Cytokines in Type 2 Immunity
- Related links
- Recombinant Proteins
- Native Proteins
- GMP Proteins
- Fluorescent Dyes
- Cytokines in Type 2 Immunity Information
- Cytokines in Type 2 Immunity
- Cytokine Regulation
- Cytokine Therapy
- Cytokines Signal Transduction
- What is Interferon
- Interferon JAK/STAT Signaling Pathway
- What are Cytokines
Chemokines
Chemokine receptors
Growth Factor & Receptor
Interleukins
Interferons
TNF Superfamily
Jak
STAT
Although multiple other cytokines can participate in type 2 immune responses, the signature cytokines associated with this immune program are IL-4, IL-5, IL-9 and IL-13. The genes for all four of these cytokines are found on the same chromosome in humans, and IL-4, IL-5 and IL-13 are found on the same chromosome in mice. The degree of homology between IL-4 and IL-13 suggests that they originated through a gene duplication event. Research from multiple groups has revealed that, while some of the functions of these cytokines can overlap, they each play important and nonredundant roles in type 2 immune responses. Transgenic overexpression or exogenous administration of each of these cytokines can lead to the development of pulmonary inflammation. The coordinated actions of these cytokines mediate the anti-helminth immune response and can promote the development of allergic lung inflammation.
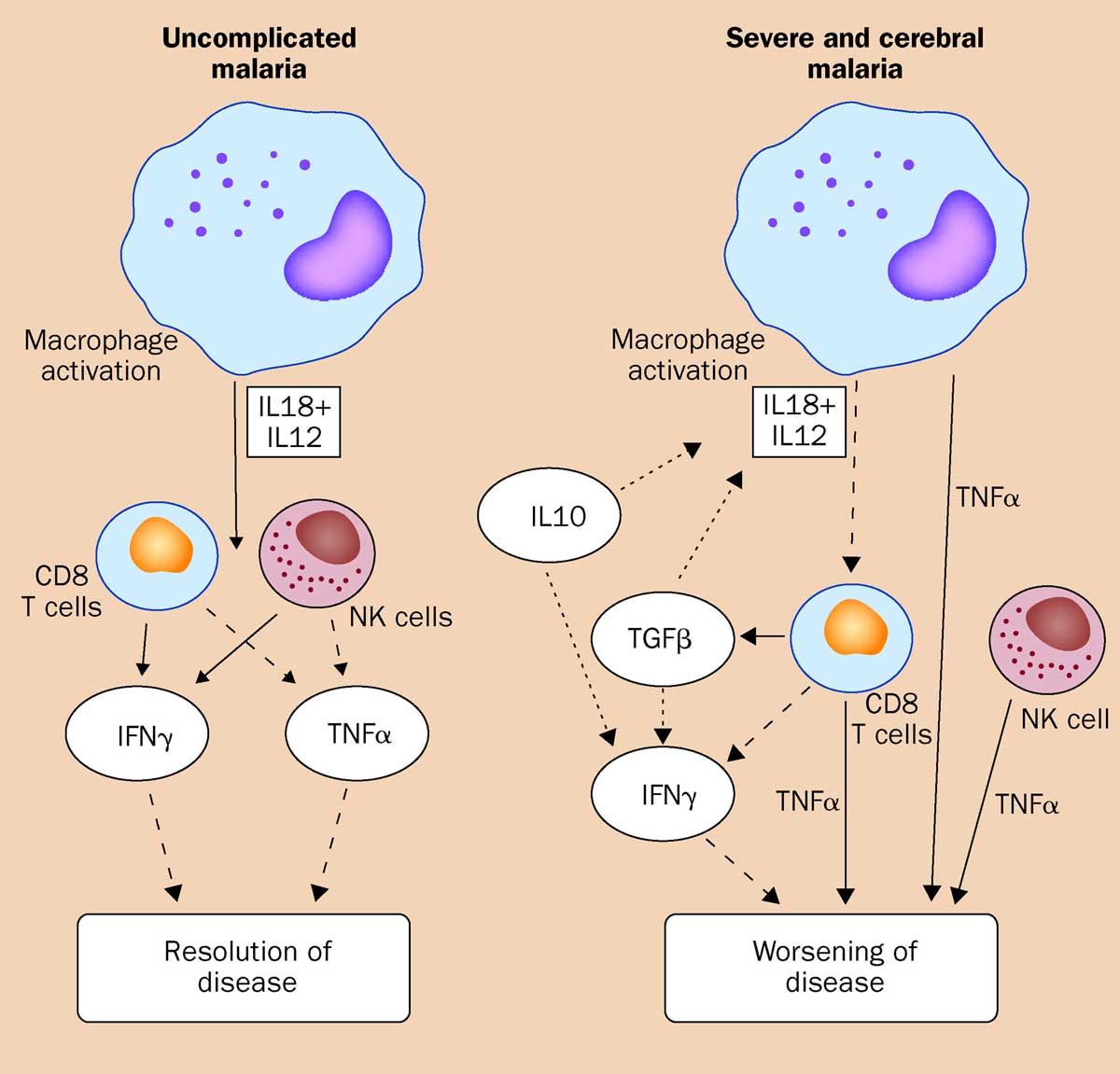 Fig. 1 Cytokines in inflammatory response.
