Cytokines Signal Transduction
- Related links
- Recombinant Proteins
- Native Proteins
- GMP Proteins
- Fluorescent Dyes
- Cytokines Signal Transduction Information
- Cytokines in Type 2 Immunity
- Cytokine Regulation
- Cytokine Therapy
- Cytokines Signal Transduction
- What is Interferon
- Interferon JAK/STAT Signaling Pathway
- What are Cytokines
Chemokines
Chemokine receptors
Growth Factor & Receptor
Interleukins
Interferons
TNF Superfamily
Jak
STAT
Cytokines Signal Transduction
Most cytokines signal primarily through activation of STATs, although the vast majority of cytokines also signal through additional pathways, which are less studied, such as insulin receptor substrate (IRS) proteins, phosphatidylinositol 3 kinase (PI3K), and the Ras/mitogen activated protein (MAP) kinase cascade. However, a few cytokines signal through unique transduction pathways. Among these, TGFβ, which signals through the Smad pathway, is one of the most important, and is involved in the present work.
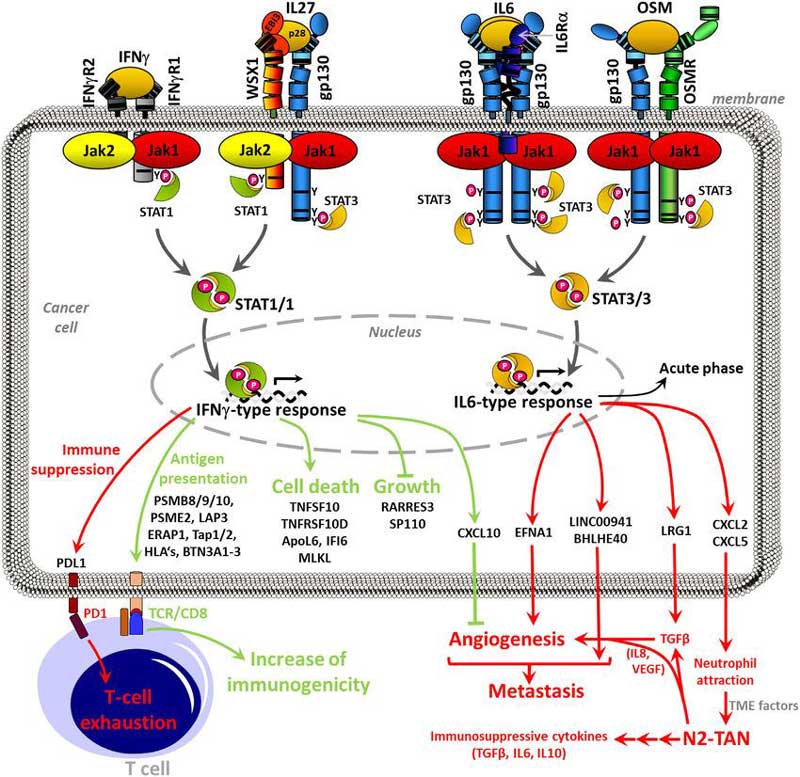 Fig. 1 Cytokine signaling.
Fig. 1 Cytokine signaling.
STAT-dependent cytokine signaling is initiated upon cytokine binding to a specific receptor, which causes association of Janus protein kinases (Jaks) with the intracellular portion of cytokine-receptor chains. Jaks can phosphorylate other Jaks as well as docking sites for many other proteins on the receptor cytoplasmic tail. STAT proteins bind to the receptor, in the domains phosphorlyated by Jaks. Upon binding, STATs are themselves phosphorylated by the Jaks. The phosphorylated STATs (pSTATs) form homodimers or heterodimers, and translocate to the nucleus. Until recent years, only dimerized pSTATs were considered to influence transcription. However, increasing evidence suggests that some unphosphorylated, monomeric, STATs can also translocate to the nucleus and influence gene expression. Still, the importance of unactivated STAT monomers in vivo remains poorly understood.
