BMP Subfamily
- Related links
- Recombinant Proteins
- Native Proteins
- GMP Proteins
- Fluorescent Dyes
- BMP Subfamily Information
- Activing/inhibin Subfamily
- BMP Subfamily
- Glial Cell-derived Neurotrophic Factor (GNDF) Subfamily
- SMAD Subfamily
- TGF-β Subfamily
- TGF-β Superfamily Receptors
- TGF-β Ligands Structure
- TGF beta Superfamily Information
- TGF-β Superfamily Signaling
TGF Family
Background of Bone morphogenetic proteins (BMPs)
What is BMPs?
Bone morphogenetic proteins (BMPs) are a group of growth factors known as cytokines and as members of the transforming growth factor beta superfamily. They were initially discovered by their ability to induce the formation of bone and cartilage. But more recent research has shown that BMPs are multifunctional proteins that regulate not only bone and cartilage formation but also embryogenesis and morphogenesis of various tissues and organs.
The discovery of BMPs dates back to the 1960s, when orthopaedic surgeon Marshall R. Urist identified an unknown activity in bone that had the ability to induce new bone formation. This activity was eventually isolated and named BMP. Since then, more than 20 structurally related members have been identified as BMPs.
Under the microscope, the structure of BMPs is quite complex. They are usually produced as large precursor proteins and subsequently processed into mature proteins by proteolytic enzymes. The mature proteins are dimeric molecules consisting of two identical or similar polypeptide chains linked via a single disulfide bridge. Despite the structural complexity, scientists have determined that the active site of BMPs is generally contained within an area formed by two loops of the protein chain. This region of the protein can bind to target cells and thereby trigger a cascade of signaling events.
What are the functions of BMPs?
The primary function of BMPs is to promote bone and cartilage formation. When bone is broken or damaged, BMPs are among the first healing factors that the body deploys to the site of the injury. By binding to specific receptors on the surface of bone-forming cells, BMPs kick-start a process known as osteoblast differentiation, which leads to the creation of new bone tissue.
Beyond that, BMPs are also known to play crucial roles in the creation of various tissues and organs during embryonic development. For example, they are believed to control the formation of the heart, kidneys, and various parts of the nervous system. They also regulate the growth and differentiation of various kinds of cells, and thus contribute to the maintenance of tissue homeostasis in adult organisms.
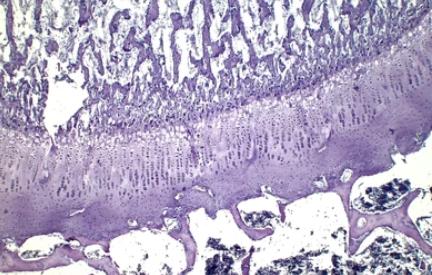
Fig1. Development of bone in the place of the cartilage model.
Meanwhile, due to their potent bone-forming activity, BMPs have been explored for their potential in the treatment of various bone diseases and conditions. For example, they have been used in spinal fusion surgeries to promote bone growth and accelerate healing. They have also been tested for their ability to treat non-healing bone fractures and other skeletal defects.
However, BMPs are far from being wonder drugs. Their use in medical treatments requires careful consideration due to several potential side-effects, including tissue overgrowth and other unintended consequences.
The Classification of BMPs
BMPs are classified into different families according to their structure and function. The largest family is the BMP-2/4 family, which includes BMP-2, BMP-4, BMP-14 (also known as GDF5), and others. Other families include the BMP-5/6/7/8 family, the BMP-9/10 family, and the BMP-11/12/13/14 family (also known as GDFs).
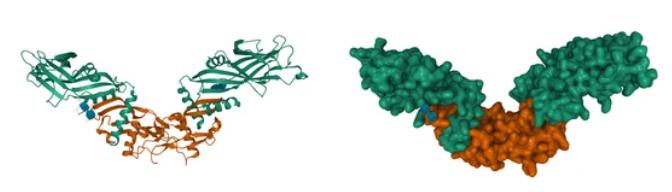
Fig2. BMP2 Stucture
Here is a basic classification:
- BMP-2 & BMP-4: BMP-2 and BMP-4 are the most extensively studied BMPs due to their high osteogenic potential.
- BMP-3: It is sometimes referred to as osteogenin. BMP-3 is taken to be a novel phenotype regulator of mesenchymal progenitor cells.
- BMP-5, BMP-6, BMP-7 & BMP-8: These BMPs are often grouped together with BMP-2 and BMP-4.
- BMP-9: Also known as growth differentiation factor 2 (GDF-2), BMP-9 has been proven to be one of the most potent BMPs to induce orthotopic bone formation.
- BMP-10: BMP-10 is critically involved in heart development.
- BMP-11: Also known as GDF-11, BMP-11 is a myostatin-homologue and holds key roles in retinal development and palate closure.
