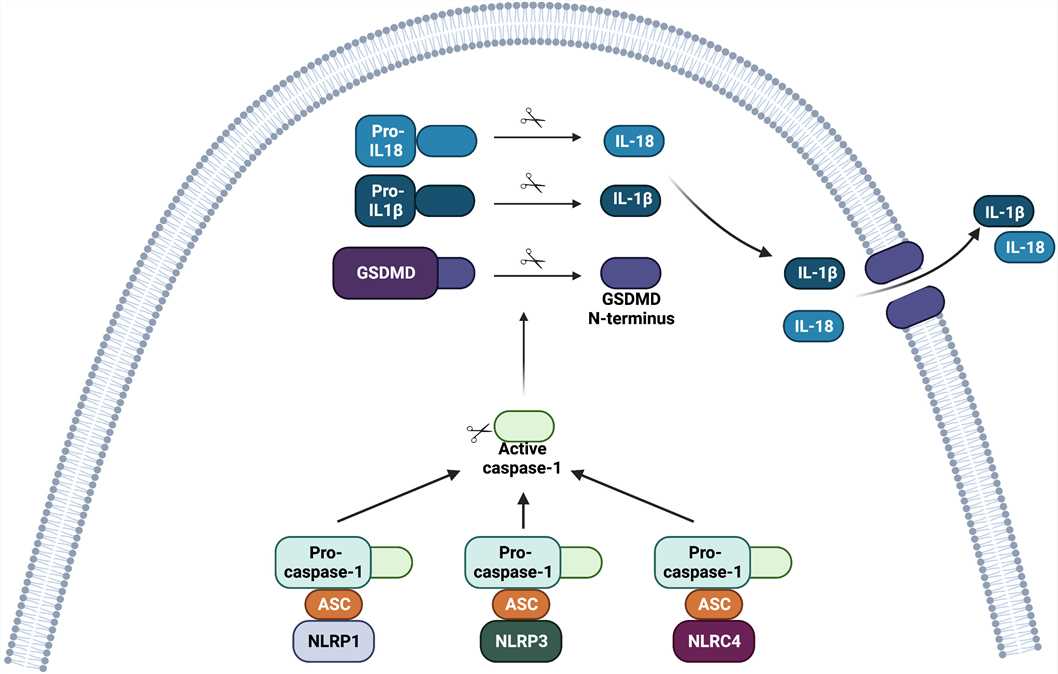 Fig. 1 Structure of the inflammasome complexes involving NOD-like receptors (NLRs). (Almeida-da-Silva CLC, et al., 2023)
Fig. 1 Structure of the inflammasome complexes involving NOD-like receptors (NLRs). (Almeida-da-Silva CLC, et al., 2023)
After ligand recognition, NLRP1, NLRP3 and NLRC4 bind to pro-caspase-1 via the adaptor ASC. The CARD domain of ASC is necessary to recruit pro-caspase-1. Nlrp1b and NLRC4 might also directly bind to pro-caspase-1 since these receptors contain the domain CARD. The assembly of the inflammasome complex leads to its oligomerization. Activation of the inflammasome complex (NLRP1, NLRP3 or NLRC4) ultimately leads to proteolytic activation of pro-caspase-1 into active caspase-1. Caspase-1 then cleaves gasdermin D (GSDMD) into a C-terminal portion and an N-terminal portion. The N-terminal portion inserts into the plasma membrane and oligomerizes to form pores that can lead to a type of cell death called pyroptosis, and contribute to the release of inflammasome-related cytokines. The active form of caspase-1 also cleaves pro-IL-1β and pro-IL-18 into their active forms, IL-1β and IL-18, respectively. The active form of these pro-inflammatory cytokines is then released from the cell.
Signaling Pathways of NOD-like Receptors and Inflammasomes
NOD-like receptors and their associated inflammasomes participate in two distinct signaling pathways: the canonical pathway and the non-canonical pathway. These pathways have unique mechanisms of activation and lead to the production of pro-inflammatory cytokines.
Canonical Signaling Pathway
- Sensing and Activation: In the canonical pathway, NLRs, such as NLRP1, NLRP3, and NLRC4, recognize specific ligands or triggers, which can be microbial components, cellular stress signals, or danger-associated molecular patterns (DAMPs). Ligand binding induces a conformational change in the NLR, allowing it to recruit an adapter protein called ASC (apoptosis-associated speck-like protein containing a CARD).