Fig. 1 Cytokines in inflammatory response.
IL-4 is often considered the hallmark cytokine of type 2 immunity. IL-4 can bind both the type I IL-4 receptor, which consists of the IL-4Rα chain paired with the common γ chain, and the type II IL-4/IL-13 receptor, which consists of the IL-4Rα chain paired with the IL-13Rα1 chain. In contrast, IL-13 does not bind the type I IL-4 receptor, but can bind the type II IL-4R or the decoy receptor IL-13Rα2, which is membrane-bound in humans and both membrane-bound and soluble in mice. Type I IL-4 receptor is mainly expressed on hematopoietic cells, while type II IL-4 receptor is expressed ubiquitously, including on nonhematopoietic cells. Binding of IL-4 to IL-4Rα leads to sequential activation of JAK-1 and STAT-6, which leads to the downstream effector functions of this cytokine. Binding of IL-4 to type I IL-4R is associated with promotion of Th2 differentiation and also the switching to IgE and IgG1 antibody isotypes by B cells. Binding of IL-4 or IL-13 to type II IL-4R mediates the tissue manifestations of allergic immunity including goblet cell hyperplasia and mucus secretion, smooth muscle contraction and the development of fibrosis. The actions of IL-4 and IL-13 and type II IL-4R signaling have also been associated with the promotion of macrophage alternative activation. However, it is important to note that differential affinities and binding patterns of IL-4 and IL-13 to the type II IL-4R are such that IL-13 is likely a more potent driver of type II IL-4R signaling. Indeed, despite utilization of the same receptor, multiple groups have demonstrated that IL-13 has multiple nonredundant functions during helminth infection and the development of allergic inflammation.
IL-5 is another cytokine associated with type 2 immune responses. IL-5 signals through a receptor comprised of IL-5Rα and common β chain subunits. Signaling through the IL-5Rα chain is associated with activation of JAK2, STAT-5 and multiple additional downstream signaling pathways. IL-5 is a growth factor for a specific subset of B cells called B1 cells, which have limited B cell receptor diversity and are major sources of IgM. IL-5 transgenic mice display a remarkable increase in the number of B1 cells, resulting in increased serum IgM, IgA and IgE, and IL-5-/-, and IL- 5Rα-/- mice have reduced numbers of B1 cells and half as many IgA-secreting cells in the intraepithelial lymphoid tissue of the intestine. IL-5 is also has multiple effects on eosinophils. IL-5 prolongs eosinophil survival by preventing eosinophil apoptosis, possesses eosinophil chemotactic activity, increases eosinophil adhesion to endothelial cells promoting migration, and also enhances eosinophil effector function. Consequently, IL-5-/- and IL-5Rα-/- also have reduced baseline eosinophil numbers, and the resulting eosinophils appear to be functionally impaired.
IL-9 production is also associated with type 2 immunity. IL-9 signals through a receptor composed of IL-9Rα and common γ chains, and signaling involves activation of JAK-1 and STAT-1, STAT-3 and STAT-5. IL-9 was first characterized as a T cell and mast cell growth factor. However, the role of this cytokine in promotion of T cell growth and function is controversial. IL-9-/- mice have reduced goblet cell hyperplasia and defects in mast cell recruitment in a pulmonary granuloma model. In contrast, IL-9-/- mice display normal levels of lung inflammation and airway hyperreactivity in a standard sensitization model, and have no observable defects in immunity to the helminth Nippostrongylus brasiliensis. Further studies are necessary to fully elucidate the role of IL-9 in type 2 immunity.
A multitude of recent studies have revealed new and important roles for the cytokines IL-25, IL-33, and thymic stromal lymphopoietin (TSLP) in the initiation of type 2 immune responses. Although multiple cellular sources for each of these cytokines have been reported, the majority of studies have focused on their expression by epithelial cells. While epithelial cells were originally viewed as merely constituting a passive barrier to infection or injury, it is becoming increasingly apparent that epithelial cells are crucial and active participants in pathways that maintain mucosal homeostasis and also in innate and adaptive immune responses. Epithelial cells express many genes that allow them to participate in these responses, including major histocompatibility complex (MHC) class I and II, innate immune receptors such as toll-like receptors (TLRs), costimulatory molecules, chemokines, cytokines, and prostaglandins. IL-25, IL- 33 and TSLP are expressed during helminth infection and allergen challenge and over expression of either of these cytokines can lead to the downstream features of a type 2 immune responses, including the production of IL-4, IL-5 and IL-13, increased serum IgE, IgA and IgG1, and tissue manifestations such as goblet cell hyperplasia and mucus production and smooth muscle hyperreactivity.
Reference:
- Dr Donato Torre, Filippo Speranza, Roberto Martegani, Role of proinflammatory and anti-inflammatory cytokines in the immune response to Plasmodium falciparum malaria. PlumX Metrics, 2002, DOI:https://doi.org/10.1016/S1473-3099(02)00449-8