- BMP-12, BMP-13 & BMP-14: Also known as growth-differentiation factor-7, -6, -5 (GDF-7,-6,-5) respectively, this group of BMPs influence tendon growth and differentiation.
The entire BMP gene family is much larger and encodes more than 20 different proteins that are functionally classified as BMPs, Growth Differentiation Factors (GDFs), Anti-Muellerian Hormone (AMH), Activins, Inhibins and Nodal proteins based on the biological processes they steer and on the structural characteristics they display.
BMPs Related Signal Pathways
They are involved in many cellular processes including cell differentiation, apoptosis, cellular homeostasis and regeneration, embryogenesis, and various signaling pathways, including:
Smad-Dependent Signaling Pathway: BMPs can regulate cell growth and differentiation through the Smad signaling pathway. When BMP ligands bind to their specific receptors, they phosphorylate receptor-regulated Smads (R-Smads), which are then able to form complexes with Smad4. These complexes translocate into the nucleus and control the transcription of target genes.
Mitogen-Activated Protein Kinase (MAPK) Signaling Pathway: BMPs also act in MAPK pathways such as Extracellular Signal-Regulated Kinase (ERK), P38 MAPK, and c-Jun N-terminal kinase (JNK) pathway. Although the role of BMPs in these pathways is not entirely clear, they appear to influence cell proliferation, differentiation, and apoptosis.
PI3K/AKT Pathway: BMPs also regulate the PI3K/AKT signaling pathway, which is crucial for cell survival, cell cycle progression, and cellular growth.
BMP/Wnt Pathway: There is evidence suggesting a cross talk between Wnt and BMP signaling in mediating various biological processes like bone formation. BMP-2 stimulates Wnt signaling from the extracellular matrix and together they regulate osteoblast differentiation and bone formation.
Hedgehog Signaling Pathway: BMPs are known to interact with the Hedgehog signaling pathway and can play a role in organogenesis, tissue polarity, cell proliferation and maintenance of stem cells.
Notch Signaling Pathway: Studies suggest an interaction between BMPs and Notch in controlling cell fate decisions and tissue patterning during embryonic development.
The roles BMPs act in these pathways generally center on supporting the proliferation, differentiation, and survival of cells, particularly in bone and cartilage formation. However, they can also contribute to other processes like embryonic development, tissue repair, and tumor development. The specific actions of BMPs can vary widely depending on the biological context, availability of other signaling molecules, and the presence of specific BMP receptors and antagonists.
Applications of BMPs
Medical applications of BMPs are mainly based on their bone-inducing properties. Here are some of their medical uses:
Bone Healing and Regeneration: This is the primary use of BMPs. They are used in surgeries and treatments that require bone healing or regeneration. In orthopedics, BMPs are used in spinal fusion surgeries and to repair fractured bones that cannot heal properly (nonunions). They are also used in dental surgeries to promote bone growth.
Tissue Engineering: BMPs are used in tissue engineering to generate new bone for use in reconstructive surgery. This involves using the proteins with a scaffold material that will gradually degrade and be replaced by the new bone.
Treatment of Osteoporosis: BMPs might also be used for the treatment of osteoporosis, a disease characterized by reduced bone mass and increased fracture risk.
Wound Healing: Some research suggests that BMPs may facilitate wound healing, including skin wounds and ulcers.
Treatment of Heterotopic ossification (HO): BMPs have a potential application in the treatment of HO, an abnormal growth of bone in the non-skeletal tissues including muscle.
Cartilage Repair: BMPs are under investigation for their capacity to induce the formation and repair of cartilage.
Although the use of BMPs in clinical practice is promising, it is important to note that they can also cause side effects which include inflammation, pain, and unwanted bone growth. Hence, use of BMPs should be under medical supervision.
Case Study
Case 1: Mayweather BA, Buchanan SM, Rubin LL. GDF11 expressed in the adult brain negatively regulates hippocampal neurogenesis. Mol Brain. 2021 Sep 6;14(1):134. doi: 10.1186/s13041-021-00845-z. PMID: 34488822; PMCID: PMC8422669.
Growth differentiation factor 11 (GDF11) is a transforming factor-β superfamily member that functions as a negative regulator of neurogenesis during embryonic development. However, when recombinant GDF11 (rGDF11) is administered systemically in aged mice, it promotes neurogenesis, the opposite of its role during development. The goal of the present study was to reconcile this apparent discrepancy by performing the first detailed investigation into the expression of endogenous GDF11 in the adult brain and its effects on neurogenesis. The findings in this article support the idea that circulating systemic GDF11 and endogenously expressed GDF11 in the adult brain have different target cells or mechanisms of action. Our data describe a role for GDF11-dependent signaling in adult neurogenesis that has implications for how GDF11 may be used to treat CNS disease.