- Inflammasome Assembly: ASC contains a caspase activation and recruitment domain (CARD) that interacts with the CARD of the NLR, facilitating the formation of a large protein complex called the inflammasome. This assembly provides a platform for the recruitment and activation of caspase-1.
- Caspase-1 Activation: Within the inflammasome, procaspase-1 molecules are brought into close proximity, enabling their autocatalytic cleavage and activation. Active caspase-1 cleaves pro-interleukin-1β (pro-IL-1β) and pro-interleukin-18 (pro-IL-18) into their mature forms, which are then released from the cell.
- Cytokine Release: Mature IL-1β and IL-18 are secreted from the cell and exert their pro-inflammatory effects, promoting immune cell activation, inflammation, and immune responses.
Non-Canonical Signaling Pathway
- Sensing and Activation: The non-canonical pathway is primarily associated with the sensing of intracellular lipopolysaccharide (LPS), a component of the outer membrane of Gram-negative bacteria. This pathway involves a specific NLR called NLRC4 and a cytosolic LPS receptor called caspase-4 (human) or caspase-11 (mouse).
- Direct Activation of Caspases: In the non-canonical pathway, cytosolic LPS directly binds to caspase-4 or caspase-11, inducing their activation. Active caspase-4/11 can then directly cleave gasdermin D (GSDMD), a pore-forming protein.
- Pore Formation and Cytokine Release: Cleavage of GSDMD leads to the formation of membrane pores, resulting in the rapid and uncontrolled release of mature IL-1β, IL-18, and other intracellular molecules. This process is often accompanied by a form of cell death called pyroptosis, which promotes the release of pro-inflammatory signals and amplifies the immune response.
Importance and Implications
The canonical and non-canonical signaling pathways of NLRs and inflammasomes highlight their roles in mediating immune responses and inflammation. Dysregulation of these pathways can lead to excessive or prolonged activation of the inflammasome and the release of pro-inflammatory cytokines, contributing to the pathogenesis of various diseases, including autoinflammatory disorders, infectious diseases, and chronic inflammatory conditions.
Understanding the distinct mechanisms of canonical and non-canonical signaling pathways is crucial for developing targeted therapies aimed at modulating inflammasome activation and cytokine release. Additionally, research into these pathways provides insights into the complex interplay between the innate immune system and disease pathogenesis.