Four Jak proteins, Jak1, Jak2, Jak3, and Tyk2, and seven STAT proteins, STAT1- STAT4, STAT5a, STAT5b, and STAT6, are expressed in mammals. Since many cells express all four Jaks, and several cytokines recruit the same Jaks to their receptors, cytokine specificity is conferred primarily by differential recruitment of particular STATs to cytokine receptors. For instance, both Jak1 and Jak3 are associated with the IL-2 and IL-4 receptors, but IL-2 primarily activates STAT5a and STAT5b, whereas IL-4 mainly causes STAT6 phosphorylation. An additional level of signaling complexity is found for several cytokines, such as IL-6, which activate multiple STATs, in this case STAT1 and STAT3. This opens the possibility of both homodimer and hererodimer formation. The ratios and compositions of the STAT1 and STAT3 dimers are clearly important because cytokines with very different effects can activate the same STATs. For instance, IL-10, an immunosuppressive cytokine, activates STAT3, but IFNγ, which is a potent immune activator, signals primarily through STAT1. Interestingly, IL-6 can be pro-inflammatory or anti-inflammatory depending on the situation, and these effects could be due to activation of STAT1 and STAT3, respectively.
In addition to different ratios of STATs, cytokines exert specific effects through many other pathways, such as insulin receptor substrate (IRS) and phosphatidylinositol 3 kinase (PI3K) activation, and the Ras/ mitogen associated protein (MAP) kinase pathway. The IRS pathway will be described briefly due to its involvement in IL-4 signaling, which is an important component of the investigations described in this document. Upon stimulation by cytokines, such as IL-4, the receptor is activated and phosphorylated at key tyrosine residues that allow recruitment of IRS proteins. After binding to the cytokine receptor, IRS proteins are phosphorylated, and serve as adaptors by recruiting many other molecules, especially PI3K, which reportedly activates Akt, promoting cell survival and inhibition of pro-apoptotic c-Jun N-terminal kinase 2 activation in a Th cell line. Others found that IRS2 was unnecessary for the anti-apoptotic effects of IL-4, but was involved in IL-4-induced proliferation. This was confirmed by studies in mice of a mutated IL-4R that lacked the tyrosine required for IRS2 recruitment, but was still able to activate STAT6. Upon stimulation, Th cells with the mutant receptor proliferated significantly less in response to IL-4 than cells with a WT receptor. Therefore, IL-4 has a reduced ability to induce proliferation in the absence of IRS activation. Thus, IL-4 effects on proliferation may be less dependent on STAT6 than are other IL-4-mediated responses.
In contrast to the IRS2, PI3K, and Ras pathways described above, which signal concurrently with STATs, receptors that signal through Smads may not transduce signals through STATs. One of the few cytokines that utilizes the Smad pathway is TGFβ. Upon ligand binding, the TGFβR forms a multimer of four separate chains, which have intrinsic kinase activity. The complex undergoes autophosphorylation, and this enables binding of either Smad2 or Smad3. After recruitment to the receptor, Smad2 and Smad3 are themselves phosphorylated. They then form homodimers or heterodimers, which bind to Smad4 to form a trimer. The Smad2/3-Smad4 complex enters the nucleus to modulate transcription. In the case of BMP receptors, Smad1, Smad5, and Smad8 are activated, and form complexes with Smad4, which translocate to the nucleus and influence transcription. Unlike STATs, which act primarily as transcription factors, the Smad proteins include two members that function as competitive inhibitors, Smad6 and Smad7. Although Smads transduce signals for relatively few cytokines, the importance of TGFβ has increased their profile as important modulators of immunity.
Reference:
- Kirchmeyer, Melanie & Servais, Florence & Ginolhac, Aurélien & Nazarov, Petr & Margue, Christiane & Philippidou, Demetra & Nicot, Nathalie & Behrmann, Iris & Haan, Claude & Kreis, Stephanie. (2020). Systematic Transcriptional Profiling of Responses to STAT1- and STAT3-Activating Cytokines in Different Cancer Types. Journal of molecular biology. 432. 10.1016/j.jmb.2020.09.011.