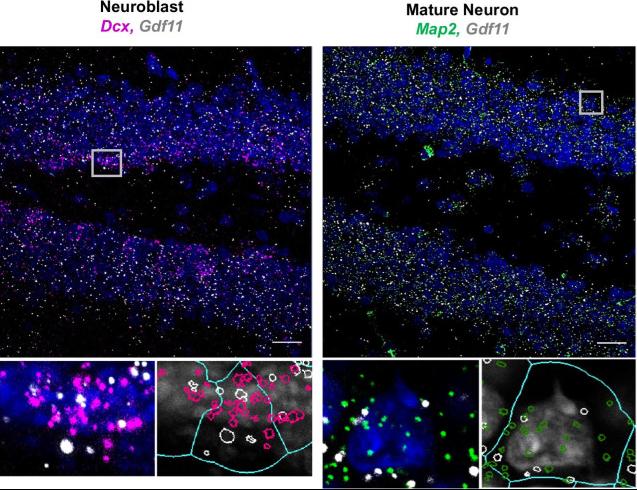
Fig1. Gdf11 is expressed by neuroblasts and mature neurons in the adult dentate gyrus. Representative RNAscope micrographs from a 3-month-old male C57BL/6 mouse probed for Gdf11 (white puncta) cell type specific markers for neuroblasts (Dcx, pink) or mature neurons (Map2, green) and stained with DAPI (blue). Gray boxes mark the regions chosen for the inserts below each micrograph shown. Scale bar = 20 μm. Inserts are magnified DAPI stained nuclei (grey) with each puncta outlined by either white, pink, or green. A light blue overlay outlines the perinuclear area used to determine puncta coexpression.
Case 2: Zhong S, Chen L, Li X, et al. Bmp8a deletion leads to obesity through regulation of lipid metabolism and adipocyte differentiation. Commun Biol. 2023 Aug 8;6(1):824. doi: 10.1038/s42003-023-05194-2. PMID: 37553521; PMCID: PMC10409762.
The role of bone morphogenetic proteins (BMPs) in regulating adipose has recently become a field of interest. However, the underlying mechanism of this effect has not been elucidated. Here this article shows that the anti-fat effect of Bmp8a is mediated by promoting fatty acid oxidation and inhibiting adipocyte differentiation. These findings indicate that Bmp8a plays a critical role in regulating lipid metabolism and adipogenesis, potentially providing a therapeutic approach for obesity and its comorbidities.
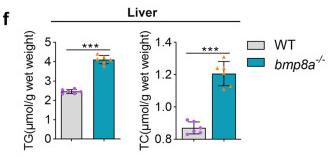
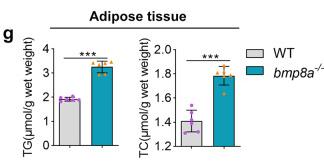
Fig1. Obesogenic phenotype of bmp8a-/- zebrafish. f, g The TG and TC level in WT and bmp8a-/- zebrafish (f, n = 6) or adipose tissue (g, n = 6).
Case 3: Marañón P, Isaza SC, Fernández-García CE, et al. Circulating bone morphogenetic protein 8A is a novel biomarker to predict advanced liver fibrosis. Biomark Res. 2023 Apr 27;11(1):46. doi: 10.1186/s40364-023-00489-2. PMID: 37106416; PMCID: PMC10142503.
Advanced hepatic fibrosis is the main risk factor of liver-related morbidity and mortality in patients with chronic liver disease. In this study, we assessed the potential role of bone morphogenetic protein 8A (BMP8A) as a novel target involved in liver fibrosis progression.
This study provides experimental and clinical evidence indicating that BMP8A is a novel molecular target linked to liver fibrosis and introduces an efficient algorithm based on serum BMP8A levels to screen patients at risk for advanced hepatic fibrosis.
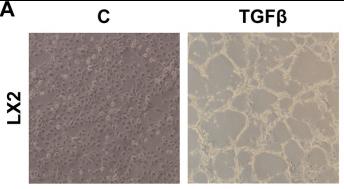
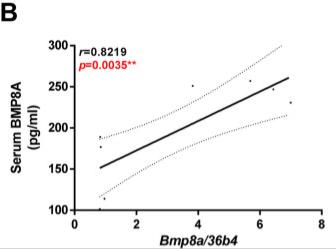
Fig1. TGFβ induces BMP8A expression and secretion in LX2 HSCs and in Huh7 cells. A. Representative phase-contrast images. Circulating BMP8A levels are increased in BDL mice. Data are expressed as pg/ml and presented as mean ± SEM. B. Correlation of matched serum BMP8A levels with hepatic Bmp8a mRNA expression