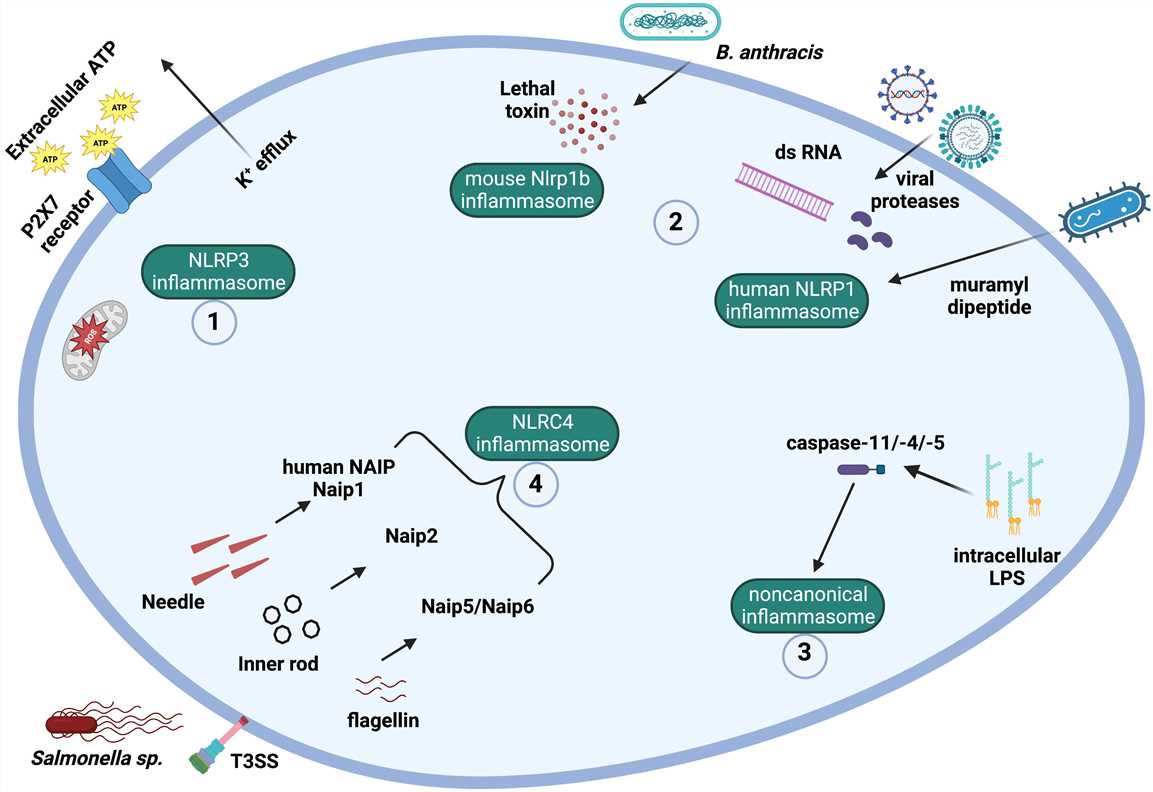 Fig. 2 Mechanisms and ligands involved in canonical and noncanonical inflammasome activation. (Almeida-da-Silva CLC, et al., 2023)
Fig. 2 Mechanisms and ligands involved in canonical and noncanonical inflammasome activation. (Almeida-da-Silva CLC, et al., 2023)
- The NLRP3 inflammasome can be activated by a plethora of signals, including potassium efflux, mitochondrial ROS production, and extracellular ATP ligation to P2X7 receptor.
- Muramyl dipeptide, SARS-CoV-2 infection, viral proteases and double-stranded viral RNA can lead to the activation of the human NLPR1 inflammasome. The lethal toxin produced by Bacillus anthracis activates murine Nlrp1b.
- The noncanonical inflammasome is activated by intracellular LPS from Gram-negative bacteria. Intracellular LPS directly binds to caspase-11 (in mice) or caspase-4/-5 (in humans), which become activated.
- The protein NAIP in humans can recognize T3SS (type III secretion system) needle protein, while the different rodent Naips can recognize different bacterial ligands, such as T3SS needle protein (Naip1), T3SS inner rod protein (Naip2), and flagellin (Naip5 and Naip 6). Following PAMP ligation, NAIPs bind to and activate the NLRC4 inflammasome.
Diseases Associated with Inflammasome-forming NLRs
Several diseases have been associated with dysregulation or mutations in inflammasome-forming NLRs. Here are some examples:
- Cryopyrin-Associated Periodic Syndromes (CAPS): CAPS is a group of autoinflammatory disorders caused by mutations in the NLRP3 gene, resulting in aberrant activation of the NLRP3 inflammasome. This leads to excessive production of IL-1β and systemic inflammation. CAPS includes three clinical phenotypes: familial cold autoinflammatory syndrome (FCAS), Muckle-Wells syndrome (MWS), and neonatal-onset multisystem inflammatory disease (NOMID)/chronic infantile neurological cutaneous and articular syndrome (CINCA).
- Familial Mediterranean Fever (FMF): FMF is an autosomal recessive disorder characterized by recurrent episodes of fever and inflammation. It is caused by mutations in the MEFV gene, which encodes the protein pyrin. Pyrin interacts with the inflammasome component ASC, modulating inflammasome activation. Mutations in MEFV can result in dysregulated pyrin function and exaggerated inflammasome activation.
- Blau Syndrome: Blau syndrome is a rare autosomal dominant inflammatory disorder caused by mutations in the NOD2 gene. NOD2 is an NLR involved in bacterial sensing and inflammasome activation. Mutations in NOD2 lead to constitutive activation of the inflammasome, resulting in chronic inflammation primarily affecting the skin, joints, and eyes.
