Cytokine Regulation
- Related links
- Recombinant Proteins
- Native Proteins
- GMP Proteins
- Fluorescent Dyes
- Cytokine Regulation Information
- Cytokines in Type 2 Immunity
- Cytokine Regulation
- Cytokine Therapy
- Cytokines Signal Transduction
- What is Interferon
- Interferon JAK/STAT Signaling Pathway
- What are Cytokines
Chemokines
Chemokine receptors
Growth Factor & Receptor
Interleukins
Interferons
TNF Superfamily
Jak
STAT
Due to the importance of cytokines as immune modulators, cells tightly regulate cytokine-mediated effects at the level of cytokine production, receptor expression, signal transduction, and signal modulation. There are many mechanisms that inhibit cytokine signaling pathways including: phosphatases, protein inhibitors of activated STATs (PIAS), and suppressors of cytokine signaling (SOCS). The SHP phosphatases and SHIP phosphatases play important roles in deactivation of many cytokine signaling pathways to prevent constant activation, but are not cytokine-specific. The PIAS group of proteins has four members, PIAS1, PIAS3, PIASx, and PIASy. PIAS proteins not only inhibit STAT binding to cytokine receptor chains, but are also capable of other mechanisms of inhibition, such as through E3 ubiquitin ligase activity.
The best known inhibitors of cytokine signaling are the SOCS proteins, which will be discussed in more detail below.
SOCS Family
The SOCS family (also known as CIS/SSI/JAB) consists of eight members, SOCS1-7 and CIS, which are characterized by a central SH2 domain and C-terminal SOCS box. Other proteins also contain SOCS boxes, such as Von-Hippel-Lindau protein (VHL) and Elongin A, and these domains allow E3 ligase complex formation, which promotes ubiquitination and proteasomal degradation. Thus, SOCS proteins can act as adaptors to induce degradation of a variety of targets, including receptors, Jaks, and STATs, leading to inhibition of cytokine signaling. In addition, SOCS proteins can bind to signaling molecules directly leading to their inhibition without degradation. The mechanism of suppression depends upon the particular SOCS protein-signaling molecule pairing.
The best studied SOCS proteins are CIS, SOCS1, SOCS2, SOCS3, and, to a lesser extent, SOCS5. These proteins will be generally reviewed, and a later section will be devoted solely to SOCS3. CIS is induced by a number of cytokines, many of which have receptors whose signaling is also inhibited by CIS, such as IL-2, IL-3, erythropoietin (EPO), GH, and prolactin. All of these cytokines signal through STAT5, and CIS inhibits STAT5 activation by blocking STAT5 docking sites. The phenotype of STAT5-/- mice and animals that over-express CIS are similar, each showing defects in growth hormone signaling and mammary gland development. On the other hand, CIS-deficient animals are phenotypically normal, suggesting possible compensatory mechanisms to downregulate STAT5 signaling.
Although it can inhibit some of the same cytokines as CIS, SOCS1 is best known as a critical inhibitor of IFNγ signaling. Thus, SOCS1-deficient animals die by one month of age due to severe, systemic inflammation. The phenotype can be significantly improved, although not eliminated, by administration of neutralizing antibody to IFNγ, or by simultaneous deletion of the gene for IFNγ. SOCS1/RAG2 double knockout mice, which lack Th cells, have significantly reduced pathology and lengthened lifespans, further strengthening the conclusion that SOCS1 is critical for inhibition of IFNγ signaling in Th cells. SOCS1 acts by inhibition of Jaks and other molecules activated by cytokines, such as IRS1. SOCS1 also inhibits signaling by IL-4 and IL-12, and SOCS1-deficient Th cells show more extreme Th1 or Th2 biasing than cells from normal mice upon treatment with either cytokine.
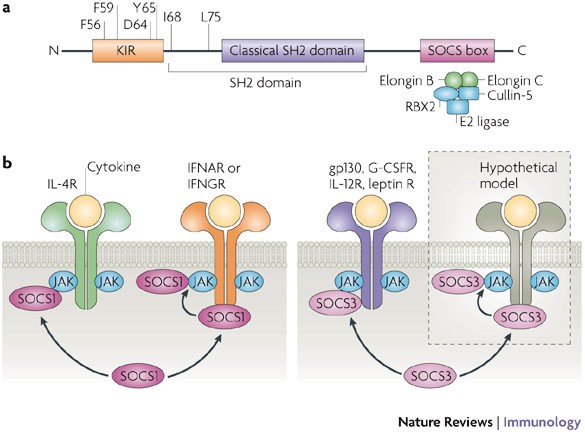 Fig. 1 Cytokines inhibited by SOCS and cytokines that induce SOCS expression.
Fig. 1 Cytokines inhibited by SOCS and cytokines that induce SOCS expression.
Less is known about the role of SOCS2 in the immune system, and the most thoroughly described effect of SOCS2 is its inhibition of STAT5 activation following growth hormone (GH) stimulation. Additionally, recent work has suggested that SOCS2 may be important for Treg development and/or function, since microarray analyses have shown increased SOCS2 expression in Tregs, compared to Th cells. This is supported by data demonstrating that over-expression of FoxP3 in Th cells is associated with upregulation of SOCS2. Another potentially important function of SOCS2 is the enhanced degradation of SOCS3 in the presence of IL-2.
Unlike the other SOCS proteins, which do not seem to skew Th cell differentiation towards a particular lineage, SOCS3 and SOCS5 are associated with Th2 and Th1 responses, respectively. Over-expression of SOCS5 seems to promote Th1 biasing in mice by binding to the IL-4R and inhibiting IL-4 signaling. However, SOCS5-deficient Th cells appear to differentiate into Th1 and Th2 cells normally, so the importance of SOCS5 in vivo remains unclear.
Reference:
- Yoshimura, A., Naka, T., & Kubo, M. (2007). SOCS proteins, cytokine signalling and immune regulation. Nature Reviews Immunology, 7(6), 454-465. https://doi.org/10.1038/nri2093