- Crohn's Disease: Crohn's disease is a chronic inflammatory bowel disease (IBD) characterized by intestinal inflammation. Variants in the NOD2 gene have been associated with an increased risk of developing Crohn's disease. NOD2 mutations may impair bacterial recognition and inflammasome activation, leading to dysregulated immune responses in the gut.
- Rheumatoid Arthritis (RA): RA is a chronic autoimmune disease characterized by joint inflammation. Polymorphisms in the NLRP1 gene have been associated with an increased risk of developing RA. Dysregulated inflammasome activity in RA may contribute to the production of pro-inflammatory cytokines and the perpetuation of joint inflammation.
- Gout: Gout is an inflammatory arthritis caused by the deposition of uric acid crystals in joints. Activation of the NLRP3 inflammasome by uric acid crystals plays a crucial role in inducing inflammation and the release of IL-1β. Dysregulated NLRP3 inflammasome activation in gout can result in excessive inflammation and recurrent flares.
These examples highlight the importance of inflammasome-forming NLRs in the pathogenesis of various inflammatory and autoimmune diseases. Dysregulation or genetic variations in these NLRs can lead to exaggerated inflammasome activation, excessive cytokine production, and chronic inflammation. Understanding the mechanisms underlying inflammasome-associated diseases can aid in the development of targeted therapies aimed at modulating inflammasome activation and managing these conditions.
Available Resources for NOD-like Receptors and the Inflammasome
Creative BioMart offers a wide range of quality tools related to NOD-like receptors and the inflammasome, including recombinant proteins and more. At the same time, we provide personalized services according to the specific needs of our customers. In addition, we provide comprehensive resources, including involved pathways, protein functions, interacting proteins, related articles, and other relevant topics, to help advance research related to NOD-like receptors and inflammasomes.
Our Featured Products
- Recombinant Mouse Tlr3, His tagged
- Recombinant Human MAPK13 protein, GST-tagged
- Recombinant Human JNK2 protein, His-tagged
- Recombinant Human RELA Protein, GST-tagged
- Recombinant Human IL18, His-tagged
- Recombinant Human IL1RAP, His-tagged
- Recombinant Mouse Map2k4, His & GST tagged
- Recombinant Human TLR6, His-tagged
- Active Recombinant Human TLR4/LY96 Complex Protein, His-tagged
- Recombinant Human IKBKE, GST-tagged
- Active Recombinant Human MAPK14 protein, His-tagged
- Recombinant Human NFKBIA, His tagged
- Recombinant Human CASP1 protein, His-tagged
- Active Recombinant Mouse Il1b protein
- Recombinant Mouse Ticam2 Protein, His/GST-tagged
- Recombinant Human TLR2, GST-tagged
- Recombinant Human TLR7, His-tagged
- Recombinant Human JUN, His-tagged
- Recombinant Human MAPK8, GST tagged
- Recombinant Human CASP4 protein, His-tagged
- Active Recombinant Human IL1R1, Fc-tagged, Biotinylated
- Recombinant Rat Ralbp1 Protein, His-tagged
- Recombinant Human TLR1, GST-tagged
- Recombinant Human TLR9 protein
- Recombinant Human MAP2K6 protein, His-tagged
- Recombinant Human RIPK1 protein, GST-tagged
If you have any questions, requirements, or cooperation intentions, please feel free to contact us. We very much look forward to working with you and helping you achieve research and commercial success.
References:
- Almeida-da-Silva CLC, Savio LEB, Coutinho-Silva R, Ojcius DM. The role of NOD-like receptors in innate immunity. Front Immunol. 2023;14:1122586. Published 2023 Mar 15. doi:10.3389/fimmu.2023.1122586
- Platnich JM, Muruve DA. NOD-like receptors and inflammasomes: A review of their canonical and non-canonical signaling pathways. Arch Biochem Biophys. 2019;670:4-14. doi:10.1016/j.abb.2019.02.008
- Ohto U. Activation and regulation mechanisms of NOD-like receptors based on structural biology. Front Immunol. 2022;13:953530. doi:10.3389/fimmu.2022.953530
- Velloso FJ, Trombetta-Lima M, Anschau V, Sogayar MC, Correa RG. NOD-like receptors: major players (and targets) in the interface between innate immunity and cancer. Biosci Rep. 2019;39(4):BSR20181709. doi:10.1042/BSR20181709

